Naming Atoms-Elements, Ions and Isotopes
advertisement

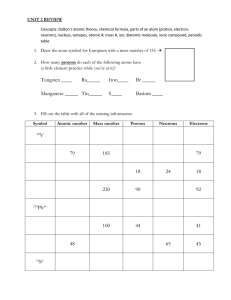
Name:__________________________________ Pd.:_____ Naming Atoms-Elements, Ions and Isotopes Pre-Lab Questions: 1. Identify the element. a. 6 protons, 6 neutrons, and 6 electrons b. 18 protons, 20 neutrons, and 18 electrons c. 3 protons, 4 neutrons, and 3 electrons 2. What is the isotope name for the following atoms? a. 8 protons, 8 neutrons, and 8 electrons b. 15 protons, 16 neutrons, and 15 electrons c. 30 protons, 36 neutrons, and 30 electrons 3. Write ions for the following atoms. a. 5 protons, 6 neutrons, and 2 electrons b. 37 protons, 48 neutrons, and 36 electrons c. 16 protons, 16 neutrons, and 18 electrons 4. An atom has 23 protons, 27 neutrons, and 23 electrons, a second atom has 23 protons, 28 neutrons, and 23 electrons, and a third atom has 23 protons, 27 neutrons, and 18 electrons. a. What is the name and symbol of the element? b. What is the atomic mass number of each atom? c. What is the charge on each atom? d. Write the symbol ( ZA X 0 ) for each atom. Procedure: 1. DO NOT OPEN THE BAGS. 2. On the lab counters are 18 bags labeled A-R. Each bag contains various amounts of black and blue marbles, with a number written on the outside. The black marbles represent protons, the blue marbles neutrons, and the number written represents the total number of electrons. 3. For each bag, record the number of protons, neutrons and electrons in the data table. 4. Using these values and the periodic table, deduce the information needed to complete the data table for each bag. Data Table: Bag Ex. A B C D E F G H I K L M N O P Q R # of Protons (black) # of Neutrons (blue) Mass Number Element 92 146 238 Uranium # of Electrons 92 Symbol A Z X 238 92 Isotope Name 0 U0 Uranium-238