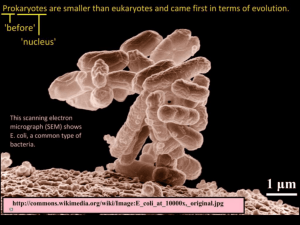
E. Coli
advertisement

Escherichia coli and Salmonella Shuzo Kaneko Andrea Tu Vipin Sandhu Escherichia Coli 1. Gram-negative, short rods 2. Common member of the normal flora of the large intestine. 3. As long as these bacteria do not acquire genetic elements encoding for virulence factors, they remain benign commensals. 4. Strains that acquire bacteriophage or plasmid DNA encoding enterotoxins or invasion factors become virulent and can cause either a plain, watery diarrhea or an inflammatory dysentery. Virulence classification of E.coli (ETEC) EHEC: Enterohemorrhagic EPEC: Enteropathogenic ETEC: Enterotoxigenic Serotype of Escherichia coli : specific Ogroup/H-antigen combinations 1. The presence or absence of specific heat-stable somatic antigens (O antigens) composed of polysaccharide chains linked to the core lipopolysaccharide (LPS) complex. 2. The H antigens are the flagellar antigens. Example: O157:H7 enterohemorrhagic (EHEC) E.coli cause the hemolytic uremic syndrome Enteropathgenic E. coli (EPEC) 1. Defined as those pathogenic E. coli whose pathogenic mechanisms are not related to the production of heat-labile enterotoxins (LT), heat-stable enterotoxins (ST), or to Shigellalike invasiveness. 2. Most EPEC strains adhere to cultured cells (Hep-2), which is rarely observed with other strains. 3. 94-kDa outer membrane protein, EPEC adherence factor (EAF) Entero-Toxigenic E.coli (ETEC) A: Heat-labile toxin (LT) 1. Related antigenically and structurally to cholera toxin. 2. Inactivated at 65oC for 30min. 3. LT1 subunits possess ADP-ribosylating activity. cAMP decreased absorption of Na+ by the tips of the intestinal villi and increased secretion of Cl- by the crypt areas between the villi. The net result in absorption of water into the intestine which manifests itself as diarrhea. B: Heat-stable toxin (ST) 1. Theses strains are designated ST. These are small polypeptide of from 1.5 to 4.0 kDa. 2. Their small size provides them with their heat stability (100 oC for 30min) as well as their non-antigenicity. Enterohaemorrhagic (EHEC) E.coli: O157:H7 1. Complete genome sequenced (2001) 2. Comparison with the nonpathogenic strain K-12 3. 1,387 new genes were found in this strain. these include candidate virulence factors Circular genome map Salmonella: Structure, Classification, and Antigenic Types 1. Gram-negative, flagellated and facultative anaerobic bacteria 2. The cell envelope contains a complex lipopolysaccharide (LPS) structure. (an outer O-polysaccharide coat, a middle portion, the R core, and an inner lipid A coat) 3. This LPS structure is thought as an endotoxin, and important in determining virulence of the organisms. Invasion of intestinal mucosa by Salmonella How were Salmonella’s virulence factors acquired? 1. Genes for the virulence factors of the Salmonella tend to cluster together. 2. The lower G-C content of the pathogenicity islands (around 38%) compared to the rest of the genome (over 50%). It is believed that these genes were acquired through horizontal gene transfer. Genes for Virulence factors 1. Pathogenicity Island 1 (SPI-1) Genes necessary for bacterial entry into the cells of the intestinal epithelium. 2. Pathogenicity Island 2 (SPI-2) Genes necessary for intracellular bacterial replication and the initiation of systemic infection of the host. Both pathogenicity islands code for protein complexes, known as Type III Secretion Systems (TTSSs), used to deliver virulence factors to host cells. Horizontal Gene Transfer Definition The movement of genetic material between bacteria other than by descent in which information travels through the generations as the cell divides. Bacterial species have acquired several mechanisms by which to exchange genetic materials. Horizontal Gene Transfer (Examples) 1. Transformation The uptake of naked DNA Common mode of horizontal gene transfer Mediate the exchange of any part of a chromosome 2. Conjugation The transfer of DNA mediated by conjugal plasmids or conjugal transposons. Requires cell to cell contact. 3. Transduction The transfer of DNA by phage Requires that the donor and recipient share cell surface receptors for phage binding Usually limited to closely related bacteria. The type III secretion system (TTSS) 1. 