Water Potential student
advertisement

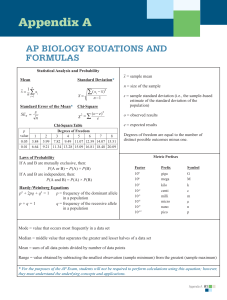
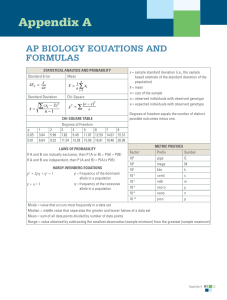
Water Potential What you need to know! • The role of diffusion (osmosis), active transport, and bulk flow in the movement of water and nutrients in plants. • How water potential explains transpiration. Water Potential Ψ • Potential Energy of Water • Determines direction of water flow • Determined by two factors: pressure potential (ΨP) and solute potential (ΨS). • The most common unit for water potential problems is bars. • The mathematical equation is: Ψ = ΨP + ΨS Pressure Potential ΨP • Pressure potential will always be provided for you – Open systems are systems w/out pressure potential – Often cell walls will provide pressure to a cellular example • When not given you may assume it is an open system – ΨP = 0 bars Solute Potential ΨS • ΨS = -iCRT • i = ionization constant – Measure of how easily the solute breaks into ions in water – The value is always given and is normally either 1 or 2 • C = molar concentration of solute – This is a measurement of the density of molecules of the solute • R = Pressure constant – This will always be the same no matter what: 0.0831 L bars/M K • T = temperature in Kelvin – The temperature will always be given – Sometimes it will be given in Celsius and you will need to convert it into Kelvin: K = 273 + °C Water Potential Ψ • Once you have both pressure potential and solute potential simply add them together: Ψ = ΨP + ΨS • Make sure to include the units with your answer • Water will always flow from higher water potential to lower water potential Practice: What is the water potential of a 0.5 M sucrose solution at 20°C in an open system? The ionization constant of sucrose is 1. Round your answer to the tenths place. Practice: What is the water potential of a plant cell with 0.3 M sucrose solution at 20°C? The cell wall applies 2 bars of pressure on the cellular solution. Round your answer to the tenths place. Practice: Determine the net flow of water if the plant cell from the second practice problem is placed into the sucrose solution from the first practice problem what will be the net flow of water? Justify your answer.