Scientific Notation - Solon City Schools
advertisement

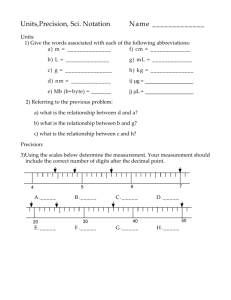
Honors Chemistry Unit 9 Measurement (2013-2014) Units of Measurement Accuracy and Precision Significant Figures Scientific Notation Unit Analysis Density Percent Error 1 We are learning to: 1. 2. 3. 4. 5. Use rules of significant figures for measurements and calculations to reflect precision. Use scientific notation. Demonstrate unit analysis. Determine density. Organize data. We are looking for: 1a. The more precise your equipment is the more exact your measurement can be. 1b. Correctly identifying the amount of sig. figs. in a measurement. 1c. Significant digits are used to show precision of a measured quantity or a calculated quantity. 2a. Use scientific notation to represent very large or small numbers. 2b. Calculate with numbers that are in scientific notation (+, -, *, /) 3a. Use unit analysis to convert units of measurements including derived units (density units). 4a. Measure the density of various samples and use the density to identify the material. 5a. Use Excel to display, manipulate and compare experimentally obtained data sets. Base SI (International System) Measurement Units Measurements: Qualitative: Quantitative: 2 Accuracy and Precision in Measurements Accuracy: Precision: Accuracy ________ _________ _________ Precision ________ __________ _________ Measuring to the Correct Precision What is an acceptable measurement reading for the line drawn below? What is an acceptable measurement reading for the liquid in the graduated cylinder? 3 Accuracy and Precision Activity Lab Equipment Precision Prediction 1–5 1 = best Your Reading Volume Reading #1 Volume Reading #2 Average Reading Actual Precision Ranking Actual Accuracy Ranking Burette 50 mL Graduated Cylinder 250 mL Beaker 250 mL Graduated Cylinder 250 mL Erlenmeyer Flask Burette 50 mL Grad Cylinder 250 mL Beaker 250 mL Grad Cylinder 250 mL Erlenmeyer Flask Actual Volume 1. Predict how precise each piece of lab equipment is by ranking them from 1-5. 1 being most precise to 5 being least precise. Put your predictions in column 1. 2. Read the volume of water in each type of lab equipment. 3. Record your answer in column 2. 4. Record the answers of two classmates in the columns labeled “Volume Reading #1” and “Volume Reading #2”. 5. Find the average reading for each piece of lab equipment. Record your answer in column 5. 6. Obtain the actual volume of water in each piece of equipment. 7. Determine the actual ranking of precision of each piece of equipment by comparing the actual reading to your average reading. Record the actual precision ranking in column 6. Discuss the precision of a ruler with markings of 1 cm to a ruler with markings of 0.1 cm. Why will one of the rulers reflect more precision amongst multiple measurements of the same item such as an index card? 4 Significant Figures What is the definition of significant figures? How do you determine the uncertain (estimated) digit when using a measuring device? 1) How long is the line below? 2) How long is the same line using the ruler below? Which ruler is more precise? How many “certain” digits are in the more precise ruler’s measurement? Which ruler will allow the measurement to have more significant figures? Therefore, the number of significant figures in a measurement is limited by the precision of the measuring device. 5 Significant Figures To determine how many significant figures a number has: P = Pacific = Present decimal = Start on Left A = Atlantic = Absent decimal = Start on Right Pass over any zeroes until you come to a non-zero digit. Count every digit starting with that digit. They are all considered significant from the non-zero digit. How many significant figures are in each of the following measurements? 1) 1.23 cm 3) 0.005 cm 5) 500 cm 2) 5.0 cm 4) 8.000 cm 6) 0.0420 cm Adding Sig Figs: Add numbers as you would a normal addition problem. To determine the number of digits in your answer: Look at the number of places after the decimal in each of the original numbers. The answer will match the value that has the fewest places after the decimal. Example: + 44.223 11.55 55.773 (From the calculator) the answer is 55.773, but 11.55 only has 2 places after the decimal so the answer must be reported to 2 places after the decimal (55.77). From the previous example if the 3 in thousandths place had been a 5 or higher, the answer would have been reported as 55.78. So before reporting places after the decimal, you must look at one place