ELAInteractiveVideo_G8
advertisement

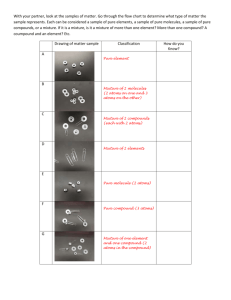
Interactive Video Script Template Course Science 7 Lesson Objective Semester A Unit 4 Lesson 3 Students will define the terms element, compound and mixture. CLIP A (Introduction) Visual <image> Audio Matter is spread all throughout the universe. It consists of anything that takes up space and has mass. http://commons.wikimedia.org/wiki/File:Rh o_Ophiuchi.jpg <image> In our solar system 99.8% of the matter is located in the sun. It is primarily made out of the element hydrogen as plasma. http://pixabay.com/en/sunset-sealandscape-317042/ <image> http://pixabay.com/en/iceberg-antarcticapolar-blue-ice-404966/ <image> Matter exists on Earth in primarily 3 states. In a solid, the molecules are fixed in place. Liquid molecules can move about. Gases are the third state of matter. Gas molecules move even faster. As atoms or molecules get heated or cooled they may switch between these states. http://pixabay.com/en/old-faithful-geyserfamous-landmark-403224/ <image> This is sulfur. It is an element because all of its atoms are the same. Each has 16 protons. http://commons.wikimedia.org/wiki/File:Sul fur-sample.jpg <image> http://pixabay.com/en/lump-sugar-sugarcubes-white-sweet-549096/ Sugar, on the other hand is a compound. All of its molecules have the same ratio of carbon, hydrogen and oxygen. Mixtures occur when more than one element of a compound is mixed but no chemical bonds are formed. <image> http://commons.wikimedia.org/wiki/File:Pla nters-Trail-Mix.jpg In some mixtures, it’s easy to see the different parts. Trail mix is a good example of this type. In others, like this juice, it’s not. <image> http://pixabay.com/en/beverage-diet-drinkfemale-fresh-15706/ Question A Stem: In this type of matter all of the atoms are identical. Answer Choices: A. Element B. Compound C. Mixture D. Plasma Correct Response (A) (Video progresses to clip B) Incorrect Response (other responses) (Video progresses to clip E) CLIP B (DOK1) Visual Audio <image> Elements are substances that are composed entirely of the same type of atoms, defined by the number of protons. http://pixabay.com/en/silver-metalmetallic-grey-gray-576793/ <superimpose “Ag (silver) atoms all have 47 protons” over the image> This bar of silver has the same number of protons in each atom. They each contain 47. <image> This open sign is a tube filled with neon gas. The element neon glows red when an electric current passes through it. http://commons.wikimedia.org/wiki/File:Is_ Open.jpg <superimpose “10 protons per atom” over the red part of the neon sign> The atoms inside the tube are all the same type. They each contain 10 protons. Question B Stem: What defines what element an atom is? Answer Choices: A. number of protons B. number of electrons C. number of neutrons D. mass of the atom Correct Response (A) (Video progresses to clip C) Incorrect Response (other responses) (Video progresses to clip F) CLIP C (Increased DOK2) Visual <image> Audio The space shuttle runs on liquid hydrogen fuel. This fuel is ignited and combined with an oxidizer which injects pure oxygen. https://commons.wikimedia.org/wiki/File:0 20408_STS110_Atlantis_launch.jpg <superimpose formula “H2 + O2 H2O + heat” over the flames> As the fuel burns the hydrogen and oxygen form bonds and the atoms of hydrogen and oxygen form a molecule. <image> Each molecule formed from this burning is water. It is a compound because each molecule has the same ratio of atoms. http://pixabay.com/en/drop-of-watercondensation-fractal-566885/ <superimpose “H2O=2 hydrogen atoms and 1 oxygen atom” over the image> Every molecule in a compound has the same ratio of atoms. Water has a 2:1 hydrogen to oxygen ratio. Question C Stem: Which type of substance is made of molecules with the same ratio of atoms? Answer Choices: A. Element B. Compound C. Homogeneous mixture D. Heterogeneous mixture Correct Response (B) (Video progresses to clip D) Incorrect Response (other responses) (Video progresses to clip G) CLIP D (Increased DOK3) Visual <image> http://pixabay.com/en/asphalt-crackbackground-line-road-315756/ <point arrows to a few of the different sized substances in the road> Audio Mixtures are all around us. They exist whenever more than one substance is combined without forming new molecules. This road is a heterogeneous mixture. It is not evenly mixed. There are particles of all different sizes. <image> http://pixabay.com/en/drink-soda-glassthirst-beverage-341489/ <image> http://pixabay.com/en/liquid-red-glassdrink-juice-pour-166772/ Soda is an example of a mixture. It is composed of sugar, water, carbon dioxide and other flavorings mixed together. Juice