Company profile, July 2010
advertisement

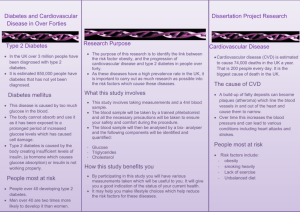
Your track to health!...™ A Truly Non-Invasive Glucose Monitor for Self Use 1 Disclaimer This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and various provisions of the U.S. Securities Act of 1933, as amended, and the U.S. Securities Exchange Act of 1934, as amended. All statements, other than statements of historical facts, included in this presentation are forward-looking statements. Forward-looking statements included in this presentation address, among other things, our future product development activities, strategies and timing of seeking regulatory approval to market our current product candidate. These statements are based on certain historical trends, current conditions and expected future developments as well as other factors the Company believes are appropriate in the circumstances. In addition to statements which explicitly describe these risks and uncertainties, readers are urged to consider statements labeled with the terms “expects”, “anticipates” and other similar words and phrases to be uncertain and forward-looking. All of the forward-looking statements made in this presentation are qualified by these cautionary statements and there can be no assurance that the actual results anticipated by the Company will be realized or, even if substantially realized, that they will have the expected consequences to or effects on the Company or its business or operations. Whether actual results will conform to the Company’s expectations and predictions is subject to a number of risks and uncertainties that may cause actual results to differ materially. See the risks described in our reports filed with the Securities and Exchange Commission, including under the heading "Risk Factors" of our Annual Report on Form 10-K for the year ended December 31, 2014. 2 About Integrity Applications (OTCQB: IGAP) CE Mark approval for a 6 month calibration of GlucoTrack® Model DF-F (March 2014) 15 distribution agreements with minimum sales quota of $28M (in order to maintain exclusivity; subject to local registration) Recruited a Director of International Marketing and Sales with proven history of launching new pharmaceutical and medical device products (July 2014) Engaged Wistron (Taiwan), a Tier 1 contract manufacturer (July 2014) 3 About Integrity Applications (OTCQB: IGAP) Completed an $8.5M Series B offering (Dec. 2014) Formed a Scientific Advisory Board with Members that are recognized as Key Opinion Leaders and Top Tier scientists (Feb. 2015) Engaged Ogilvy CommonHealth (Paris) to create a marketing and communication campaign, designed to increase awareness of the GlucoTrack Submitted pre-submission documents to the US FDA for clinical trials in the US (Aug. 2015) Gained amended CE Mark approval for short calibration (<30 min.) and intended to use also by pre-diabetics (Aug. 2015) 4 Prevalence of Diabetes: 20-79 Years Old 5 Prevalence of Diabetes: 20-79 Years Old 6 Challenges with Self Monitoring of Blood Glucose Vast majority of Diabetics do not follow the IDF guideline on the daily number of spot measurements of blood glucose (IDF: International Diabetes Federation) Conventional (invasive) devices for spot measurement of blood glucose are painful Conventional (invasive) devices for spot measurement of blood glucose are expensive Conventional (invasive) devices are subject to human factors which (meaningfully) decrease the accuracy of the measurement 7 Worldwide Statistics The number of Type 2 diabetics constantly increases 382 million people have diabetes 316 million people have Impaired Glucose Tolerance (IGT) 175 million people are undiagnosed as diabetics Diabetes caused 5.1 million deaths in 2013 — Every 7 seconds a person dies from Diabetes — 11% of worldwide healthcare expenditures were — spent on diabetes care Source: Diabetes Atlas, 6th Edition, Rev. 2014 8 Benefits of Frequent Glucose Monitoring Frequent glucose monitoring is an essential component to effective diabetes management The Diabetes Control and Complications Trial (DCCT) demonstrated that intensive management reduced the risk of complications by: 76% for eye disease 60% for nerve disease 50% for kidney disease 9 HbA1c as a Function of SMBG Source: The American Journal of Medicine, Vol. 111, July 2001 American Diabetes Association stated in June 2012: Reducing HbA1c a Little Less than 1 Point Reduces Cardiovascular Risk by 45% ! 