Experiment 2 Physical Properties
advertisement

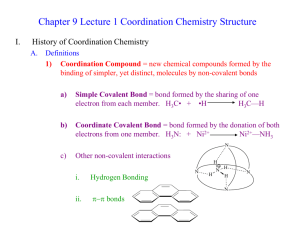
Chapter 22 Transition Elements Transition Elements – d- and f-block • Used in construction and manufacturing (iron), coins (nickel, copper, zinc), ornamental (gold, silver, platinum). • Densest elements (osmium d=22.49 g/cm3, iridium d=22.41g/cm3). • Highest melting point (tungsten, mp=3410oC) and lowest melting point (mercury, mp=-38.9oC). Metal Chemistry • Radioactive elements with atomic number less than 83 (technetium 43; promethium 61). • All elements are solids, but mercury. • Have metallic sheen, conduct electricity and heat. • Are oxidized and form ionic compounds. • Some are essential to living organisms: Cobalt (vitamin B12), iron (hemoglobin and myoglobin), molybdenium and iron (nitrogenase). • Compounds are highly colored and used as pigments: Fe4[Fe(CN)6)3 14 H2O (prussian blue), TiO2 (white). • Ions give color to gemstons: Iron(II) ions give yellow color in citrine and chromium(III) ions produce the red color of a ruby. Electron Configurations • General: [noble gas core] nsa (n-1) db • Valance electrons for transition elements reside in the ns and (n-1) d subshells. Reactions • All metals undergo oxidation with oxygen, halogens, aqueous acids. • First the outermost electron is removed, followed by one or more d electrons. • Some generate cations with unpaired electrons = paramagnetism. • Are colored. • For first transition series common oxidation numbers are +2 and +3. Fe: [Ar]3d64s2 Fe + Cl2 Fe + HCl Fe + O2 Fe + Cl2 Fe + HCl Fe + O2 Trends: Oxidation number Most common Trends: Atom Radius Trends: Density Trends: Melting Point Metallurgy: Element Sources Pyrometallurgy • Involves high temperature, such as Fe • C and CO used as reducing agents in a blast furnace Fe2O3 + 3 C ---> 2 Fe + 3 CO Fe2O3 + 3 CO ---> 2 Fe + 3 CO2 • Lime added to remove impurities, chiefly SiO2 SiO2 + CaO ---> CaSiO3 • Product is impure cast iron or pig iron Hydrometallurgy • Use aqueous solutions (flotation). Some use bacteria. • Add CuCl2(aq) to ore such as CuFeS2 (chalcopyrite) CuFeS2(s) + 3 CuCl2(aq) --> 4 CuCl(s) + FeCl2(aq) + 2 S(s) • Dissolve CuCl with xs NaCl CuCl(s) + Cl-(aq) --> [CuCl2]• Cu(I) disproportionates to Cu metal 2 [CuCl2]- --> Cu(s) + CuCl2 (aq) + 2 Cl- Azurite, 2CuCO3•Cu(OH)2 Native copper Coordination Compounds – Definition: __________________________ ___________________________________ ___________________________________ _____________________. •• H N H H •• H O H •• CH2 CH2 - Cl Pt Cl Cl Coordination Chemistry Pt(NH3)2Cl2 Co(H2O)62+ “_____________” - a cancer chemotherapy agent Cu(NH3)42+ Coordination Chemistry An _______________, the basic unit of hemoglobin Myoglobin / Hemoglobin p.1084 Coordination Chemistry Vitamin ________ A naturally occurring cobalt-based compound Co atom Coordination Chemistry • Biological nitrogen fixation contributes about half of total nitrogen input to global agriculture, remainder from Haber process. • To produce the H2 for the Haber process consumes about 1% of the world’s total energy. • A similar process requiring only atmospheric T and P is carried out by N-fixing bacteria, many of which live in symbiotic association with legumes. • N-fixing bacteria use the enzyme nitrogenase — transforms N2 into NH3. • Nitrogenase consists of 2 metalloproteins: one with Fe and the other with Fe and Mo. Coordination Chemistry Nickel ion: coordination compounds Nomenclature • [Ni(NH3)6]2+ • A Ni2+ ion surrounded by ____ __________________ • Gives coordination complex ion with ______charge. Ligand: monodentate Coordinate to the metal via a single Lewis base atom. Nomenclature Inner coordination sphere Ligand: polydentate also chelating ligands Coordinate with more than one donor atom. (Bidentate) Co3+ + 2 Cl- + 2 neutral ethylenediamine molecules Cis-dichlorobis(ethylenediamine)cobalt(II) chloride + Cl- Bidentate Ligands Acetylacetone (acac) Ethylenediamine (en) Bipyridine (bipy) Oxalate (ox) Bidentate Ligands Acetylacetonate Complexes Commonly called the “acac” ligand. Forms complexes with all transition elements. Multidentate Ligands EDTA4- - ethylenediaminetetraacetate ion Multidentate ligands are sometimes called ________________ ligands Multidentate Ligands Co2+ complex of EDTA4- Give the formula of a coordination compound A Co3+ ion bound to one Cl- ion, one ammonia molecule, and two ethylenediamine (en) molecules. 