heckRedoxRelay
advertisement
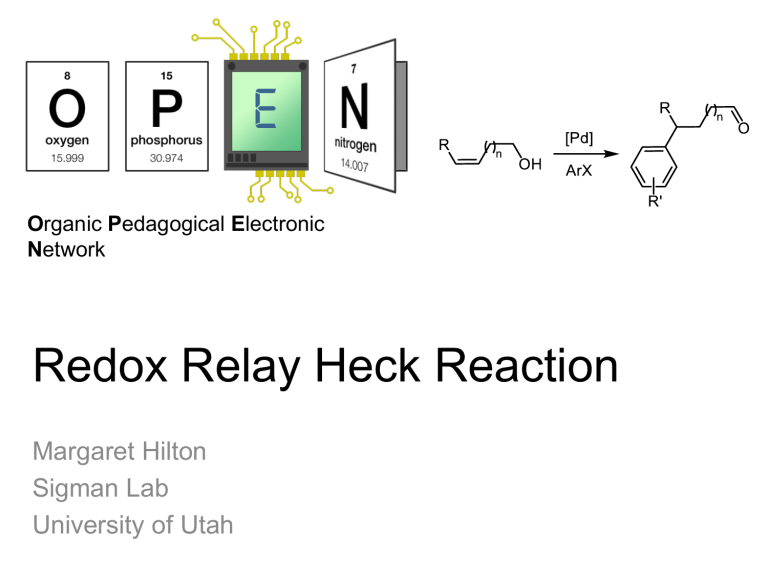
Organic Pedagogical Electronic Network Redox Relay Heck Reaction Margaret Hilton Sigman Lab University of Utah Heck Reaction Since Heck’s seminal publication in 1968, the Heck reaction has been a powerful synthetic tool for the functionalization of olefins with aryl groups. General Mechanism* Heck’s Conditions: Heck, along with Negishi and Suzuki, won the Chemistry Nobel Prize in 2010 for their work in cross coupling. *Note: this reaction may also proceed through an oxidative mechanism, where a PdII precatalyst , a transmetallating aryl reagent, and a terminal oxidant are used, such as with Heck’s conditions shown. Review: Chem. Rev. 2000, 100, 3009. Other Examples and Applications: Overman JACS 1993, 115, 11028 , Sarpong JACS 2008, 130, 7222, Shibasaki JOC 1996, 61, 4876, Sigman JACS 2010, 132, 13981, Sigman JACS 2011, 133, 9692. 2010 Nobel Prize Chain-Walking After the initial migratory insertion, which forms a C-C bond, a βhydride elimination will occur, producing a Pd-H intermediate. If the alkene remains coordinated to palladium and the hydride reinserts at the opposite carbon, the palladium catalyst is now positioned one more carbon down the alkyl chain. This is called a “relay” or “chain walking” and produces alkene isomers. Hayashi JACS 1991, 113, 1417 Curran, JACS 2007, 129, 494 Examples of Relay via Palladium Catalysis Can chain-walking be controlled? Redox-Relay Heck Reaction Sigman and coworkers have developed a redox-relay Heck reaction, where the relay by palladium is controlled by a thermodynamic sink (an alcohol) on the substrate. The unsaturation of the alkene is transferred to the alcohol to form aldehydes or ketones. Classical Variant Oxidative Variant Sigman Science 2012, 338, 1455 Sigman JACS, 2013, 135, 6830 Problems 1. Keay, JOC, 2007, 72, 7253