2. This system is made up of 20 proteins, and ensure controlled secretion and delivery of virulence factors to the host cells. The base of the complex assembles in step wise order. The PrgI needle structure is secreted by the incomplete TTSS already in place. PrgI SPI-1 TTSS is necessary for Salmonella’s initial interaction with host intestinal epithelial cells 1. Salmonella contains genes encoding two forms of TTSS, one located on SPI-1 and the other located on SPI-2. (Mutants lacking working copies of these genes are no longer virulent.) 2. SPI-1 TTSS probably mediates the entry of Salmonella into host cells, by a process termed bacteria-mediated endocytosis. 3. Entry into the host cell then activates the expression of the Salmonella Pathogenicity Island-2 Type III Secretion System (SPI-2 TTSS). Bacterial effector proteins trigger cytoskeleton rearrangements in host cell 1. Cytoskeleton-associated proteins relocate to the site of bacterial entry when Salmonella contact with the host cell. 2. The apical cell surface itself undergoes structural changes, causing its brush border to resemble the membrane ruffles. 3. These changes trigger the endocytosis of the bacteria in membrane-bound vesicles. Bacterial effector proteins trigger cytoskeleton rearrangements in host cell (continue) 4. This process is different from the route of receptor-mediated endocytosis followed by most pathogens into the host cell. 5. After internalization some of these vesicles translocate to the basolateral surface of the cell, allowing the apical membrane to reassume its normal, non-ruffled structure. SipA and SipC : SPI-1 effector protein for host invasion SipA: Binds actin and stabilizes actin filaments, thus decreasing the minimum cellular concentration required for actin polymerization. Therefore, SipA maximizes the efficiency of Salmonella invasion of the host cell. SipC: Aids the entry of other SPI-1 effector proteins. It is believed to activate actin monomers to polymerize and stimulate actin filament bundling, indicating a role in Salmonellainduced host cytoskeleton rearrangements. SopB : the primary virulence factor 1. Two domains homologous to eukaryotic inositol polyphosphate 4-phosphatases. 2. SopB as a enterotoxin is associated with the SPI-1 TTSS. 3. SopB induce an increase the concentration of cellular inositol polyphosphate. SopB : the primary virulence factor (continue) 1. Increased concentration of inositol polyphosphate causes an increase in chloride secretion from the intestinal epithelium into the lumen. 2. This efflux of chloride ions and influx of sodium ions alters the charge equilibrium across the epithelium, leading to a massive fluid secretion designed to restore equilibrium. 3. The secreted fluid causes the watery stool. Dam (DNA adenine methylase) mutants are unable to colonize deep tissues and are avirulent 1. The Dam enzyme methylates adenine residues in the genome. 2. Regulate the timing of biological processes including DNA replication, mismatch repair, and transposition. 3. Dam regulates the expression of at least 20 genes encoding virulence factors, such as various pili operons and the Pag genes. Types of E. coli Commensal Strains Extraintestinal Pathogenic Strains (ExPEC) Intestinal Pathogenic Strains Urinary Tract Infection Entrotoxigenic E. coli (ETEC) Abdominal Infection Entropathogenic E. coli (EPEC) Meningitis Entroinvasive E. coli (EIEC) Pneumonia Entroaggregative E. coli (EAggEC) Cellulitis/Musculoskeletal Infection Diffusely Adherent E. coli (DAEC) Endovascular Infection Entrohemorrhagic E. coli (EHEC) Bacteremia Miscellaneous Infection Commensal Strains • Facultative fecal flora. • Colonize the intestines of virtually all warm blooded animals shortly after birth. • Does not cause disease within the intestinal tract. • Does not usually cause disease outside of the intestinal tract except in presence of other factors. • Strains lack virulence traits. ExPEC Strains • Also present in normal human fecal flora, but possess special genes that code for virulence factors. • Entry into the extraintestinal site, and not acquisition, is the limiting factor for infection. • ExPEC infections can occur in nearly every organ and site, and in all age groups and host types, regardless of human or animal. • Coincidental disease and abnormalities in host defenses result in more adverse outcomes. • Primarily community-acquired, but is also the most frequently isolated gram neg. bacteria in nosocomial environments. ExPEC: Urinary