Balancing Equations
advertisement

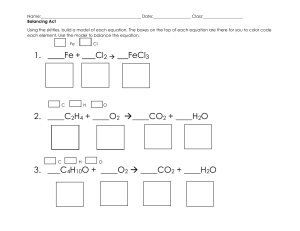
Balancing Equations Step 1 • Write the skeleton equation. Step 2 • Balance each element by placing coefficients in front of the chemical formulas. • Balance in this order: – metals – non-metals – hydrogen – oxygen last “M”et a “n”on “h”airy “o”xen Step 3 • Select coefficients that will give the same number of atoms on each side of the equation. • If you have an odd number of oxygen atoms, multiply the coefficient by 2 first to make it an even number. Then try to balance the equation. Step 4 • Check all other elements to see whether the equation is balanced. • Other elements may have become unbalanced. • Make adjustments to balance all the elements. Tips for Balancing Hydrocarbons • If you have a hydrocarbon reaction such that when you divide the subscript for the number of hydrogens by two and the answer is odd or a fraction, then first start the balancing process out by making the hydrocarbon’s coefficient two. • Ex: 2 C2H6 +7O2 4 CO2 + 6 H2O (6 ÷ 2 = 3, therefore multiply the hydrocarbon by 2 before balancing.) Next, balance the CO2, then H2O, then O2. Balance these equations. 1. HgO 2. Mg 3. KClO3 4. AgNO3 + H2S + O2 Hg + O2 MgO KCl + O2 Ag2S + HNO3 Balance these equations. 5. Al(OH)3 + H2SO4 Al2(SO4)3 + H2O 6. Zn(OH)2 + H3PO4 Zn3(PO4)2 + H2O 7. FeCl3 NaOH + Fe(OH)3 + NaCl 8. 2C3H6O3 + 6 CO2 + 6 H2O 6 O2 Begin your Balancing worksheet-finish for hw