Gas Properties Notes
advertisement

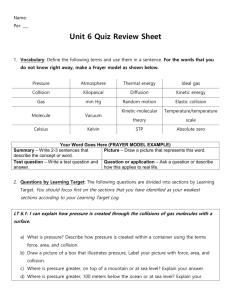
GASES Behavior of Gases Gases behave much differently than liquids and solids and thus, have different laws. Because gas molecules have no forces keeping them together, they fill whatever container in which they are kept. This distance between the molecules also means they can be compressed. Kinetic Molecular Theory of Gases Model explaining gas behavior. 1) A gas consists of small particles (atoms or molecules) that move randomly with high velocities. 2) The attractive forces between gases can be ignored. Kinetic Molecular Theory of Gases 3) The actual volume that the gas molecules occupy is usually much smaller than the volume of the container. - Therefore, gases can be easily compressed because of all the empty space. 4) The average kinetic energy (temperature) of gas molecules is proportional to the Kelvin temperature. Kinetic Molecular Theory of Gases 5) Gas particles are in constant motion, moving rapidly in straight paths until collide with another gas molecule and change direction. - When they hit each other or the walls of the container, they create gas pressure. - As collisions increase, pressure increases. Properties of Gases The following properties are used to describe gases: Pressure Volume Temperature Amount of gas Pressure of a Gas Pressure of a gas occurs when gas molecules strike each other as well as the sides of a container. As collisions increase between molecules, pressure increases. The pressure of the air molecules around us is called atmospheric pressure. Atmospheric pressure – decreases with altitude because there are fewer air molecules present Common Units of Pressure Atmosphere (atm) Millimeters of Mercury (mmHg) Torr (torr) Pounds per square inch (lb/in2 ) Pascal (Pa) Barometric Pressure -As air pressure increases (downward) the barometer reading (Hg) rises upward. Volume of a Gas The volume of a gas equals the size of the container in which the gas is placed. Volume of gases are generally measured in liters (L) or cubic decimeters (dm3 ) which equal the same amount. Also, 1mL = 1 cm3 Temperature of a Gas Temperature reflects the average kinetic energy due to the movement of a gas. Measured in Kelvin (K) Amount of a Gas Uses moles to describe the relative quantity of molecules of a gas that are present. What happens to the pressure of a gas when volume decreases? 1. Pressure decreases 2. Pressure increases 3. No change 4. Unable to tell 0% te ll 0% to bl e Un a No ch an ge 0% in cr ea se s Pr es su re Pr es su re de cr ea se s 0% What is the relationship between pressure and volume? 1. Direct 2. Inverse 3. Exponential 4. No relationship 0% 0% t io ns hi re la No Ex po n en t ia p l 0% In ve rs e Di re ct 0% Boyle’s Law Boyle’s Law – If temperature is held constant, gas pressure is inversely proportional to the volume of the gas. Boyle’s Law: P1 V1 = P2 V2 What happens to the volume of a gas when temperature increases? 1. Volume decreases 2. Volume increases 3. No change 4. Unable to tell Vo l 0% te ll to bl e No ch an ge 0% Un a um e in cr de cr ea se s e um Vo l 0% ea se s 0% What is the relationship between temperature and volume? 1. Direct 2. Inverse 3. Exponential 4. No relationship 0% 0% t io ns hi re la No Ex po n en t ia p l 0% In ve rs e Di re ct 0% Charles’ Law As temperature increases, average kinetic energy of gas molecules increases. As the molecules move faster, volume increases. Charles’ Law – If pressure is held constant, there is a direct relationship between volume and temperature. Charles’ Law V1 T1 = V2 T2 Note: Temperature is measure in Kelvin Kelvin = Celsius + 273 What happens to the pressure of a gas when temperature increases? 1. Pressure decreases 2. Pressure increases 3. No change 4. Unable to tell 0% te ll 0% to bl e Un a No ch an ge 0% in cr ea se s Pr es su re Pr es su re de cr ea se s 0% What is the relationship between temperature and pressure? 1. Direct 2. Inverse 3. Exponential 4. No relationship 0% 0% t io ns hi re la No Ex po n en t ia p l 0% In ve rs e Di re ct 0% Gay – Lussac’s Law As temperature increases, collisions increase and pressure increases as well. Gay – Lussac’s Law – If volume is held constant, there is a direct relationship between pressure and temperature. Gay – Lussac’s Law P1 T1 = P2 T2 Gay-Lussac’s Law is true if number of moles of the gas as well as the volume is held constant. Note: Temperature is measure in Kelvin Kelvin = Celsius + 273 Combined Gas Law P 1 V1 T1 = P 2 V2 T2 The combined gas law applies as long as the number of moles of a gas are held constant. Note: Temperature is measure in Kelvin Kelvin = Celsius + 273 What happens to the volume of a gas when the number of atoms increases? 1. Volume decreases 2. Volume increases 3. No change 4. Unable to tell Vo l 0% te ll to bl e No ch an ge 0% Un a um e in cr de cr ea se s e um Vo l 0% ea se s 0% What is the relationship between volume and moles of a gas? 1. Direct 2. Inverse 3. Exponential 4. No relationship 0% 0% t io ns hi re la No Ex po n en t ia p l 0% In ve rs e Di re ct 0% Avogadro’s Law V1 n1 = V2 n2 Avogadro’s Law is true if pressure and temperature are held constant. Note: n = moles of the gas Avogadro’s Law V1 n1 = V2 n2 The combined gas law applies as long as the number of moles of a gas are held constant. Note: Temperature is measure in Kelvin Kelvin = Celsius + 273 True or False: Increasing the volume of a gas increases the pressure. 1. True 2. False se 0% Fa l Tr ue 0% True or False: Increasing the pressure of a gas increases the temperature. 1. True 2. False se 0% Fa l Tr ue 0% True or False: 1 mole of iron metal has a volume of 22.4L at STP. 1. True 2. False se 0% Fa l Tr ue 0% True or False: Decreasing the temperature of a gas increases the pressure. 1. True 2. False se 0% Fa l Tr ue 0% True or False: Decreasing the temperature of a gas increases the volume. 1. True 2. False se 0% Fa l Tr ue 0% True or False: Increasing the moles of a gas increases the pressure. 1. True 2. False se 0% Fa l Tr ue 0% STP STP – Standard Temperature and Pressure STP: T = 273K (or 0o C) P = 1 atm or 760 mmHg Molar Volume ***** At STP, according to Avogadro, any gas has a molar volume of 22.4L. Conversion factor: 1 mole (of any gas) = 22.4L Ideal Gas Law – used for conditions not at STP PV = nRT R = Ideal Gas Constant Rearranged, R = PV/nT At STP for one mole, calculate R values for units of atm and mmHg. Ideal Gas Law At 2 atm pressure, what is the volume of 10 moles of hydrogen gas at 200K? What pressure would 4.04g of hydrogen gas (H2) create at 100oC? Law of Partial Pressures The Law of Partial Pressures states that the total pressure of a gas is equal to the sum of all of the individual pressures. Ptot = P1 + P2 + … Pn Volume (mL) Volume (L) of gas at STP 1000mL = 1 L Volume (L) Molarity Molar Mass 6.02 x 1023 Mass (g) Moles of A Mass (g) Mole Ratio 6.02 x 1023 Moles of B Molar Mass Molarity Volume (L) Volume (L) of gas at STP 1000mL = 1 L Volume (mL) Atoms or Molecules Atoms or Molecules