Assitance Lecturer Amjad Ahmed Jumaa Aromatic hydrocarbon
advertisement

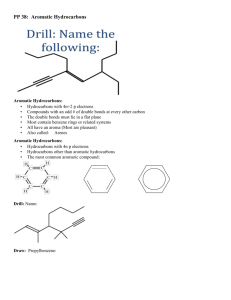
Organic and biochemistry Aromatic hydrocarbon Benzene Assitance Lecturer Amjad Ahmed Jumaa www.soran.edu.iq 1 Aromatic hydrocarbons Benzene Aromatic hydrocarbons: are benzene and compounds that resemble benzene in chemical behavior. Aromatic properties are those properties of benzene that distinguish it from aliphatic hydrocarbons. They are characterized by a tendency to undergo ionic substitution. www.soran.edu.iq Molecular formula. Isomer number. KeKule' structure 1-benzene has the molecular formula C6H6. Benzene was known to contain six carbon atoms and six hydrogen atoms. in 1865, he offered an answer to the question of benzene : these carbon chains can sometime be closed to form rings. KeKule' structure of benzene represented as today (I) www.soran.edu.iq Other structure consistent with the formula C6H6. For example the structures II-V. Of all these, KeKule's structure was accepted as the most nearly satisfactory www.soran.edu.iq 4 2-benzene yield only one monosubstitution product, C6H5Y.only one bromobenzene, C6H5Br. Similarly only one chlorobenzene, C6H5Cl . Or one nitrobenzene, C6H5NO2. From this fact we see each hydrogen must be exactly equivalent to each other hydrogen. www.soran.edu.iq Thus the structure (I) above is the acceptable structure for the benzene ring. Nomenclature of benzene and its derivatives For many of these derivatives we prefix the name of the substituent group to the word –benzene, for example, chlorobenzene, bromobenzene, etc. www.soran.edu.iq 6 Special names: we see methylbenzene is always known as toluene, amino benzene is called aniline, hydroxybenzene as phenol, and so on. www.soran.edu.iq For several substituents (more than one groups that attached benzene ring), the names are differentiated by use the names ortho, Meta, and para, for example: www.soran.edu.iq If the two groups are different, we simply name the two groups successively and the end the word with –benzene, for example: www.soran.edu.iq 9 For special name the compound is named as a derivatives of that special compound as for example, nitro toluene, bromophenol. For more than two groups numbers are used to indicate their relative positions, for example: www.soran.edu.iq For more than two groups numbers are used to indicate their relative positions, for example: www.soran.edu.iq 11 Solution: www.soran.edu.iq Follow problem: draw the structure for the following benzene derivatives: 1-(3-ethyl-2-methylaniline). 2-(2, 4-dinitrobenzene). 3-(1-ethyl-3-nitrobenzene). 4-(3,4,6-tribromobenzene). 5-(1-iodobenzene). 6-(p-methylbenzene). 7-(ethylbenzene). 8-(isopropylbenzene). www.soran.edu.iq 13