1. 2. Determine the net charge (sum the charges of the various components). Place the formula in brackets and the net charge attached. [Co(H2NCH2CH2NH2)2(NH3)Cl]2+ Determine the metal’s oxidation number and coordination number Pt(NH3)2(C2O4) [Co(NH3)5Cl]SO4 Nomenclature Cis-dichlorobis(ethylenediamine)cobalt(III) chloride 1. Positive ions named first 2. Ligand names arranged alphabetically 3. Prefixes -- di, tri, tetra for simple ligands bis, tris, tetrakis for complex ligands 4. If M is in cation, name of metal is used 5. If M is in anion, then use suffix -ate CuCl42- = tetrachlorocuprate 6. Oxidation no. of metal ion indicated in roman numerals. Nomenclature Co(H2O)62+ Hexaaquacobalt(II) H2O as a ligand is aqua Pt(NH3)2Cl2 Cu(NH3)42+ Tetraamminecopper(II) diamminedichloroplatinum(II) NH3 as a ligand is ammine Nomenclature Tris(ethylenediamine)nickel(II) [Ni(NH2C2H4NH2)3]2+ IrCl(CO)(PPh3)2 Vaska’s compound Carbonylchlorobis(triphenylphosphine)iridium(I) Geometry of Coordination Compounds Defined by the arrangement of donor atoms of ligands around the central metal ion. Valdosta State University Isomerim of Coordination Compounds • Two forms of isomerism – Constitutional – Stereoisomerism • Constitutional – Same empirical formula but different __________ _________________________________. • Stereoisomerism – Same atom-to-atom connections but different ____________________________________. Geometric and Optical Constitutional Isomers O Aldehydes & ketones CH3-CH2-CH O H3C C CH3 3C, 1O, 6H - Coordination isomerism: it is possible to exchange a ligand and the uncoordinated counterion. Example: [Co(NH3)5Br]SO4 and [Co(NH3)5SO4]Br (violet) (red) - Linkage isomerism: it is possible to attach a ligand to the metal through different atoms. Usually: SCN- and NO2- Constitutional Isomers NH3 2+ H3N NO2 sunlight Co H3N NH3 NH3 2+ NH3 H 3N ONO Co H3N NH3 NH3 Such a transformation could be used as an energy storage device. Pentaamminenitritocobalt(III) Pentaamminenitrocobalt(III) Stereoisomerism • One form is commonly called geometric isomerism or cis-trans isomerism. Occurs often with square planar complexes. cis trans Note: there are VERY few tetrahedral complexes. Would not have geometric isomers. Geometric Isomers Cis and trans-dichlorobis(ethylenediamine)cobalt(II) chloride Geometric Isomers For octahedral complexes (MX3Y3): fac isomer has three identical ligands lying at the corners of a triangular face of octahedron (fac=facial). mer isomer ligands follow a meridian (mer=meridional). fac isomer mer isomer Stereoisomers • Enantiomers: stereoisomers that have a nonsuperimposable mirror image. • Diastereoisomers: stereoisomers that do not have a non-superimposable mirror image (cistrans isomers). • Asymmetric: lacking in symmetry—will have a non-superimposable mirror image. • Chiral: an asymmetric molecule. Enantiomers [Co(NH2C2H4NH2)3]2+ Stereoisomers [Co(en)(NH3)2(H2O)Cl]2+ N N Cl Co 2+ NH3 NH3 OH2 NH3 2+ N NH3 Co N Cl OH2 NH3 2+ These two isomers N Cl have a plane of Co N OH2 symmetry. Not chiral. NH3 NH3 2+ N NH3 Co N OH2 Cl These two are asymmetric. Have non-superimposable mirror images. Stereoisomers [Co(en)(NH3)2(H2O)Cl]2+ These are non-superimposable mirror images: enantiomers Bonding in Coordination Compounds • Model must explain – – – – Basic bonding between M and