Tract Infection (UTI) • The urinary tract is the most common site of ExPEC infection (90% of ambulatory UTIs; 25-30% long-term care and hospital UTIs). • Serotype O4:H5 is commonly associated with UTI. • UTI is the second most common infection responsible for hospitalization. • 7 epidemiological groups: 1) children under 1 years old 2) school-age girls 3) premenopausal women 4) men with prostatic or other causes of urinary tract obstruction 5) postmenopausal women 6) individuals with neurogenic bladders 7) patients with indwelling catheters ExPEC: Urinary Tract Infection (UTI) • Inflammation of urethra and bladder common, with symptoms of painful urination, frequency and pain in the suprapubic region. • Complications include kidney infection, characterized by fever and/or back pains. • Prolonged or increasing fever may indicate intrarenal abscess and/or obstruction, which may led to renal cell damage or renal failure. • Prostate infection is also a complication in men. • UTI inducing ExPEC is sometimes referred to as Uropathogenic E. coli (UPEC). ExPEC:Abdominal Infection • The abdomen is the second most frequent site of ExPEC infections. • Events resulting in a distruption in the bowel mucosa can lead to an acute peritoneal infection. • Peritoneal abscess formation may also occur following acute peritonitis or as a consequence of subclinical fecal spillage. • Acute peritonitis and intraperitoneal abscesses often involve multiple organisms - both aerobes and anaerobes, with E. coli as the commonest organisms. ExPEC:Meningitis • Most cases of E. coli meningitis occurs in newborn infants. • ExPEC is the primary cause of meningitis in the first month of life (25%-33% of cases), and almost all are caused by the K-1 strain (75% of E. coli possess the Capsular K-1 antigen). • Premature or low-birth weight babies are at higher risk. • Even with treatment, morbidity rate is 20%, and survivors may suffer permanent brain damage. • Infection may occur during delivery. • Otherwise, E. coli meningitis is rare (2% of all meningitis cases), and occurs comorbid to cirrhosis or as a result of a disruption of the meninges. • Neonatal meningitis causing ExPEC is sometimes referred to as Neonatal Meningitis E. coli (NMEC). ExPEC:Pneumonia • Only 2%-5% of community-acquired pneumonia are caused by enteric gram-negative bacteria (GNB) such as ExPEC. • Enteric gram-negative bacteria such as ExPEC are a common cause of pneumonia acquired long-term care homes, and the primary cause of pneumonia acquired in a hospital setting. • GNB caused pneumonia will result in severe illness and mortality rates of 20%-60%. ExPEC:Cellulitis/Musculoskeletal Infection • ExPEC often causes infection of decubitus ulcers and some lower-extremities infections in diabetic patients. • Cellulitis, burn site or surgical infections can also be caused by ExPEC, especially if near the perineum. • Inflammation of the bone as a result of ExPEC acquired through the blood can also occur. • Though rare, E. coli may also cause orthopedic deviceassociated infections and hematogenously acquired inflammation of voluntary muscles. ExPEC:Endovascular Infection • Infections primarily occur in in relation to intravascular devices, and rarely in native heart valves. • ExPEC is a rare cause of prosthetic valve endocarditis, aneurysm infections, and vascular graft infections. ExPEC:Bacteremia • E. coli bacteremia can be a complication of an ExPEC infection of any site; can result in death in the immunocompromised. • E. coli is one of the most common blood isolates (16%-37%), with about equal cases of community-acquired and nosocomial bacteremia. • Approximately 15% of bacteremias are complicated by septic shock. • 60% of bacteremias arise from the urinary tract. • The second most common source of bacteremias is the abdomen, accounting for 25% of all cases. • Soft tissue, bone, and pulmonary infection are the third most frequent sources for bacteremia. • ExPEC strains are among the most common causes of sepsis in neonates ExPEC:Miscellaneous Infection • • • • ExPEC infections can occur in nearly every organ and site. Can cause a significant number of surgical site infections Also known to cause complicated cases of sinusitis. ExPEC may be a cause of Sudden Infant Death Syndrome (SIDs) (Possible toxin mediated). Diagnosis of ExPEC • ExPEC is easily identified by a clinical microbiology lab