The colloidal state
advertisement

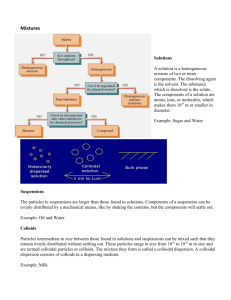
The colloidal state Solids with particle size in the range of 10-7 – 10-4 cm are said to be in the colloidal state. Such articles are too small to be visible to the naked eye. They have particle size which is intermediate between that in a true solution and particles in precipitate. A suspension of a colloidal particles in a liquid scatter light so that the solution appears cloudy when seen at right angles to the beam of light passing through it (i.e. scattering of light falling on large sized particles). In true solution, the solute particles are present as molecules or ions giving a homogenous mixture consisting of a single phase. In colloidal solution, the unit of particles of dissolved substance are either very large molecules (starch, gelatin) or aggregate of large number of molecules. The colloidal solution consists of dispersed phase (small concentration) and dispersion medium (large concentration) like solvent. Properties of colloids 1- The particles of the dispersed phase are relatively large, however they pass through ordinary filter media. 2- The dispersed phase don't dissolve in the dispersion medium. 3- They scatter light (Tyndal effect). 4- Particles show random motion (Brownian motion), due to collision with molecules of the dispersion medium. 5- Particles adsorb ions (its own ions in preference to others). 6- Particles may have an electrical charge which lead to repulsive forces which stabilize the colloid dispersion and prevent its coagulation. 7- When the particles of the dispersion phase join together, they coagulate and separate due to gravity. 8- Particles have large surface area. 9- Colloidal suspensions have negligible effects on colligative properties. 59 ∗ Terminology and Classification 1- Lyophilic colloids (solvent loving). They are so called because of affinity of particles for the dispersion medium. Solutions of lyophiles are prepared by simply dissolving the material in the solvent. Because f attraction between the dispersed phase and dispersion medium, salvation (hydration in case of water) of the particles occur. Most of these colloids are organic n nature e.g. gelatin, acacia, insulin, albumin. The solution is viscous because of strong affinity for water (called gels). 2- Lyophobic colloid The dispersed phase has little attraction to the solvent (solvent hating). Their properties differ from the lyophilic (hydrophilic). They are usually inorganic n nature e.g. gold, silver, sulphur. In contrast to lyophilic colloid, it is necessary to use special method to prepare hydrophobic colloid. a) Dispersion (reduce particle size to colloidal dimentions using high density ultrasonic generators of colloidal mill etc.). b) Condensation from materials in the subcolloidal dimentions which are caused to aggregate into particles in the colloidal state or condensation reactions e.g. hydrolysis, oxidation reduction etc. They are stabilized by an acquiring a charge. 3) Association colloids (Amphophiles) (Accumulate on surface between 2 fluids). These are surfactants. Their molecules have two opposing solution affinities (lypophilic part and hydrophilic part). When these are present in a liquid at low concentration they exist separately in subcolloidal dimentions. If the concentration is increased, aggregation occurs over a narrow range (50 monomer or more), thus forming what is called micelles. The concentration at which micelle is formed is the critical micelle concentration. 60 ∗ Example of pharmaceutical colloids Colloidal AgCl, AgI, Ag proteinate (effective germicide), colloidal sulphur. Many natural and synthetic polymers are important n pharma-ceutical practice. Polymers: These are macromolecules formed by polymerization or condensation of small non-colloidal molecules e.g. proteins, natural colloids, plasma proteins which are responsible for binding certain drug molecules so that the pharmacological action of the drug molecule is affected by them. Starch and hydroxymethylallulose, cyclodeztrin are also examples. ∗ Stability of colloidal system The colloidal particles are not stable in the dispersion medium. The particles of colloidal substance attain their stability in solution if they don't adhere to each other and coagulate. The surface of the particle is covered by different substances : a) In the dispersed phase particles are lyophilic (hydrophile in case of water). Stabilization is brought about by surrounding the particle with a protective solvent sheath that prevents mutual adherence when the particle collide as a result of Brownian motion. b) If the dispersed phase is lyophobic e.g. (hydrophobe; heats water). The particles are stable only in presence of an electrolyte that be adsorbed on the surface in the form of either positive or negative charge, so as the particles become similarly charged so they don't coagulate. The stability may increase by collecting solvent molecules around them after adsorbing ions (salvation) i.e. two stabilizing layers. The amount fo electrolyte must be of certain value that is necessary for maximum adsorption (if less no stability). 