10 Why GlucoTrack? Pain: truly non-invasive Cost: no incremental cost 11 GlucoTrack® Model DF-F Main Unit (MU) Personal Ear Clip (PEC) 12 GlucoTrack Unique Approach Three independent (non-optical) technologies Attempts (by others) to develop non-invasive glucose monitors have mostly been based upon optical technologies, and have not been successful Patented combination of technologies (simultaneously): Ultrasound Electromagnetic (Conductivity) Thermal (Heat Capacity) Unique algorithms Multi sensor Calibration is only required every 6 months Calibration is conducted by a simple (new) process 13 Calibration Process Short (less than half an hour); and Simple (3 data pairs) process; Required every 6 months Times are given for reference only 14 Users Acceptance Feedback (from clinical trials participants) The device is easy to use When available to the market, I will use the device consistently 7% 7% 10% 6% 43% 12% 4% 48% 35% 28% 15 “…Like an iPod!” (Prof. Steven Edelman) Just Clip it! ™ 16 Compelling Economics Direct Cost Comparison per User: GlucoTrack Versus Invasive Device $10,000 $9,000 $8,000 5 invasive measurements daily Virtually unlimited non-invasive measurements ($1,000) 1 2 Invasive 3 Years GlucoTrack 4 $2,900 $2,700 $2,975 $1,350 $0 ($275) $1,000 $2,100 $2,000 $2,300 $3,650 $3,000 $2,500 $5,475 $4,000 $4,600 $7,300 $5,000 $6,225 $9,125 $6,000 $1,825 Cost / Saving $7,000 5 Saving Assumptions: Invasive measurement costs $1.00 each Retail price for GlucoTrack DF-F: $2,000 (including one PEC) PEC costs $100; replaced every 6 months 17 Clinical Trials and Data Initial trials involving more than 25,000 readings from over 450 patients during the past 8 years Clinical data collected since 2009 demonstrate positive correlation between GlucoTrack model DF-F readings and those obtained from conventional invasive devices Safety and performance clinical trial conducted on 135 subjects, yielded 6,275 measurements, of which 96.5% were within clinically acceptable (A & B) zones of the Clarke Error Grid Expected to begin clinical trials in United States in early 2016, pending U.S. FDA protocol approval 18 GlucoTrack Results Demography: 276 subjects 148M; 128F 36 T1DM; 240 T2DM Body Mass Index (BMI): 18.9 – 47.3 Kg/m² Clinical trials, conducted by third party, covered virtually the entire demography, including race and levels of education Age: 18 - 81 years CEG Zone Data Points Percent A+B Zones A 4,309 46% 97% B 4,730 51% C 180 2% D 135 1% E 5 0% Total 9,359 100% 97% of the results fall into the clinically acceptable A&B zones of the Clarke Error Grid (CEG) GlucoTrack model DF-F has CE Mark approval 19 Non-Invasive Measurement Advantages Invasive Non-Invasive Under-utilized Frequently utilized Complex Extremely simple use High cost Cost-effective Preparation required (washing hands, etc.) Just clip it!™ “Stays in the garage” (not being used) “Hits the road” (frequently used) 20 Marketing and Sales Sales are conducted through distributors Distributors are cleared to sell in: Israel Turkey Thailand Spain Hong-Kong Lithuania Uruguay Latvia Estonia Italy Philippines 21 Marketing and Sales GlucoTrack is pending approval for sale in the following countries: o China (expected in 8-11 months) o Japan (expected in 14-17 months) o South Korea (expected in 2-5 months) o Australia o Belarus We intend to add distributors for additional countries during the balance of 2015 22 Marketing and Sales We are increasing our sales, support and field engineering teams to assist our distributors in making sales We hired and keep seeking to hire more business development managers to serve as local liaisons and manage relationships with our distributors We have