using standard biochemical criteria. • Grows aerobically and anaerobically. • Grows within 24 hrs. • More than 90% are rapid lactose fermenters. Intestinal Pathogenic Strains • • • • • Rarely encountered in human fecal flora. Obligate pathogens. Cause gastroenteritis or colitis when ingested. Contamination via fecal-oral route. Incapable of causing disease outside the intestinal tract. Can be divided into 6 virotypes based on the following criteria: • Clinical characteristics • Attachment to host cells • Effects of attachment • Toxins • Invasiveness Entrotoxigenic E. coli (ETEC) • Incubation period: 1-7 days; Duration of Illness: 2-6 days. • Inoculum: ID50 = 106 to 1010 colony-forming units(CFU) • Transmission occurs via contaminated food and water; humans are the major reservoir. • Mediated by Heat-Labile Toxins (LT) and Heat-Stable Toxins (STa); stimulate fluid secretion. • Mild symptoms, including cramps, nausea, vomiting, watery diarrhea. However, can also cause death, especially when health care is poor. • Children: causes death in developing countries. • Adults: Partial immunity; Most common cause of traveler's diarrhea (25%-75% of cases). • ETEC uncommon in the U.S. and other developed countries. • Serotype O148:H28 is a common ETEC. Entropathogenic E. coli (EPEC) • Incubation period: 1-2 days; Duration of Illness: weeks. • Inoculum: unknown, is suspected to be low. • Transmission occurs via rapid person-to-person spread; humans are the major reservoir. • Adheres to enterocyte plasma membrane and destroy microvilli; exhibits patchy adherence; process called attaching-effacing. • Not toxin mediated, symptoms result from invasive damages to intestinal walls; involves the entire intestines. • Symptoms: watery diarrhea with mucus but no blood, deformation/destruction of microvilli. • Primarily causes infant/neonatal diarrhea in developing countries; breast-feeding diminishes risk of infection. May also cause some cases of traveler's diarrhea. • EPEC is uncommon in the U.S. and other developed countries • Serotype O126:H11 is a common EPEC. Entroinvasive E. coli (EIEC) • Incubation period: 1-3 days; Duration of Illness: 7-10 days. • Inoculum: ID50 = 108 to 1010 CFU. • Transmission occurs via contaminated food and water; humans are the major reservoir. • Not toxin mediated; Invades the epithelial cells and proliferate, resulting in death of cells and ulceration of the mucosa. • EIEC is fairly uncommon as a cause of diarrhea; In underdeveloped countries, sporadic EIEC induced diarrhea is identified in children and travelers. • Symptoms: an invasive, dysenteric form of diarrhea, including fever, cramps, tenesmus and frequent stool containing blood, leukocytes and mucus. • EIEC rare in the U.S. and other developed countries. Entroaggregative E. coli (EAggEC) • Duration of Illness: long (persistent diarrhea). • Inoculum: high amount required for infection. • Transmission occurs via contaminated food and water; humans are the major reservoir. • Noninvasive, will not alter the histology of host cells; EAggEC forms a “stacked-brick” adherence pattern. • Mediated by Entroaggregative Stable Toxin (EAST) • Symptoms: may cause persistent watery diarrhea, instead of acute diarrhea. • Primarily causes diarrhea in developing countries, affecting young children and immunocompromised individuals. May also cause some cases of traveler's diarrhea. • EAggEC is uncommon in the U.S. and other developed countries Diffusely Adherent E. coli (DAEC) • Duration of Illness: long (persistent diarrhea). • Inoculum: high amount required for infection. • Transmission occurs via contaminated food and water; humans are the major reservoir. • DAEC forms a “stacked-brick” adherence pattern, but in a more diffused pattern than EAggEC. • Symptoms: may cause persistent watery diarrhea, instead of acute diarrhea. • Primarily causes diarrhea in developing countries, affecting young children and immunocompromised individuals. May also cause some cases of traveler's diarrhea. • Relatively little is known about DAEC. • DAEC is uncommon in the U.S. and other developed countries. Entrohemorrhagic E. coli (EHEC) • Incubation period: 3-9 days; Duration of Illness: 1-2 weeks. • Inoculum: ID50 = less than 103 CFU. • Transmission occurs via contaminated food and water, as well as person to person. • Reservoir for EHEC: 1. Mostly in intestine of cattle 2. Also found in chickens, deer, sheep and pigs. 3. Does not make the carrier animal sick. 4. Meat typically becomes contaminated during the slaughtering process. 