Chapter 10: Energy
Chapter 14: Liquids and Solids
Purpose
Energy can be transferred or it can change form, but it cannot be created or destroyed. Matter can be described
by its physical properties. The physical properties of a substance depend on the spacing between the particles of
the substance and the forces of attraction between the particles. Heating curves and phase diagrams graphically
relate temperature, energy, phase of matter, and strength of intermolecular forces.
A and B
3/17 and 3/18
In-Class
Chapter 10 & 14 Notes
3/19 and 3/20
Go over Chapter 10 &14
Worksheet #1
Specific Heat Lab
Go over Chapter 10 & 14
Worksheet #2
Food Energy Lab
Go over Chapter 10 & 14
Review
Chapter 10 & 14 Test
3/23 and 3/24
3/25 and 3/26
HW Assignments
Chapter 10 & 14
Worksheet #1
Chapter 10 & 14 Worksheet
#2
Chapter 10 & 14 Review
Vocabulary
energy
thermodynamics
temperature
kinetic energy
potential energy
heat
endothermic
exothermic
enthalpy
heat of reaction
entropy
calorie
Joules
specific heat capacity
calorimeter
Hess’s Law
intermolecular forces
intramolecular forces
dipole-dipole
hydrogen bonding
London dispersion forces
heat of vaporization
heat of fusion
sublimation
evaporation
condensation
triple point
critical point
deposition
melting
freezing
By the end of this Topic, you should be able to demonstrate proficiency in the following areas:
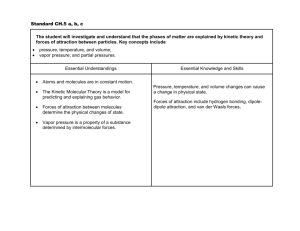
Essential Understandings
The phase of a substance depends on temperature and pressure.
Forces of attraction (intermolecular forces) between molecules determine their state of matter at
a given temperature. Forces of attraction include hydrogen bonding, dipole-dipole attraction, and
London dispersion (van der Waals) forces.
Solid, liquid, and gas phases of a substance have different energy content. Pressure, temperature,
and volume changes can cause a change in physical state. Specific amounts of energy are absorbed
or released during phase changes.
A heating/cooling curve graphically describes the relationship between temperature and energy
(heat). It can be used to identify a substance’s phase of matter at a given temperature as well as
the temperature(s) at which it changes phase. It also shows the strength of the intermolecular
forces present in a substance.
Molar heat of fusion is a property that describes the amount of energy needed to convert one
mole of a substance between its solid and liquid states. Molar heat of vaporization is a property
that describes the amount of energy needed to convert one mole of a substance between its
liquid and gas states. Specific heat capacity is a property of a substance that tells the amount of
energy needed to raise one gram of a substance by one degree Celsius. The value of these
properties are related to the strength of their intermolecular forces.
Essential Knowledge, and Skills
In order to meet this standard, it is expected that students will
distinguish between an endothermic and exothermic process.
identify how hydrogen bonding in water plays an important role in many physical, chemical, and
biological phenomena.
graph and interpret a heating curve (temperature vs. time)
interpret a phase diagram of water.
calculate energy changes, using specific heat capacity.
explain the relationship between intermolecular forces and relative size of heat of fusion/vaporization.
calculate the energy changes, using molar heat of fusion and molar heat of vaporization.
SOL Standards
CH.5 The student will investigate and understand that the phases of matter are explained by kinetic theory and
forces of attraction between particles .
Key concepts include
d) phase changes;
e) molar heats of fusion and vaporization;
f) specific heat capacity;
 0
0