Groups/Families on the Periodic Table
advertisement

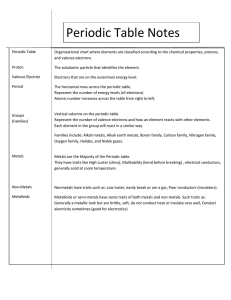
Groups/Families on the Periodic Table (PART ONE) August 15, 2012 Catalyst • Mystery Elements! Use your periodic table and the clues below to determine the identity of 2 mystery elements: 1. This element is located in Group 1 and Period 2 of the periodic table. It is an ingredient in batteries. 2. This element is located in Group 2 and Period 7 of the periodic table. It is radioactive and was discovered in 1898. Objectives • I can compare and contrast properties of metals, nonmetals, and metalloids (semimetals). • I can locate metals, nonmetals, and metalloids (semimetals) in the Periodic Table. Agenda • Catalyst • Notes: Properties of Metals, Nonmetals, and Semimetals. • Color coding periodic table • Unit 1 Quiz – Open Note! • Article: Where do elements come from? Just like you and your family share similarities… So do elements! • 3 main groups 1. Metals 2. Non-metals 3. Semi-metals We will talk about more specific groups later in the week! 1. METALS LOCATION on the Periodic Table: To the left, to the left… Quic kTime™ and a dec ompres sor are needed to see this pic ture. 1. METALS • Physical properties – – – – – – – Solids at room temperature (250C) Luster (shininess) Good conductors of heat and electricity High density (Heavy for its size!) High melting point Ductile (thin wire) Malleable (thin sheets) QuickTime™ and a decompressor are needed to see this picture. 1. METALS • Chemical properties – Tend to LOSE electrons when bonding • form positive ions – ex. K+, Al3+ – Sometimes corrode (disappear slowly by chemical damage) or rust (reacts with oxygen) 2. NON-METALS LOCATION on the Periodic Table: To the right, and with the exception of Hydrogen! 2. NON-METALS • Physical properties (tend to be the opposite of metals) – – – – – – Mostly gases at room temperature No luster (dull) Non-conductors of heat and electricity Low density Low melting point Brittle (breaks easily) QuickTime™ and a decompressor are needed to see this picture. 2. NON-METALS • Chemical properties – Tend to GAIN electrons when bonding 3. SEMI-METALS (Metalloids) LOCATION on the Periodic Table: the Semi-metal “Staircase” What are the semi-metals? B, Si, Ge, As, Sb, Te 3. SEMI-METALS (Metalloids) • Tend to have both metallic and nonmetallic physical and chemical properties. • For example, a semi-metal may be shiny like a metal, but cannot conduct electricity. Please color-code your periodic tables. • The majority of elements are… METALS B Si Ge As Sb Te Unit 1 Quiz: Open Notes! 1. What are isotopes? Give an example. 2. What are ions? Give an example. 3. What part of an atom is the most dense? (Where is most of its mass?) 4. Write a paragraph explaining the relationship between atomic structure and the periodic table. Use these words: atom, proton, neutron, electron, nucleus, atomic number, mass number.