is also a mixture. These drinks are so evenly mixed that it is hard to tell the different parts. It’s a homogeneous mixture. Question D Stem: What kind of mixture is not evenly mixed? Answer Choices: A. Homogeneous mixture B. Heterogeneous mixture C. Solution D. Alloy Correct Response (B) (Video progresses to Success Alert) Incorrect Response (other responses) (Video progresses to clip H) CLIP E (Remedial 1) Visual Audio <image> Matter is anything that takes up space and has mass. Salt is a compound because all of its molecules are the same. http://pixabay.com/en/salt-nature-eat-foodspice-91539/ <superimpose “1 Na and 1 Cl per molecule” over the image> Each salt molecule has one sodium and one chlorine atom. Salt has very different properties from its parent elements. <images side by side> By themselves both sodium and chlorine are highly reactive. When they are in a compound, though, their properties change. http://commons.wikimedia.org/wiki/File:So dium.jpg http://commons.wikimedia.org/wiki/File:Chl orine-sample.jpg <image> Mixtures are made when matter is mixed together but the molecules are not combined into new types of molecules. http://pixabay.com/en/blender-mixerjuicer-food-processor-10934/ Question E Stem: What type of matter is composed of different substances that have not formed chemical bonds? Answer Choices: A. Elements B. Compounds C. Mixtures D. Liquids Correct Response (C) (Video progresses to clip B) Incorrect Response (other responses) (Video progresses to clip F) CLIP F (Remedial 2) Visual Audio <image> http://pixabay.com/en/balloons-colorfulhappy-party-466169/ These balloons are filled with the gas form of the element helium. That means that all of the atoms in the balloon will be identical. <superimpose “2 protons per He atom” over the balloons> The elemental identity of an atom is determined by the number of protons in the nucleus, these have 2 each. <image> Pencil lead is composed of carbon atoms. They are no longer made from actual lead. The atoms are loosely compacted and good for marking. http://pixabay.com/en/school-pen-greatpointed-leave-93200/ <image> In graphite, each atom contains 6 protons. The atoms of a diamond also have 6 each. They’re both made of carbon, but they are http://pixabay.com/en/diamond-gem-cubic- not quite the same! zirconia-jewel-123338/ Question F Stem: Which type of substance contains atoms that contain the same number of protons? Answer Choices: A. Homogeneous mixture B. Heterogeneous mixture C. Element D. Compound Correct Response (C) (Video progresses to clip C) Incorrect Response (other responses) (Video progresses to Intervention Alert, bringing students back to clip B) CLIP G (Remedial 3) Visual <image> Audio The rust on these chains is made of a combination of iron and oxygen. http://pixabay.com/en/chain-rust-pastbondage-history-232930/ <image> It is a compound that forms when pure iron is combined with oxygen. Iron oxide is made of the elements iron and oxygen. http://pixabay.com/en/iron-chains-chainschain-link-steel-502883/ <superimpose “iron + water +oxygen Chemical reactions must occur in order for rust + water” over the image of the chains> new compounds to form. In this case the element oxygen is added to iron. <image> New compounds are continuously forming through reactions that are rearranging the ratio of atoms in molecules. http://www.morguefile.com/archive/display /151843 Question G Stem: What happens when the ratios of atoms in a molecule changes? Answer Choices: a. New compounds form b. New elements form c. Oxygen is destroyed d. Iron is destroyed Correct Response (A) (Video progresses to clip D) Incorrect Response (other responses) (Video progresses to clip F) CLIP H (Remedial 4) Visual Audio <image> The water in the ocean is an example of a mixture. It is hard to tell what the different parts are because it is so evenly mixed. http://commons.wikimedia.org/wiki/File:Ale utian_islands_ocean_water.jpg <superimpose salt + water = salt water mixture> The salt and water in the ocean can be separated by evaporating off the water in a sample. <image> If you were to go from the ocean to the beach you would notice that the sand is also a heterogeneous mixture. http://pixabay.com/en/rocks-beach-windsand-ocean-555736/ <point arrows to the rocks on the beach> This is because there are many different types of materials in the sand, which is mixed unevenly. Question H Stem: Which type of mixture is mixed so evenly that the individual parts cannot be seen? Answer Choices: A. Heterogeneous B. Homogeneous C. Element D. Compound Correct Response (B) (Video progresses to Success Alert) Incorrect Response (other responses) (Video progresses to clip G)