Document 10409921
advertisement

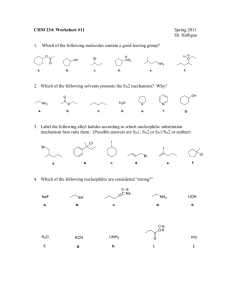
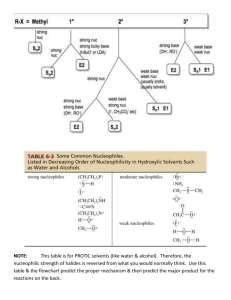
Trends in Alkyl Substitution and Elimination Reactions Nucleophile/Base Weak Bases/ Strong Nucleophiles Uncharged Nucleophiles Strong Bases (pKas > 15) (-9 < pKa <10) - - - - (-2 < pKa < 15) - (I , Br , SCN , N3 , CH3COO , RS , CN , etc) Alkyl Chain Mainly SN2 reactions. Neighboring carbon atoms (on the β-carbon) reduce the rate of substitution, i.e., neopentyl chloride does not undergo SN2 reactions. Primary R-CH2-X Secondary ( H3C C C -­‐ -­‐ -­‐ ,CH3O , OH , NH2 , -­‐ (C3H7)2NLi, (CH3)3CO ) (H2O, ROH, RSH, R3N, NH3) Mostly SN2 but some E2 products. E2 reactions predominate when conditions like heat are used or with highly substituted βcarbon atoms. Elimination reactions are favored by the use of strong bulky bases like, i.e. tert-butoxy. Mainly E2. Use of strong bulky bases like, i.e., tertbutoxy favor the formation of the least substituted alkene – Hoffman rule. SN2 reactions. (Nucleophilic activity order: N ≈ S >> O) Almost no substitution reactions. Mainly E2 even with weak bases. Mainly E2. Only traces of substitution, if any. SN1 in polar protic solvents – solvolysis (H2O, CH3OH). E2 with nucleophiles that have nitrogen. Heat favors E1 reactions. Expect SN2 reactions with ease in primary benzylic compounds. Expect also some SN1. SN2 expected in primary benzylic compounds. E2 in secondary benzylic compounds with β-hydrogen atoms. In polar protic solvents expect SN1 with primary benzylic compounds. SN2 reactions with ease with primary allylic compounds. Expect also some SN1. SN2 reaction in primary allylic compounds. E2 in secondary allylic with βhydrogen atoms. In polar protic solvents expect SN1 with primary allylic compounds. Expect SN2 for primary benzylic compounds and nucleophiles with nitrogen or sulfur. Solvolysis (SN1) mainly occurs in polar protic solvents and primary benzylic compounds. Secondary benzylic halides give primarily E2, some E1 also expected. Expect SN2 for primary allylic compounds and nucleophiles with N or S. Solvolysis (SN1) in polar protic solvents and primary allylic compounds. Secondary allylic halides give mainly E2 products, some E1 expected. SN2 & E2. Bases weaker than acetate (pKa < 4.8) give higher substitution rates. Highly branched alkyl compounds provide greater elimination rates. Mostly SN2. Polar protic solvents (H2O, CH3OH, etc.) favor SN1 & E1. Polar aprotic solvents (acetone, DMSO, etc.) favor SN2 & E2. Heat favors E2. Tertiary Benzylic Allylic Prepared by José Laboy