past the correct answer to see if that digit is 5 or more. If it is, the last reported digit must be rounded up by one. 6 Example: + 28.218 1.55 29.768 Since 1.55 only has 2 places after the decimal, the answer would be 29.76 BUT since an 8 is in the place after the 6, the answer is reported as 29.77. Subtracting Sig Figs: Follow the same rules as Addition: Look at the places after the decimal. The answer must match the number with the fewest places. Multiplying Sig Figs: Multiply the numbers as you normally would. To determine the number of digits to report, calculate the number of significant figures for each of the numbers being multiplied (decimal present or absent). The answer will have the same number of significant figures as the number being multiplied with the lowest number of sig figs. Example: x 33.874 (left : 5 sig figs) 11.24 (left : 4 sig figs) 380.74376 (From the calculator), the answer is 380.74376 BUT 11.24 only has 4 sig figs, so the answer can only contain 4 sig figs. Start counting from the left: 380.74 123 4 Look at the digit in the 5th place to see if it is a 5 or higher. If it is a 5 or higher, the 4th digit would be rounded up by one. In this example, it is a four so no rounding will take place. The correct answer is 380.7. Dividing Sig Figs: Follow the same rules for multiplication. 7 Significant Figures Part 1. Determine the number of significant figures in each of the following measurements and write your answer in the space provided. 1) 8 675 309 g _________ 4) 30 200 s __________ 2) 0.035 6 m _________ 5) 0.080 20 g __________ 3) 801.50 mL _________ 6) 1000 K __________ Part 2: Round the following quantity to the specified number of significant figures. 7) 650,000 mL to one sig fig. ____________________________ 8) 0.001 342 94 g to four sig figs. ____________________________ 9) 49 203.03 g to three sig figs. ____________________________ 10) 48 412 g to two sig figs. ____________________________ 11) 0.000 823 938 0 g to five sig figs. ____________________________ 12) 7600 g to one sig fig, ____________________________ Part 3: Perform each of the following calculations and express the answer to the correct number of significant figures. 13) 69.24 cm + 14.2 cm = ____________________________ 14) 13.5 mg – 8 mg = ____________________________ 15) 45.90 dam x 5.41 dam = ____________________________ 16) 34.9 km / 11.169 km = ____________________________ 17) 0.0023 Mg x 787 Mg = ____________________________ 8 Scientific Notation When writing a number in scientific notation, there are two parts: 1. Coefficient: a number equal to or greater than 1 and less than 10 (this number will also reflect the significant figures in a measurement) 1 ≤ n < 10 2. a power of 10 Example: 2.37 x 105 Writing a number in scientific notation Example: # of electrons in a circuit: 6,250,000,000,000,000,000 Step 1: If original # is larger than 1, the power is going to be POSITIVE! Step 2: Put imaginary decimal point at the end of the number. 6,250,000,000,000,000,000. Step 3: Put another imaginary decimal point in the position where the number is between 1 and 10 6.250 000 000 000 000 000. Step 4: Move the decimal at the end of the number until it is on top of the decimal in the 6.25 position. Count the positions as you move. Step 5: The number of times you move is equal to the power value. Step 6: Write the value that is now 1 ≤ n < 10 followed by x 10 and The number of times the decimal moved should be written as a superscript on the 10. Add units to the answer Answer: 6.25 x 1018 electrons/sec To undo the scientific notation, move the decimal 18 times to the right (since the power is positive, the new value will be larger than 10) Adding / Subtracting with Scientific Notation: When adding or subtracting with scientific notation, the powers to all of the numbers being added (or subtracted) must be the same. 2.0 x 107 +1.07 x 107 3.07 x 107 Last step: check the places after the decimal. 2.0 only has 1 place after the decimal so that is the limiting number. The answer can only have one digit after the decimal. In this example, the 0 is followed by a 7 so the 0 is rounded up to a one, making the answer 3.1 x 107 . Notice that the power value is just carried down…since they are the same. 9 Adding / Subtracting with Scientific Notation: If the powers are not equal then one of them must be changed to make them match before addition or subtraction can take place: 9.42 x 109 +1.12 x 108 Since both of these powers are positive, it means as the decimal is moved to the right the answer is going to get bigger, while the power number is going to get smaller. Example: 9.42 x 109 = 94.2 x 108 = 942. x 107 So, changing this first number to be to the power of 8, the problem becomes: 94.2 x 108 +1.12 x 108 Now, adding them together, the answer looks like 95.32 x 108 Last step – check for places after the decimal. Limiting value is 94.2, so the answer will contain one decimal place: 95.3 x 108 Multiplying and Dividing in Scientific Notation Multiplying: 1) Multiply the coefficients (numbers before the powers of 10). 