61 If large amount of electrolyte is used this would cause coagulation of the hydrophonic particles. ∗ Purification of colloidal solution 1- Dialysis True solutions pass through semipermeable membrane (e.g. Parchment, Collodin,) while colloidal particles not. They pore size of the semipermeable membrane permits the passage of small molecules or ions such as sugar, urea, NaCl pass. Bag of dialyzing medium Water (water, ions and small molecules that has dialyzed water out) The colloid is placed in a plastic bag and the bag is suspended n large water bath in which water is constantly changing. This process a slow, and it can be accelerated by applying potential difference to attract ions (electrodialysis). water → Sol + + electrolyte - solution Dialyzing membrane (2) Ultrafilteration colloidal particles pas through ordinary filter paper, so filtration is conducted under suction (negative pressure) through a dialysis 62 membrane supported in Buchner funnel using unglazed porcelain as filter media. (3) Electrophoresis As the lyophile particles are surrounded by ions. Therefore the colloidal particles will move to the cathode or anode under influence of applied potential difference. (-) (+) Colloidal soln. The electrophoresis cell, fitted with two electrodes contain the colloidal solution. The rate of particle migration is observed by means of an ultramicroscope. The motion of colloidal particles under effect of electric field is used to known the type of charge carried on the surface of the particle. Examples of negative colloids : metal sulphides, gum, starch, and for positive colloids are ferric hydroxice, aluminum hydroxide chromium hydroxide, etc. Also electrophoresis is used to separate the colloidal solution because the colloids will form several fronts according to the charge density (used to purify colloids). Sometimes the charge of the particles is due to part of the colloid itself (not adsorbed on the surface) e.g. protein (polyamino acids) molecules. _ + H2N COO NH3+ _ _ OOO H3N + NH3+ COO COO _ 63 Proteins at different pH values will move to positive or negative pole. At certain pH it don't move (isoelectric point) at which the protein in neutral). It is pH at which protein is neutal. ∗ Methods of Preparing Colloids Condensation methods Dispersion methods True colloid coagulated True colloid coagulated soln. solution colloid sol. solution colloid → ← 1- Condensation method We start with ions or molecules, then convert them to large article by chemical reaction . e.g. (a) Oxidation : H2S + ½ O2 → H2O + S ↓ (colloid) S atoms collect together to give colloidal suspension (b) Reduction : Ag2O + CO (or H2) → 2Ag + CO2 (or H2O) (colloid) © Hydrolysis : Fe3+ + 3H2O → Fe (OH)3 + 3Hcolloid (d) Solvent exchange : Addition of a new will solvent (in which the dispersed phase is not freely soluble), this will cause separation of dispersed phase in a colloidal dimention. 2- Dispersion methods (Large particles : coagulated or solid → colloid) a) Mechanical dispersion : disintegratin using colloidal mill. The substance is ground with the solvent and electrolyte (If it lyphobe). disintegration is possible. 64 Also electrical (b) Peptization : By electrolyte if the substance is lyphobe it is precipitated because there is no enough electrolyte. On adding electrolyte it can be changed to colloid again. FeCl3 + 3NH4=OH → Fe(OH)3 → Fe(OH)3)Fe3+ Peptization by washing with water of a coagulated colloid (e.g. washing with water (hydrophile). In case of hydrophobe added solvent and electrolyte will cause colloid formation. Addition of electrolyte to gel (hydrophilic colloid) may coagulate the colloid by salting out. ∗ Coagulation of colloids For lyophobe: remove adsorbed electrolyte, by washing or adding another electrolyte to neutralize adsorbed electrolyte. For lyophile: (1) Cause particles t move at large speed so the adsorbed solvent molecules are removed so the particles will adhere to each other (e.g. ultra-centrifugation). (2) Increase temperature so that the particles gain kinetic energy and move faster and colloid and adhere together to coagulate. Also adsorbed solvent gain energy, so separates leading to coagulation. ∗ Pharmaceutical application of colloids 1- Protein binding Drugs are bound to plasma proteins forming complexes or products that reliberate the drugs at different rates. Positively changed drugs and negatively charged drugs and negatively charged are bound by different mechanisms. This can be tested by examining binding effect of γ globulins to drugs at certain pH (depending on the nature on the nature of drug). Then drug is separated by dialysis to estimate the extent of drug-protein binding. 65 2- Colloids in which the particles of a liquid are dispersed in another liquid is called emulsion (e.g. colloidal suspension of oil in water (o/w or w/o emlsion). These are only formed in the presence of an emulsifying agent. Emulsifying agents form a protective layer on the surface of each colloidal particle. Unless the emulsifying agent is added, the proplets would combine and the two liquids separate. The emulsifying agent contains a hydrophobic part and hydrophilic part (amphiphile). Example : milk is an emulsion o.w, casein is emulsying agent; mayonnaise is o/w emulsion, egg yolk is an emulsifying agent. Other examples are liquorice, Tr quallia, Tweens, polymers containing amphiphilic parts are also surfactants. 4- Detergents the simples is soaps. E.g. C17H35 COO- Na+ ←→ ←→ long chain HC water soluble (oil soluble) Oily and greasy dirts adhere firmly to clothes and using soap which is an emulsifying agents brings about dirt into colloidal dispersion. The hydrocarbon end of the soap is soluble in oil or grease, the anion end is insoluble in oil or grease, protrudes from colloid drops making them soluble in water and repelling other drops, thus preventing coalescence. O _______________ (oil soluble) HO oil H2O ________________ (H2O soluble head) far soluble tail) Synthetic detergents using sulponic acid derivatives. 66 SO3-Na+ straight chain SO3-Na+ branched sod. alkyl benzene sulphonate 67 68