5. Bacteria on cow’s udders and equipment can also contaminate milk. • EHEC occurs more in developed countries, both outbreaks and sporadic, peaks in summer season. Entrohemorrhagic E. coli (EHEC) • • Mediated by Shiga-like toxin (STX). Serotype O157:H7 is the most common EHEC; result of EPEC + Phage (STX) Typical Symptoms of EHEC Infections 1. GI tract → Large Intestines. 2. Binds tightly via attachment-effacement; Inflammation. 3. Hemorrhagic Colitis (more than 90% of cases): a) Sudden onset of sever cramps and abdominal pain b) Diarrhea within 24 hrs. c) Diarrhea becomes watery. d) Diarrhea becomes grossly bloody. e) Nausea and vomiting. f) Little or no fever. Complications of EHEC:HUS Hemolytic Uremic Syndrome • First described in 1955, now most common cause of kidney failure in childhood. • 5% - 10% of EHEC cases will develop HUS,usually within 2-14 days after diarrhea. • HUS more common in younger children, less so in adolescents. • 90% of HUS is caused by strain E. coli O157:H7. • Caused when E. coli enters the circulation through the inflamed intestinal wall and releases SLT. • SLT attach to receptors on the endothelial cells. • Kidney, pancreas and brain most susceptible. • Characteristic symptoms include hemolytic anemia, thrombocytopenia, and renal failure. • Neurological consequences are also possible (Thrombotic Thrombocytopenia Purpura - TPP); blood clot in brain. Complications of EHEC:HUS • In the active stage of HUS (1-2 weeks): – 50% Require dialysis due to kidney failure – 25% Experience seizures – 5% Suffer diabetes mellitus – Majority require blood transfusions Complications of EHEC:HUS • There is no known therapy to stop disease. • With intensive care, mortality rate is 5% in America. • Of survivors, 5% will develop end stage renal failure, and require dialysis or transplantation. • Another 5%-10% of survivors with have neurological or pancreatic problems. • In children, apparent recoveries may result in hypertension, renal insufficiencies and urinary abnormalities later on in life. Comparison of Intestinal Pathogenic Strains Diagnosis of Intestinal Pathogenic Strains • • • • Differentiate between inflammatory (EIEC and EHEC) and non-inflammatory (ETEC, EPEC, EAggEC and DAEC) diarrhea. ETEC, EPEC, EAggEC and DAEC: either rare or self-limiting = definite diagnosis is not necessary. EIEC: rare, self-limiting = does not require definite diagnosis. Definite diagnosis of EHEC required: a) Most common: does not ferment in sorbitol (sorbitolMacConkey (SMAC) agar), followed by subsequent serotyping for O157 b) More sensitive/rapid/specific: testing for presence of STX. Treatment of Pathogenic E. coli Infection • Antibacterial medication is the primary treatment for extraintestinal pathogenic (ExPEC) strains. • For self-limiting infection resulting from EIEC, ETEC, EPEC, EAggEC and DAEC strains, empirical antimicrobial treatment is also administered. • In the case of EHEC, antibiotics should be avoided, as they may increase the risk of complications such as HUS and TPP. • Diarrheal symptoms require replacement of lost water and electrolytes. Antidiarrheal agents such as Imodium should also be avoided. How Can An E. Coli Infection Be Prevented? • Eating undercook beef is the biggest risk factor for acquiring E. coli O157:H7. Cook all ground beef to at least 160º F. • Don’t eat undercooked beef patties in restaurants. • Prevent harmful bacteria from spreading in the kitchen by keep raw meat separate from ready to eat foods. • Wash all fruits and vegetables thoroughly, especially those that will be eaten raw. • Alfalfa sprouts may also contain E. coli. There is currently no method available to decontaminate alfalfa sprouts. How Can An E. Coli Infection Be Prevented? • Drink only pasteurized milk, juice or cider. • Drink municipal water that has been treated with effective disinfectants (ie chlorine), and bottled water that has been sterilized with ozone or reverse osmosis. • Avoid swallow water when swimming in lakes or pools, esp. public swimming facilities. • Persons with diarrhea should be careful in preventing person-toperson transmission. • Outbreaks caused by person-to-person transmission can also occur at hospitals, nursing homes, and in particular, at day care