hired a Coordinator of Social Media to both monitor social network activity with respect to diabetics and blood glucose measurement devices, and to assist in publicizing GlucoTrack through social networking activity 23 Road Map for Current Year (Model DF-F) Improvement of the device characteristics Expect to ramp commercial sales of GlucoTrack FDA process for clinical trials pathway 24 Contract Manufacturers Provide full turn-key manufacturing services o AY Electronics (Israel) o Wistron Corporation (Taiwan, manufacturing arm of Acer Computers) Wistron expects its production line to be operational at the end of 2015 / early 2016 25 Long Term Products Road Map DF-B: Basic model for use in developing countries DF-C: Continuous monitoring of glucose level DF-N: Provides Night time advanced warning of hypoglycemic episodes DF-D: Warn Drivers of low glucose level DF-I: System for pediatric Incubators DF-P: Device for (IGT) Pre-diabetic patients The Company doesn’t guarantee that it eventually will develop all or part of these models. 26 Competitive Landscape # Company Product Technology 1 Echo Therapeutics (MA, USA) Ultra Sound Symphony (ISF) 2 Grove Instruments (MA, USA) Gi-200 3 Cnoga Medical (Israel) TensorTip Optical; BioCGM – parameters; Combo Look-up table 4 Freedom Meditech (CA, USA) Auto I Sugar X Fluorescence (in No data front of the eye) Spot In R&D stage 5 Google (USA) Google Lens No data No data Conceptual stage 6 NovioSense (Germany) NovioSense Tear Glucose No data Continuous In clinical trials Optical Bridge Tear drops Calibration Measurement Picture Comments 12 hours between calibrations Continuous For ICU; In clinical trials No data Spot In clinical trials Long process. Many (>200) Spot points CE Mark Approved; “Selling” 27 Scientific Advisory Board Prof. Lutz Heineman (Chairman): Business Development of Profile Institute for Metabolic Research Ltd., Neuss, Germany Prof. Michael Heise: University of Applied Science of SouthWestphalia, Iserlohn, Germany Prof. Mark A. Arnold: Professor of Laser Chemistry at the University of Iowa Prof. Jens Sandahl Christiansen: Clinical professor at the Department of Endocrinology and Diabetes of Aarhus University Hospital, Denmark Prof. Jan Bolinder: Professor of Clinical Diabetes Research at the Department of Medicine, Huddinge, Sweden 28 Management Avner Gal: President & CEO, Chairman (Co-Founder) o Has held multiple senior management positions in the fields of engineering, technology, and intelligence systems o Served as development engineer and help initialize and bring to life Electronic Warfare combat suite on board the fleet of the Israeli Navy ships David Malka: Executive VP & COO (Co-Founder) o Multiple years of experience building and heading operations departments in a variety of companies o Substantial background introducing products to market Ron Roobroeck: Director of Int’l Marketing & Sales o o o Over 20 years of international experience of marketing, sales and business development Background in pharmaceutical, medical devices, biotech and diagnostic field Experience gained from Novartis, Roche, Medtronic, Cyberonics and Terumo Eran Hertz: CFO o Over 15 years of finance experience in global publicly traded companies o Extensive experience in the areas of finance, compliance and financial reporting o Certified Public Accountant in Israel and in the USA 29 Conclusions The benefits of GlucoTrack will encourage people with diabetes to use it more frequently and regularly Non-Invasive glucose monitoring for home use was a dream. Now it’s reality! Capitalization Approximately 5.32 million shares of common stock outstanding (December 31, 2014) Approximately 2.74 million shares of common stock are issuable upon conversion of outstanding shares of convertible preferred stock Approximately 5.35 million shares of common stock are issuable upon exercise of outstanding warrants Approximately 0.50 million shares of the Company’s common stock are issuable upon exercise of outstanding stock options 31 Your track to health!...™ www.integrity-app.com Thank You! 32