2) Add the exponents on the powers of 10. 3) If the new coefficient is larger than 10 or smaller than one, adjust the placement of the decimal and adjust the exponent on the 10 accordingly. (Hint: LADD; left add) 4) Round the coefficient to the correct number of significant figures. Ex) (6.2x1034 ) x (5.1x1056) 6.2 x 5.1 = 31.62 34 + 56 = 90 31.62 x 1090 Round for significant figures Dividing: 1) 2) 3) 4) Move decimal and adjust exponent 3.162 x 1091 3.2 x 1090 Divide the coefficients. Subtract the exponents on the powers of 10. Adjust the coefficient if needed. Round the coefficient to the correct number of significant figures. Ex) (4.5x1034) / (6.1x1056) 4.5 / 6.1 = 0.7377049 34 – 56 = -22 0.73377049 x 10-22 Move decimal and adjust exponent 7.3377049 x 10-23 Round for significant figures 7.3 x 10-23 10 Scientific Notation Change the following numbers to proper scientific notation: 1. 65.7 g 2. 0.005 45 g 3. 22 450 000 g 4. 3 450 678 001 g 5. 679.3 g 6. 0.0803 g Change the following numbers to standard notation: 7. 6.5 x 10-2 g 8. 9.75 x 106 g 9. 3.4009 x 10-5 g 10. 1.847 x 102 g 11. 8.85 x 10-1 g Addition / Subtraction Problems: 12. 2.367 x 10-13 mL + 5.4 x 10-12 mL 13. 6.50 x 107 mL + 4.321 x 108 mL 14. 4.89 x 10-13 mL + 2.17 x 10-16 mL 15. 9.875 x 108 mL - 2.343x1010 mL 11 Multiplication Problems (Make sure the answer is in scientific notation form): 16. (2.87 x 105 cm) x (3.514 x 109 cm) 17. (5.0 x 10-2 m) x (7.85 x 104 m) 18. (1.042 x 10-1 km) x (4.002 x 10-5 km) 19. (2.21 x 105 cm) x (1.807 x 10-7 cm) Division Problems (Make sure the answer is in scientific notation form and has the right number of sig figs.) : 20. (9.4 x 107 km) / (1.24 x 105 hr) 21. (2.4 x 106 g / 5.49 x 109 cm3) 22. (1.92 x 10-2 g) / (2.3 x 106 mL) 23. (9.2 x 10-3 g) / (6.3 x 10-5 g/cm3) 12 Sig. Figs. & Sci. Notaion Practice Determine the number of significant figures in the following measurements: 1.) 640 cm3 _____ 2.) 0.000 000 009 234 00 mL _____ 3.) 6,650.000 kg _____ 4.) 790,001 dag _____ Perform the following calculations and express the result in the correct number of digits: 5.) 22.0 m + 5.28 m + 15.554 m = ____________ 6.) 0.0050 m2 x 0.042 m = ____________ 7.) 14000 kg + 8000 kg - 590kg = ____________ 8.) 300.3 L / 180. sec = ____________ 9.) (0.054 m + 1.33 m) x 5.4 m2 = ____________ 10.) 13.75 mm x 10.1 mm x 0.91 mm = ____________ Express the following quantities in scientific notation (remember to keep the same amount of sig. figs. from the original number in your coefficient) : 11.) 8,800,000,000 cm = _____________________ 12.) .000 000 06 L = _____________________ 13.) 0.210 5 g = _____________________ 14.) 4813.67 hm = _____________________ 15.) 654,321.789 Gg = _____________________ 16.) 0.000 683 5 mL = _____________________ Take these measurements out of scientific notation and put them into standard notation: 17.) 8.4356 x 10-4 cm = _____________________ 18.) 6.574839 x 103 kg = _____________________ 19.) 4.21 x 106 ml = _____________________ 20.) 9.21 x 10-7 L = _____________________ more on the back 13 Carry out the following calculations. 21.) 2.48 x 1012 kg + 9.17 x1013 kg + 7.2 x 1013 kg = _____________________ 22.) 4.07 x10-15 g + 3.966 x 10-14 g + 7.1 x 10-13 g = _____________________ 23.) 3.890 x 108 km / 1.97 x 103 s = _____________________ 24.) 1.111 1 x 105 cm x 5.82 x 104 cm = _____________________ Without using a calculator, solve the following problems: 25.) 2.000 x 10120 m x 3.0 x 1044 m = _____________________ 26.) 5.000 x 10341 g / 5.00 x 10141 mL = _____________________ 14 Honors Chemistry Worksheet "Significant Figures and Scientific Notation" Part 1: Determine the number of significant figures in each of the following measurements and write your answer in the space provided. 