ME_U1_L10 - Heat Transfer_ Work and First Law
advertisement

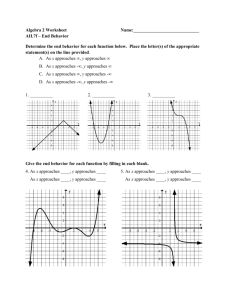
Matter and Energy Drill What mode of heat transfer best describes the scenarios below: Energy transfer through a brick wall Air blowing over a hot surface The sun warming your face on a cold day Matter and Energy Thermodynamics - “the study of systems and energy transfer” Heat (Q) – energy transfer caused by a temperature difference Conduction T Q cond - kA x Convection Radiation Q conv hA T AT 4 Q rad t QQ Matter and Energy Energy is transferred from a warm room at 20C inside a house to the outside air at -10C through a single-pane window. The glass is 5mm thick with an area of 0.5 m2 and a conductivity of 1.4 W/m K. What is the rate of heat transfer through the glass? Known: Heat flows through a window pane (A=0.5m2, ∆x = 5 mm, k = 1.4 W/m K) Q Find: cond , [W] Q cond Sketch: Troom = 20C = 293 K Solution: ∆x Assumptions: Closed System Toutside = -10C = 263 K T Q cond - kA x 2 293K 263K Q (1.4 W/m K)(0.5 m ) cond 1m 5mm( 1000 ) mm Q cond - 4200 W Matter and Energy Cold air at -10C blows over a warm window-pane with a surface temperature of 12C. The glass has a surface area of 0.5 m2 and the convective heat transfer coefficient is h =100 W/m2 K. What is the rate of heat transfer through the glass? Known: Heat flows through a window pane (A=0.5m2, 100 W/m2 K) , [W] Q Find: conv Assumptions: Closed System Toutside = -10C Sketch: = 263 K Q conv Tglass = 12C = 285 K Solution: Q conv hA T 2 2 Q (100 W/m K)(0.5 m )( 285K 263K ) conv Q conv 1100 W Matter and Energy A small light bulb with a surface area of 0.025m2 and an emissivity of 0.6 fluoresces at a temperature of 100C. What is the rate of heat transfer from the light bulb? Known: Heat radiates from a bulb (A=0.025m2, Tbulb = 100 W/m2 K) , [W] Q Find: rad Assumptions: Closed System Sketch: Q rad Tbulb = 100 C = 373 K Solution: AT 4 Q rad (0.6)(5.67x10 -8 Q rad 16.5 W Q rad W m 2 -K 4 )(0.025 m 2 )(373 K ) 4 Matter and Energy Thermodynamics - “the study of systems and energy transfer” A piston cylinder containing a gas was compressed over a period of 20 seconds. The change in energy of the system was 300 kJ and the average rate of heat transfer from the cylinder was 12 kW. Find the work done on the gas in kJ. Known: A gas is compressed inside of a piston-cylinder Find: W, [kJ] Sketch: Assumptions: Closed System Q Solution: E1-2 Q1-2 - W1-2 W1-2 - E1-2 Q1-2 t - E W1-2 Q W1-2 - 300 kJ (-12 kJ/s)(20s) W1-2 - 540 kJ Matter and Energy A vertical piston-cylinder device (D = 5 cm) contains a trapped mixture of gases. The walls of the cylinder are insulated, but heat is allowed to enter through the floor of the cylinder at a rate of 2.75 W over 20 seconds. The absolute gas pressure is 101.3 kPa, initially. If the change in energy of the system is 41 J, how far does the piston rise (in cm) during this process? Known: A gas mixture is heated and expands inside of a piston-cylinder Find: rise of piston, h [cm] Sketch: Assumptions: Closed System , Vertical-piston cylinder -> Isobaric Solution: V1-2 r 2 h h πr 2V1-2 h 3 0.00013 πr 2 8 m h π(0.025 m) 2 h 0.07 m 7 cm W1-2 P W (V V ) V1-2 12-2 1 P 1-2 W1-2 PV 14J W V 1 2 V11--22 101.3 P kPa V1-2 0.000138 m 3 E W11--22 Q11--22 --W E1-22 Q Qt Q(2.75 Q )(t20 s) Q W 55J 41J WW 1- 21- 2 Q1- 2 - E1- 2 W1-2 14J Q (55 J)