centers. • Contamination from farm animals and manure is also possible. Avoid contact and wash your hands thoroughly with soap and hot water. Children under 5 should also be extra cautious around cattle, including in petting zoos. • More research is also necessary to reduce the number of cattle that carry E. coli O157:H7, and the contamination of meat during slaughtering. Types of Salmonella NonTyphoidal Salmonella Typhoidal (Enteric) Salmonella Gastroenteritis Typhoid Fever Bacteremia and Endovascular Infections Paratyphoid Fever Localized Infections Intraabdominal Infections Central Nervous System Infections Pulmonary Infections Urinary and Genital Tract Infections Bone, Joint and Soft Tissue Infections Nontyphoidal Salmonellosis • Caused by S. typhimurium and S. enteritidis. • The elderly, infants and immunocompromised individuals more susceptible. • Rainy season of tropical climates; Warm season of temperate climates. • Nontyphoidal salmonellosis is growing rapidly in the U.S; five fold increase between 1974-1994. • Centralization of food processing makes nontyphoidal salmonellosis particularly prevalent in developed countries. • Resistance is a concern, especially with multidrug-resistant strains such as Definitive Type 104 (DT104), such as Definitive Type 104 (DT104). Nontyphoidal Salmonellosis • • • • General Incubation: 6 hrs-10 days; Duration: 2-7 days Inoculum: ID50 = 103 CFU. Transmission occurs via contaminated food and water. Reservoir: a) multiple animal reservoirs b) mostly from poultry and eggs (80% cases from eggs) c) fresh produce and exotic pets are also a source of contamination (more than 90% of reptile stool contain salmonella bacterium); small turtles ban. • Salmonella spreads in hens both vertically and horizontally. • General Symptoms: diarrhea with fever, abdominal cramps, nausea and sometimes vomiting. Nontyphoidal Salmonellosis: Gastroenteritis • Incubation: 6-48 hrs; Duration: 3-7 days for diarrhea & 72hrs for fever. • Inoculum: large. • Mediated by the SopB toxin; attaches to wall of intestines and cause inflammation. • Symptoms include nausea, vomiting, abdominal cramps and fever of 100.5 – 102.2 ºF. Also accompanied by loose, nonbloody stool in moderate volume; Pseudoappendicitis (Rare). • Stool culture will remain positive for 4-5 weeks; less than 1% will be come carriers. • Neonates, elderly and immunocompromised individuals most susceptible. Nontyphoidal Salmonellosis: Bacteremia and Endovascular Infections • 5% develop bacteremia; 5%-10% of bacteremia patients develop localized infections. • Infants, the elderly and immunocompromised patients or those with underlying infections are most susceptible. • Endocarditis: Salmonella often infect vascular sites; preexisting heart valve disease risk factor. • Arteritis: Elderly patients with a history of back/chest + prolonged fever or abdominal pain proceeding gastroenteritis are particularly at risk. • Both are rare, but can cause complications that may led to death. • Serotypes S. choleraesuis and S. dublin are invasive and cause sustained bacteremia and fever. Nontyphoidal Salmonellosis: Localized Infections • INTRAABDOMINAL INFECTIONS: 1) Rare, usually manifested as liver or spleen abscesses. 2) Risk factors: hepatobiliary abnormalities, abdominal abnormalities and sickle cell disease. 3) Treatment: surgery to correct anatomic damages and drain abscesses. • CENTRAL NERVOUS SYSTEM INFECTIONS: 1) Usually meningitis (in neonates, present with severe symptoms e.g. seizures, hydrocephalous, mental retardation, paralysis) or cerebral abscesses. • PULMONARY INFECTIONS: 1) Usually lobar pneumonia. 2) Risk factors: preexisting lung abnormalities, sickle cell disease, glucocorticoid usage. Nontyphoidal Salmonellosis: Localized Infections • URINARY AND GENITAL TRACT INFECTIONS: 1) Urinary infections usually cystitis or pyelonephritis. 2) Genital infections rare, usually ovarian and testicular abscesses, prostatitis, or epididymitis. 3) Both can be complicated by abscess formation. • BONE, JOINT AND SOFT TISSUE INFECTIONS: 1) Bone infections usually in the femus, tibia, humerus or lumbar vertebrae. 2) Risk factors: sickle cell disease and preexiting bone disease. 3) Treatment: prolonged antibiotic usage to prevent relapse/chronic osteomyelitis. 