1) 8 675 309 g ________ 4) 30200 s ________ 2) 0.0356 m ________ 5) 0.080 20 g ________ 3) 801.50 mL ________ 6) 1 000 000 K ________ Part 2: Round the following quantity to the specified number of significant figures. Standard Notation Scientific Notation 7) 695,900 mL to three sig. figs. 8) 0.001 342 94 g to four sig. figs. 9) 49 203.03 g to three sig. figs. 10) 0.000 000 775 2 mg to two sig. figs. 11) 0.000 293 749 0 in to four sig. figs. 12) 3400 kg to one sig. fig. Part 3: Perform each of the following calculations and express the answer to the correct number of significant figures. Standard Notation Scientific Notation 9) 69.24 dm + 144. 2 dm = 10) 13.5 mg – 8 mg = 11) 245.90 dam x 9.41 dam = 12) 34 km2 / 1581.169 km = 13) 0.0023 Mg x 77 Mg = 14) 5.44 cm x 31 cm x 0.0984 cm = 15) 0.043 kg / 452.1 kg/mL = 16) 300 ft x 9.7600 ft = 15 16 Factor Label Method (Notes on how to convert using conversion factors) I. Conversion Factor: a ratio that can be used to convert from one unit to another. The numerator and the denominator are equal to each other The denominator’s unit should be the same as the given numbers unit The numerator’s unit will be the unit you want to convert to Example of a conversion factor: 4 quarters or 1 dollar 12 eggs 1 dozen II. Factor Label Method Procedure: 1. Write the given number and unit 2. Set up a conversion factor (fraction used to convert one unit to another) 3. Place the given unit as denominator of conversion factor 4. Place desired unit as numerator 5. Cancel units 6. Solve Problem III. Factor Label Method Procedure (Metric to Metric): 1. Write the given number and unit 2. Set up a conversion factor (fraction used to convert one unit to another) 3. Place the given unit as denominator of conversion factor 4. Place desired unit as numerator 5. Place a “1” in front of the larger unit 6. Determine the number of smaller units needed to make “1” of the larger unit 7. Cancel units 8. Solve Problem 17 Metric System Units (Number of base units needed to make one) The Tera T 1,000,000,000,000 Grand Giga G 1,000,000,000 Master Mega M 1,000,000 King Kilo k 1,000 Henry Hecto h 100 Died Deka da 10 By Base Unit Liter, Meter, Gram (Number needed to make one base unit) Drinking deci d 10 Chocolate centi c 100 Milk milli m 1,000 Monday micro μ 1,000,000 Night nano n 1,000,000,000 Prior pico p 1,000,000,000,000 Other Important Conversions: 12 in. = 1ft 2 pt = 1 qt 1 lb = 454 g 1 in = 2.54 cm 3 ft = 1 yd 4 qt = 1 gal 1 lb = 16 oz 1 m = 39 in 5280 ft = 1 mi 1 qt = 0.946 L 1 metric ton = 2200 lb 1 mi = 1.61 Km 1760 yd = 1 mi 1 qt = 32 fl oz 1 mL = 1 cm3 18 "Problems Using The Factor-Label Method" Use the factor-label method to show your work to convert each of the following measurements to the specified unit. 1) 5 dm to m 2) 700 mm to nm 3) 1400 cm to km 4) 8 km to mm 5) 250 mL to L 6) 43000 mg to kg 7) 560 mL to L 8) 12 L to mm3 9) 10 dm3 to L 10) 54 yd3 to m3 11) 45,200 sec. to days Medicines were at one time dispensed in the Apothecaries’ Measuring System as listed below: 1 Apothecaries’ pound = 12 Apothecaries’ ounces 175 Apothecaries’ pounds = 144 English pounds 1 kilogram = 2.205 English pounds 16 English ounces = 1 English pound 12) If someone offered you an ounce of gold, would you prefer an English ounce or an Apothecaries’ ounce? Determine the kg equivalent of each type of ounce to answer this question. 19 Name ______________________________________________________________ Practice Conversions 1) 38.02 m 56.29 m Find the area of the above garden in ft2 (hint: 2.54 cm = 1 inch). 2) How many mL would a small cubed container hold that is 2.00 inches on each side? 3) Convert 65 miles/hour to meters/sec. (hint: 1 mile = 1.61 km) 4) A box has dimensions of 1.20 ft, 2.40 ft, and 4.80 ft. What is the volume of this box in mL? 5) The height of a tree is 15.0 yards. A tree trimming company charges $30.00 per meter to cut down the tree. How much is it going to cost to cut the tree down? 6) You want to buy genuine Italian marble tiles for your kitchen floor. The dimensions of your kitchen are 12.0ft by 16.0 ft. The tiles are made in Italy, therefore they measure using metrics. Each of the tile will cover 935 cm 2. They only sell them in quantities of 10 tiles per box. It costs 37.44 Euros per box. How much will the tiles cost for your kitchen floor? (1 Euro=$1.37) You will need to work this out on a separate sheet of paper. 20 Density Density is a measure of mass per volume. D= Answer the following questions on density and experimental error. Show all your work with units and round your answer to the correct number of sig. figs.! 1. What is the density of a cardboard if 6.2 g occupy 8.56 cubic centimeters? 2. What is the density of a gold nugget having a volume of 2.39 cubic centimeters and a mass of 45.58 grams? 