Chemical Equation
advertisement

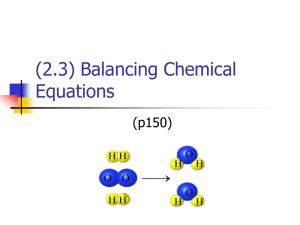
Chemical RXNs SIGNS OF CHEMICAL RXNS Color change - new substances might reflect different colors of light. Heat change - the container may get hotter or colder. Gas produced - bubbles or smoke may be observed. Precipitate formed - an insoluble solid might form in a liquid. CONDITION FOR A CHEMICAL RXN: •reactants have ability to form chemical bonds. •Bonds are formed by valence electrons of reactant atoms. “If valence electrons of substances brought together do not match to complete the Octet Rule, there will be no chemical reaction”. Parts of a rxn: Reactants •Substances that interact with each other; left side of the arrow in a chemical equation. Products •New substances formed during a reaction; right side of the arrow in a chemical equation. Reactants Products TYPES OF CHEMICAL REACTIONS A +B C C A + B FORM SEPARATE SWITCH 1 A + BX AX + B AX + BY AY + BX SWITCH 2 Chemical reaction types: synthesis, decomposition, single replacement, double replacement, and combustion. Synthesis: two simple substances combine to form a more complex one. Example: element + element A + B compound C 2Na + Cl2 4Fe + 3O2 2NaCl 2Fe2O3 REACTION OF COPPER POWDER •WORD EQUATION •CHEMICAL EQUATION •BALANCED CHEMICAL EQUATION REACTION OF COPPER POWDER 2Cu + O2 (2CuO) Copper Oxide (2CuO) Pesticide Fungicide TYPES OF CHEMICAL REACTIONS A +B C C A + B FORM SEPARATE SWITCH 1 A + BX AX + B AX + BY AY + BX SWITCH 2 Chemical reaction types: synthesis, decomposition, single replacement, double replacement, and combustion. Synthesis: two simple substances combine to form a more complex one. Example: element + element A + B compound C 2Na + Cl2 4Fe + 3O2 2NaCl 2Fe2O3 REACTION OF COPPER POWDER •WORD EQUATION •CHEMICAL EQUATION •BALANCED CHEMICAL EQUATION REACTION OF COPPER POWDER 2Cu + O2 (2CuO) Copper Oxide (2CuO) Pesticide Fungicide Decomposition: a complex substance breaks down into two simple substances. compound C 2H2O H2CO3 element + element A + B 2H2 + O2 H2O + CO2 Lab: Separating Water What will happen to water when an electric current is allowed to run through it? MAKING OBSERVATIONS What predictions can you make based on the given information? • What do you think will happen when water separates? • What do you expect to observe during the separation? • Is there a way to identify hydrogen? oxygen? • What do you know about H and O2? What phase of matter will they exist? 2H2O --> 2H2 + O2 Single replacement: a single element replaces an element in a compound. element + compound Zn + 2HCl 2Na + 2H2O compound + element ZnCl2 + H2 2NaOH + H2 SINGLE REPLACEMENT: Reaction of Metals (Mg, Al, FE, Cu, Zn) to HCl Metal Word/Chemical Equation Zinc Zn + 2HCl H2 + ZnCl2 Iron (II) Magnesium Aluminum Chemical Equations 1 WORD EQUATION Copper oxide (ore) plus hydrogen gas produce copper metal plus water 2 CHEMICAL EQUATION CuO + H2 Cu + H2O Coefficient 5H2 Subscript Element BALANCED EQUATION + --> Reactants Products Reactants Products CuO + H2 --> Cu + H2O BALANCED EQUATION 1 copper 1oxygen 2 hydrogen 1 copper 2 hydrogen 1 oxygen BALANCED EQUATION + --> Reactants Products Reactants Products 2 H2 + O2 --> BALANCED EQUATION Reactants Products __H2 +__ O2 --> __H2O BALANCED EQUATION Reactants Products 2 H2 + O2 --> 2 H2O BALANCED EQUATION 4 hydrogen 2 oxygen 4 hydrogen 2 oxygen Chemical Equation: 2 H2 + O2 --> 2 H2O a way to represent chemical reactions on paper. Law of conservation of mass: “During a chemical rxn, atoms are neither created nor destroyed. The number of atoms remains constant throughout the reaction. Since the number of atoms doesn't change, the mass must remain constant as well”. There are times when mass might appear to change during a chemical reaction. If you experience this during an experiment, be sure to remember the law of conservation of mass. The change in mass must be accounted for in a way other than destroying or creating atoms. Equations must be balanced because: Atoms can be neither created nor destroyed in an ordinary chemical reaction, so there must be the same number of atoms on both sides of the equation. Balance equations by changing coefficients never by changing formula subscripts Interactive Tutorials on Balancing Equations Click on this site: GenChem - Balance Equation http://www.wfu.edu/~ylwong/balanceeq/balanceq.html