Siderophores - Princeton University
advertisement

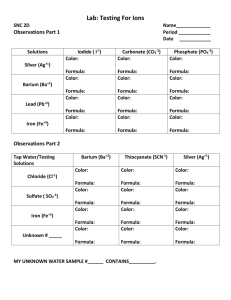
Synthesis, Characterization and Iron-Acquisition of Hydroxamic Acid Derivatives -Functional studies based on the subunits of amphiphilic siderophores O OH O NH O N NH2 HO O NH HN OH N HN + O HO HO OH O NH H O HN HN O O NH3+ N H O Linda M. Johnson University of North Carolina at Chapel Hill Class of 2003 Dr. John T. Groves/ Minkui Luo Princeton University May 6, 2004 Mercer County Community College Iron – An Important Element • 4th most abundant element in the world • An important nutrient for the body. • Helps with growth and development in the body, especially in children. • Iron is forever cycled from a liver storehouse in a protein called ferritin. The Paradox of the Limited Abundance of Iron Lack “iron” in such an iron-abundant planet ? •Extremely limited bioavailability. •Bacteria battles to acquire iron to obtain nutrients that to help contribute to growth Siderophores • Small weight compounds that have a high affinity for Fe (III). • Help with Iron Transport. • Three subunits of siderophore’s chelating groups. A Common Strategy of Bacteria --- IronDelivery Shuttle, Siderophore R' 3+ N + Fe (aq) OH R O R' R O 1 N O Fe R + Fe (aq) OH O HO O + Fe3+(aq) OH Fe O OH O 2H N HN + O Fe HO 2H+ OH O HN O + O O HO NH H O O O O OH HN R HO NH HN + NH O N NH2 + 2 R H+ R 3+ HO + N H O O NH3+ Pseudobactin Albrecht-Gary A. M.; Crumbliss A. L. Metal Ions in Biological Systems 1998, 35, 239 Water-phase Iron-acquisition Kinetic of Citrate-based Amphiphilic Siderophore O NH N HO OH O OH O O HO NH N -1 0.15 Apo-Acinetoferin O O NH N HO OH O OH O O HO NH N Apo-Rhizobactin O O Iron-acquisition Rates / s O 0.12 Acinetoferrin Rhizobactin SAF 0.09 0.06 0.03 0 NH N HO OH O OH O O HO NH N 0 1 2 3 4 FAC Concentration / mM Apo-SAF kobs km [ FAC ] kp [ FAC ] [ FAC ] 1 / Kp 5 Purpose of Experiment • To examine and compare the structures of siderophores’ sub-unitshydroxamic acids. • To understand how hydroxamic acids facilitate the iron chelating process of siderophores by measuring their ironacquisition rate. Synthesis of N1-BOC propane diamine H2N 2 equi. BPO 10% TEA/MeOH NH2 HN (BOC)2O O NH2 HN O O N H O Compound 1 Compound 2 NaOH/ MeOH HN O N H O Compound 3 OH OBz Synthesis of Compound II & III • Synthesized Bocprotected amine • Use Flash Column Chromatography (FCC) to help separate compound. • Perform Thin Layer Chromatography (TLC) to help locate the final product. • Check purity of compound with 1H-NMR. Five substrates used to react with the amine: • Acetyl Chloride O Cl Cl • Cinnamoyl Chloride O • Butyryl Chloride O Cl O Cl • Crotonyl Chloride • Hydrocinnamoyl Chloride Cl O Procedure of Analog Synthesis • Performed test reactions with each halide. • Check TLC and take 1H-NMR for each reaction. • Once reaction is successful, upscale amount of material for future experiments. Reaction Between Amine and Acetyl Chloride • Equation: O O H N O NH Cl H N O OH O CH2Cl2/ Reflux N OH O tert-butyl 3-(hydroxyamino)propylcarbamate • Results: • 1st attempt: No distillation or either substrate nor solvent. Some reaction occurred, but it was not complete, even after adding 10% base to help push reaction. • 2nd attempt: Distilled Chloride and Solvent (CH2Cl2). Reaction completed. Rxn. time estimate: ~0.5 hr. Equations for Other Hydroxamic Acid Analogs With Butyryl Chloride o O OH NH O NH O O Cl NH NH O CH2Cl2/ Reflux O With Crotonyl Chloride o O OH NH O NH O Cl NH NH O CH2Cl2/ Reflux O With Hydrocinnamoyl Chloride o O OH O NH NH Cl O O NH NH O CH2Cl2/ Reflux O Reaction Between Amine and Cinnamoyl Chloride • Equation: O H Cl N O H N O H N O N OH H N O OH O O O Reflux O N OH • Result: Did not completely react even after distilling both chloride and solvent. • 2 ways attempted to push reaction - No heat, extra base added - With heat and extra base added. Procedure (Cont.) • Once the analogs were synthesized, solvent was removed with rotavapor and oil pump • 1H-NMR, 13C-NMR and Mass Spec. were taken of each derivative to check purity. O 1H-NMR of H N O N OH O Four Final Analogs Used for Iron Acquisition Measurement O H N O O N OH O H N O N OH O O H N O N OH O O H N O N OH O Procedure for Measuring the Iron-Acquisition Rate • Iron stock solution was made (.6394g/200ml distilled water) • 10mM Buffer solution -Ammonium Fe(III) Citrate (FAC) w/Hepes buffer • Used SF 61 DX2 Mixing Stopped-Flow Machine • Conditions: - pH of buffer solution: 7.4 - Temp-37oC Results of iron acquisition of hydroxamate analogs O O 0.2 H N O O N HC CR OH O O H N Iron-acquisition Rate / s -1 O O H N N OH AC BT N OH O O H N O O N OH 0.15 0.1 0.05 0 0 1 2 3 Fac Concentration / mM 4 5 Results and Conclusions • There was a general curve that formulated for each analog. • The shorter the substrate, the higher the iron acquisition rate. • Having a unsaturated substrate also increased the iron-acquisition rate. Future Studies Adjust conditions to successfully react the following: 2 equi. BPO 10% TEA/MeOH H2N NH2 (BOC)2O HN O NH2 HN O O N H OBz O Cl CH2Cl2/ Reflux O H N O O O N OH NaOH/ MeOH HN N O O OBz O To continue to further examine the chelating process of iron acquisition due to hydroxamic acid structure. Acknowledgements Dr. John T. Groves Princeton University Department of Chemistry Minkui Luo Dr. Groves’ Research Team Mercer County Community College Prof. Helen Tanzini Family and Friends THANK YOU ALL FOR YOUR SUPPORT!