Lesson 3 Properties of Chemicals
advertisement

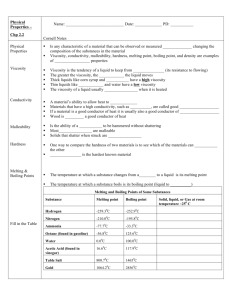
Properties and Characteristics of Matter Physical Properties of Matter • These are observed characteristics – Colour, Lustre, Clarity (by sight) • Lustre is how shiny or dull something appears • Clarity measures how transparent or opaque – Texture (by touch) – Odour (by smell) – Taste • (please don’t use this one) States of Matter Solid • A solid has a fixed shape (DEFINITE) – It does not change shape simply by placing it in a container • A solid has a fixed volume (DEFINITE) – Its’ volume is unchanging – (volume = space it occupies) States of Matter Liquid • A liquid will take the shape of the container holding it, so its’ shape can change (INDEFINITE) • A liquid has a fixed volume (DEFINITE) – If you pour a test tube of water into a beaker, then back into the test tube, it will still be the same volume as before States of Matter Gas • A gas will take the shape of the container (INDEFINITE) • A gas will expand to fill whatever space is provided (INDEFINITE) Other physical properties of matter include: • • • • • • • • Hardness Malleability Ductility Melting/Boiling points Crystal form (structure) Solubility Viscosity Density Hardness The resistance of material to deformation. A rating of how difficult it is to scratch, dent, or otherwise deform a solid. This is often tested by scratching materials together to see which one is marked and which is not. The scale shown to the left is merely an order and not a rating. That is to say, diamond is not twice as hard as apatite. Solids are merely place between materials more, or less hard, than themselves. Malleability The property of a material to be deformed by compression without cracking or rupturing. Malleable materials can be pounded into thin sheets, without breaking or separating. Gold, lead and aluminum are malleable metals. The opposite of malleable, is brittle. Ductility The property of a material which permits it to be reduced in cross sectional area without fracture. This means it can be stretched into long, thin wires of a relatively uniform thickness, without breaking. Ductility and Malleability are related to each other, and ductile materials tend to be more malleable, and malleable materials tend to be more ductile. Boiling Point The temperature at which a substance will change from liquid to gas. The reverse of vapourization (boiling) is called condensation. These temperatures are listed in the textbook, but it should be noted that it is based on standard atmospheric pressure. Increase or decrease the pressure of the atmosphere, and you will change the boiling point of a substance. Melting Point The temperature at which a substance will change from solid to liquid. The reverse of melting is freezing. These temperatures are listed in the textbook, but it should be noted that it is based on standard atmospheric pressure. Increase or decrease the pressure of the atmosphere, and you will change the boiling point of a substance. Things to remember about changes in state (melting, boiling, etc.) • Never think about something becoming cold or gaining cold – objects either gain or lose heat. – A cold item against your skin does not make you cold: you transfer heat to that item, and you lose heat in the process • Some things like carbon dioxide (dry ice) is never a liquid on the earth (under normal pressure). In this case, the process to change from solid to gas is called SUBLIMATION. – Carbon Dioxide can be found in a liquid state, on other planets or moons where the pressure is dramatically different, as well as in a laboratory under the right conditions Crystal Form The shape and structure of the three-dimensional lattice formed by a crystalline material. Some solids like ice, quartz, salt, pyrite and diamond, form crystals when in a solid state. Depending on the shape, nature and composition of the material, it can form wildly different shapes and structures at the atomic level. Diamond Quartz Salt Ice Graphite Solubility A measure of how much of a given substance will dissolve in a liquid. Salt in water vs. rocks in water The substance that is being dissolved is called the solute. The substance doing the dissolving is called the solvent. Water is the most common of solvents. Viscosity The thickness of a liquid or its’ resistance to flow. Molasses and honey = very viscous liquids. Olive oil and other cooking oils = moderately viscous liquid. Water and alcohol = low viscosity. Think of viscosity as the “resistance to pouring”. Temperature will have an effect on viscosity. Lower temperatures will often cause an increase in viscosity. Density The measure of mass per unit volume. Less dense materials will float in liquids of a greater density. Eg. Sweeten colas are more dense than unsweetened colas. The diet coke floats, while regular coke sinks. So what are “non-physical” properties? Those would be CHEMICAL properties. • The two we will deal with are: – Combustibility (if a substance is flammable or combustible) – Reaction with Acid (does it dissolve, do bubbles form, does nothing happen) • In either of these cases, new substances will be formed out of the original substances. Combustion An oxidation reaction accompanied by heat and light. It may or may not require the presence of an open flame to begin the reaction. However, once consumed by the reaction, the process is not reversible. (Have you ever tried turning a lump of charcoal back into a wooden board?) Give-aways to look for: - Light - Heat - Water produced - Carbon Dioxide produced Reaction to Acid A substance can be immersed in acid or have the acid poured over its’ surface. Observe if there are any changes or reactions. Any changes will be a chemical change. Like combustion, once the acid has dissolved the zinc it is not easy to put the zinc back! A common reaction is the formation of bubbles. Often these bubbles are a flammable gas. Another is the material will often be dissolved/disintegrate in the acid. Some metals will react with acid, while others, like gold, do not. Questions • Which properties of matter can be classed as QUALITATIVE and which are QUANTITATIVE? • Questions: p 159 # 1-2