4) Septic arthritis: usually in knee, hip, shoulders. (same risk factors) 5) Individuals with HLA-B27 histocompatibility antigen at risk for developing reactive arthritis and Reiter’s syndrome (the combination of arthritis, conjunctivitis and urethritis) following Salmonella gastroenteritis. 6) Soft tissue infections rare, usually in the immunocompromised, at sites of local trauma. Treatment of Nontyphoidal Salmonellosis • Gastroenteritis: Antibiotics are usually not recommended for Salmonella gastroenteritis; increases rates or relapse and prolonged carriage of bacterium. • Diarrhea: Rehydration of lost fluids and electrolytes. • Bacteremia: Third-generation cephalosporin or quinolones for 7-14 days. • Endovascular Infection: IV ß-lactam antibiotics for 6 weeks. • Extraintestinal Nonvascular Infections: antibiotic therapy for 2-4 weeks. • 2-3 days of preemptive antibacterial treatment for patients susceptible to spreading infection. • Surgical intervention to remove damaged cells. Typhoidal Salmonellosis • Caused by S. typhi and S. paratyphi. • Transmission occurs via person-to-person spread; Human are the only known hosts/reservoir. • Children under 1 year of age are particularly susceptible. • Rare in developed countries; Global health problem, endemic in India, South/Central America, Asia, and recently in eastern Europe. • The large number of global cases serve as a reservoir for U.S. incidence of typhoid fever. Typhoidal Salmonellosis • Inoculum: ID50 = 103 to 108 CFU. • Incubation: 3-60 days; Duration: a few days. • Symptoms: a) Prolonged fever (75% of cases) b) Non-specific symptoms preceding fever c) Variable GI symptoms, such as abdominal tenderness (majority), abdominal pain (20%-40% of cases) and diarrhea. d) “Rose Spots” (30% of cases); Duration: 2-5 days. e) Enlargement of the spleen/liver, nose bleeds, bradycardia. f) Neuropsychiatric symptoms of “muttering delirium” or “coma vigil” Typhoidal Salmonellosis • Late stage complications include intestinal perforation and gastrointestinal hemorrhage • Immediate care such as increase antibacterial medications or surgical resection of bowel. • Other rare complications include inflammation of the pancreas, endocardium, pericardium, myocardium, testis, liver, meninges, kidneys, joints, bone, lungs, and parotid gland and hepatic/splenic abscesses. • In general, symptoms for paratyphoid fever is similar to typhoid fever, but milder with a much lower fatality rate. • Majority clear bacteria from the stool within 8 weeks. However, 1% -5% become asymptomatic chronic carriers; the gall bladder is the primary source of bacterium. Treatment of Typhoidal Salmonellosis • Third-generation cephalosporins or quinolones is the current treatment for typhoidal salmonellosis. • IV or IM ceftriaxone (1g-2g) is also prescribed; usually 10-14 days (5-7 days for uncomplicated cases.) • Multi Drug Resistant (MDR) strains of S. typhi: quinolones are the only effective oral treatment. • Nalidixic acid-resistant S. typhi (NARST) must be tested for sensitivity to determine course of treatment. • Sever typhoid fever (altered consciousness, septic shock): dexamethasone treatment • Chronic carriers: 6 weeks of treatment with either oral amoxicillin, ciprofloxacin, or norfloxacin. • Surgical intervention to remove damaged cells. Diagnosis of Nontyphoidal and Typhoidal Salmonella • Methods for identifying Salmonella bacterium: a) Stool sample or blood sample grown on agar culture plate. b) ELIZA; pinpoints exact serovar via antibody detection. c) Stool sample grown on agar and exposed to bacteriophages. d) Rapid detection of salmonella via PCR of stool isolate e) Other diagnostic methods include detecting for leukopenia and neutropenia. Colony Color Key: •Salmonella = pink •Klebsiella, Enterobacter = blue/green •Serratia = black •E.coli, G+ = white • How Can Nontyphoidal Salmonellosis Be Prevented? Most commonly found in chicken eggs . • Cook egg until the yolk is solid; heat poultry to 180ºF. • Do not store food at room temperature; instead, store at temp above 145ºF or below 39ºF. • Ensure proper hygiene during food preparation, especially in centralized/mass food preparation. • Use pasteurized eggs and poultry products, especially in hospitals and long-term care facilities and commercial food-service establishments. • Avoid antibacterial products to prevent emergence of new resistant strains. • In avoiding typhoidal Salmonella, monitor food and water intake during travel and consider vaccination.