0.2 Macromolecules - SandyBiology1-2
advertisement

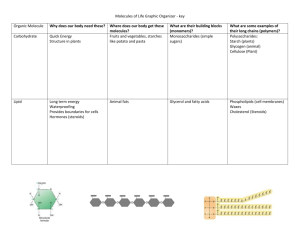
Introduction to Organic Macromolecules Organic Macromolecules • Large molecules • All contain carbon , hydrogen and oxygen • Some contain other elements – eg phosphorus or nitrogen Monomers the building blocks of more complex molecules, called polymers. Monomers water water Dehydration reaction Water molecules removed from monomers bind them to form polymers + + Monomers the building blocks of more complex molecules, called polymers. water water Dehydration reaction Water molecules removed from monomers bind them to form polymers Polymer consist of repeating molecular units which usually are joined by covalent bonds. + + Polymer water Hydrolysis The addition of water molecules breaks the bonds in a polymer Monomers water Carbohydrates (CHO) Saccharides = sugars 5C 6C Monosaccharides = single/simple sugars Disaccharides = double sugar Polysaccharide = many/complex sugars Carbohydrates - Starch: • is a storage polysaccharide composed entirely of glucose monomers Long chain of glucose molecules 200-500 units • Used as an energy store in plants. • Not soluble. • Forms solid grains inside plant cells (often inside chloroplasts). • The chains coil up into a basic spiral shape making the molecules compact. • Hydrogen bonds hold the polysaccharide chain in the compact spiral shape. Carbohydrates -Glycogen Carbohydrates- Cellulose • Most abundant organic molecule. • 300-10,000 + glucose units • It is very slow to decompose. • 20-40% of the plant cell wall. • Hydrogen bonding between monosaccharide molecules in the chain gives strength. • Hydrogen bonding between cellulose molecules cause bundles called microfibrils to develop. These are held together in fibres. • A cell wall will have several layers of fibres running in different directions gives great strength almost equal to steel. • Provides support in plants and stops plant cells bursting. • Freely permeable to water and solutes. Lipids ( CHO[P]) : are built with glycerol and fatty acids • Structure: – glycerol (3C alcohol) + fatty acid fatty acid = long HC “tail” with COOH group at “head” enzyme dehydration synthesis Lipids:Building Fats • Triacylglycerol – 3 fatty acids linked to glycerol – ester linkage = between OH & COOH Lipids:Saturated fats • All C bonded to H • No C=C double bonds – long, straight chain – most animal fats – solid at room temp. • contributes to cardiovascular disease (atherosclerosis) = plaque deposits Lipids:Phospholipids • Hydrophobic or hydrophilic? – fatty acid tails = hydrophobic – PO4 = hydrophilic head – dual “personality” interaction with H2O is complex & very important! Lipids: Cholesterol • Important cell component – animal cell membranes – precursor of all other steroids • including vertebrate sex hormones – high levels in blood may contribute to cardiovascular disease Lipids:Cholesterol Important component of cell membrane helps keep cell membranes fluid & flexible Amino Acids • Are the building blocks of proteins. • Contain a carboxylic acid group and an amino group on the alpha () carbon. • Each contain a different side group (R). R │ H2N—C —COOH AA Proteins or polypeptides are built with amino acids (CHON[S]) 18 Summary of Protein Structures Nucleic Acids are built with nucleotides (NCHOP) DNA IS MADE OF TWO STRANDS OF POLYNUCLEOTIDE Hydrogen bonds P G C P P C G P P C G P P A T P P T A P P T A P