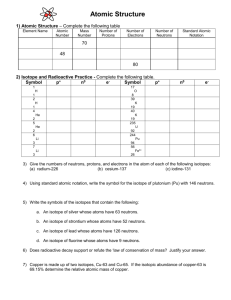
PP - myndrs.com
advertisement

Chapter 7: Radiation Remember from Chemistry: • Mass number: the number of protons + number of neutrons • Atomic number: the number of protons in an atom of an element. • Isotopes: atoms of the same elements that have a different number of neutrons and therefore a different mass number There are many isotopes of carbon. • For example: carbon-12, carbon-13 and carbon-14 • How many protons in each of these atoms of carbon? – 6 • How many neutrons in each of these atoms of carbon? • Carbon-12 = ____ neutrons • Carbon-13 = ____ neutrons • Carbon-14 = ____ neutrons Standard Atomic Notation • Carbon-12 = 12 C 6 • Carbon-13 = _____ • Carbon-14 = _____ Mass number Atomic number Isotopes of Hydrogen • Hydrogen: 1 proton and 0 neutrons • Deuterium: 1 proton and 1 neutron • Tritium: 1 proton and 2 neutrons Radioactive Decay: • The nuclei of some isotopes are stable, whereas, others are not. • In unstable nuclei, the nuclear force is not enough to overcome the repulsive electrical forces with in the nucleus. • As a result the nuclei are radioactive. • When an unstable nucleus emits radiation, it undergoes a radioactive decay Types of radioactive decay • Alpha Decay: • Particle emitted: Helium nucleus • Result: Atom with two fewer protons and neutrons Sample Equations: Parent isotope Daughter isotope • U → _____ + He • U → Th + _____ Types of radioactive decay • Beta • Particle emitted: Electron • Result: # of neutrons decrease by one and the # of protons increase by one = new element Sample Equations: Parent isotope Daughter isotope • C → ____ + e • C → N + ____ Types of radioactive decay • Gamma • Particle emitted: No particle, but gives off excess energy • Result: No change to type of nucleus, it just has less energy. Sample Equations: Parent isotope Daughter isotope • Ba → _____ + Gamma radiation • Ba → Ba + ______ On your data sheets: p12 Radiation Assignment: • Fill in the 7.1 worksheets p123-5 (all) and p126 (pick two alpha, two beta and two gamma questions) • You are going to have to look up some of this information in your textbook • Quiz next class – Identifying and completing decay equations – Given # of neutrons and number of protons can you determine, mass #, atomic #, the element, isotope name and isotope symbol. Half Life Section 7.2 PLO: Unit 2 #5.2 Half Life: • Half Life: The time required for half the nuclei of a sample of a radioactive isotope to decay. • The half life is the same regardless of the starting amount of the isotope. • Because the amount of all radioactive isotopes decrease by half in each half life, graphs that show the amounts of these isotopes at various times look similar. • The only difference is the time scale for decrease. Examples of half life: 120 100 80 60 40 20 0 carbon 0 Uranium 20000 40000 60000 80000 Time (Years) sample of uranium amount of sample left carbon 600 500 400 300 200 100 0 Uranium 0 50000 100000 Time (years) 150000 Half Life Problems • Figure out what the problem is asking you to find. • Fill in the table with the information given to you in the problem. Half Life Table for Solving Word Problems Time # of Half lives Amount of parent isotope ** optional ** Amount of daughter isotope 0 0 Original Amount 0 1 2 3 4 Homework: • Fill in the 7.2 worksheets p134 (odd) and 135 • For p 134 you must show the half life tables Half Life Table for Solving Word Problems Time # of Half lives Amount of parent isotope ** optional ** Amount of daughter isotope 0 0 Original Amount 0 1 2 3 4

![tutorial #14 [nuclear physics and radioactivity] .quiz](http://s3.studylib.net/store/data/008407305_1-1884988a9e5162a6b7a2b0d0cf8c83c5-300x300.png)