COVALENT Bonds
advertisement

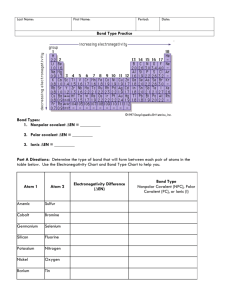
COVALENT BONDS DO NOW What is the ionic formula for Calcium and Fluorine? WORD WALL Big Question (3) Diagram two chlorine atoms bonding. Is this an ionic bond? NO BECAUSE ONE ATOM IS NOT STEALING AN ELECTRON FROM THE OTHER Today‘s Topic COVALENT BOND: a bond formed when atoms shares electron pairs Diagram two oxygen atoms bonding. What do we notice? We need to share two pairs of electrons Vocabulary SINGLE BOND: one pair shared DOUBLE BOND: two pairs shared TRIPLE BOND: three pairs shared DIATOMIC MOLECULE: two of the same element bonded How to Draw Covalent Bonds Hydrogen + Chlorine Bond: H—Cl Formula: HCl Start with formula CO2 Single, Double, or triple? DOUBLE O=C=O Start with the Formula N2 Single, Double, or triple? TRIPLE Together NH3 Together Hydrogen and Oxygen Formula? H2O Try this one… CH4 Try This One Sulfur and Fluorine F—S—F Independent Practice DIAGRAM AND WRITE FORMULA Two hydrogens Carbon and Fluorine PBr3 CH4 Challenge: Nitrogen and Oxygen What is the different between ionic and covalent? Give an example of a possible diatomic molecule Ionic or Covalent? K2S Ionic SO2 IONIC BOND: Between a metal and non-metal Covalent NaCl Ionic P2 Covalent COVALENT BOND: Between two nonmetals You Try! Ionic or Covalent? SrCl2 NO2 Cl2 BeBr2 Are these the same type of bond? CH4 SHARE EVENLY Non-polar CH3F SHARE UNEVENLY Polar Types of Covalent Bonds Non-polar covalent: even sharing of electrons CH4 Polar covalent: uneven sharing of electrons CH3F Polar or Nonpolar? NH3 THIS IS POLAR BECAUSE NITROGEN PULLS ON HYDROGEN MORE THAN HYDROGEN PULLS ON NITROGEN Polar or Nonpolar? CO2 This is nonpolar because they all pull equally on Carbon Polar or Nonpolar? Water is polar because Oxygen pulls hydrogen more than hydrogen pulls oxygen Polar or Nonpolar? NaCl