3. What is the mass of a piece of aluminum having a volume of 15.12 cubic centimeters and a density of 2.70 grams per cubic centimeter. 4. Cerium sulfate has a density of 3.17 grams per cubic centimeter. What is the volume of .54 grams of this substance? 5 What is the density of a brick if 51.21 g occupy 31.32 cubic centimeters? 6. Cerium sulfate has a density of 3.17 grams per cubic centimeter. What is the volume of 1.25 grams of this substance? 7. Tin has a density of 7.28 grams per cubic centimeter. What is the volume of 11.2 grams of this substance? 21 Percent Error (Experimental Error) Density and Percent Error Practice 1) You measure the density of substance in the lab as 4.25 g/mL. The true value of the density of the substance is 4.32 g/mL. Calculate your percent error. 2) Your measurement of the volume of a sample of tap water is 9.6 mL, 9.52 mL and 9.553 mL using 3 different graduates. What is the average volume of the tap water? 3) The average mass of this tap water is 9.2 g, what is the density? 4) The standard density of water is 1.00 g/mL, What is your percentage error for the above problem? 22 Layered Solutions Activity Objective: To create a column of distinct layers of different solutions. Procedure: 1. In five different cups, place the following ingredients: Chart 1: Ingredients for layered solution (you must use at least 20mL but each cup must use a different volume of water and each must differ by at least 10 mL). Cup Number Salt (g) Warm water (mL) Calculated Density (g/mL) 2. Once you have added all of the necessary materials, stir each one with a spoon for at least one minute until all of the salt is completely dissolved. 3. Using sig figs, calculate the density for each cup and record the density in the last column of Chart 1. 4. Using the balance and 10 mL of each solution you just made, measure the density of each solution. Record these values for density in Table 1 on the back of this sheet. 5. Determine the order to put the liquids into the graduated cylinder. Which one should go in first, the least dense or the most dense? 6. Add food coloring to create a rainbow effect with the different layers. 7. Using your pipette, carefully transfer 20 mL from each cup into the 100 mL graduated cylinder. Observations: Table 1: Data from the Layered Solution Activity Cup Number Measured Density from solutions made (g/mL) Projected order to fill graduated cylinder The best layers will win a prize!!! 23 Lab: Graphing and Density Name:________________________ Class Period:_____ Purpose: -Determine the density of a liquid from a graph of mass and volume. -Determine the layering order of three liquids if poured together into a graduated cylinder. Problem: Are density and the layering order of liquids in a graduated cylinder related? Hypothesis: (If…,then…) Experiment: Materials: 25mL graduated cylinder balance rubbing alcohol water dropper calculator Procedure: 1) Determine the mass of 5mL of water and record this in the data table. a. Place the empty graduated cylinder on the balance and record this mass below: i. Mass of empty cylinder:_____________g b. Place the 5mL of water in the cylinder and carefully place it on the balance. c. Subtract the mass of the empty cylinder. d. Record the mass in the data table. 2) Repeat step 1 for 15mL and 25mL of water. 3) Repeat step 1 for 5mL, 15mL, and 25mL of rubbing alcohol. a. Use the beaker of rubbing alcohol that is at your table. Return the alcohol to the beaker when finished for the next class to use!! 4) We will not be measuring the values for silicone oil because it is too messy. The data has already been given to you in the data table. 5) Graph the data on the provided graph under the data table. a. Use a different colored pencil for each line. b. Provide a key to identify each color. c. Make a title for the graph. d. Make a best-fit line for each color. The line must go through 0,0. e. Determine the slope of each line and show your work. 24 Data: Volume (mL) Water Mass (g) Rubbing Alcohol 5 Silicone Oil 4.60 15 13.70 25 23.10 ___________________________________ Slope determination of each line to calculate the density of each liquid: (You MUST show your work! Remember to put units with your work and answer.) Circle your answer. Water: Rubbing alcohol: Silicone oil: Given your results, how will the liquids be layered if poured carefully into a graduated cylinder? Make sketch show which liquid would form the bottom… will be in the middle, and … will be on top. 25