ligand Color and color changes Magnetic behavior Structure • Two models available – Molecular orbital – Electrostatic crystal field theory – Combination of the two ---> ligand field theory Bonding • As ligands L approach the metal ion M+, – L/M+ orbital overlap occurs – L/M+ electron repulsion occurs • Crystal field theory focuses on the latter, while MO theory takes both into account Ligand Field Theory [Ar] 4s All electrons have the same energy in the free ion five 3d orbitals • Consider what happens as 6 ligands approach an Fe3+ ion: Orbitals split into two groups as the ligands approach. energy t2g eg d(x2-y2) dxy dxz dz2 dyz D0 Value of ligand field sppliting: ∆o depends on L: e.g., CN- > H2O > Cl- Octahedral Ligand Field Tetrahedral and Square Planar Ligand Fields Crystal Field Theory • Tetrahedral ligand field. • Note that ∆t = 4/9 ∆o and so ∆t is small. • Therefore, tetrahedral complexes tend to absorb “red wavelengths” and be colored blue. energy e t2 dxy dxz d(x2-y2) dyz dz2 Dt Ways to Distribute Electrons • For 4 to 7 d electrons in octahedral complexes, there are two ways to distribute the electrons. – High spin — maximum number of unpaired e– Low spin — minimum number of unpaired e- • Depends size of ∆o and P, the pairing energy. • P = energy required to create e- pair. Magnetic Properties of Fe2+ energy eg t2g • High spin d(x2-y2) dxy dz2 DE small dxz dyz 2 2 Paramagnetic energy t2g eg 2 d(x -y ) dxy dxz dz dyz Diamagnetic • Weak ligand field strength and/or lower Mn+ charge • D0 is smaller than P • [Fe(H2O)6]2+ • Low spin D E large • Stronger ligand field strength and/or higher Mn+ charge • D0 is larger than P • [Fe(CN)6]4- High and Low Spin Octahedral Complexes High or low spin octahedral complexes only possible for d4, d5, d6, and d7 configurations. Why are complexes colored? Fe3+ Co2+ Ni2+ Cu2+ Zn2+ Why are complexes colored? – Note that color observed is transmitted light. Red and blue are absorbed Why are complexes colored? – Note that color observed is transmitted light. – Color arises from electron transitions between d orbitals (d-to-d transitions). – Color often not very intense. • Spectra can be complex – d1, d4, d6, and d9 --> 1 absorption band – d2, d3, d7, and d8 --> 3 absorption bands • Spectrochemical series — ligand dependence of light absorbed. The ability to split the d orbitals is determined by spectroscopy. Light absorption by octahedral Co3+ complex d(x -y ) dxz dxy dz2 Ground state + energy (= D0 ) dyz 2 (light absorbed) t2g eg d(x2-y2) dxy t2g 2 dz2 dxz eg energy energy Excited state Usually excited complex returns to ground state by losing energy, which is observed as heat. dyz Spectrochemical Series • d orbital splitting (value of ∆o) is in the order: small ∆o large ∆o I- < Cl- < F- < H2O < NH3 < en < phen < CN- < CO As ∆ increases, the absorbed light tends to ________(red or blue), and so the transmitted light tends to _________ (red or blue). Other ways to induce color • Intervalent transfer bands (IT) between ion of adjacent oxidation number. – Aquamarine and kyanite are examples – Prussian blue • Color centers – Amethyst has Fe4+ – When amethyst is heated, it forms citrine as Fe4+ is reduced to Fe3+ Prussian blue contains Fe3+ and Fe2+ End of Chapter • Go over all the contents of your textbook. • Practice with examples and with problems at the end of the chapter. • Practice with OWL tutor. • Practice with the quiz on CD of Chemistry Now. • Work on your OWL assignment for Chapter 22. 22.4. Match up the isoelectronic ions on the following list: Cu+ Mn2+ Fe2+ Co3+ Fe3+ Zn2+ Ti2+ V3+ 22.2. Identify two transition metal ions with the following electron configurations. (a) [Ar]3d 6 ;(b) [Ar]3d 10 ; (c) [Ar]3d 5 ; (d) [Ar]3d 8 22.8. One of the following nitrogen compounds or ions is not capable of serving as a ligand: NH4+, NH3, NH2−. Identify this species and explain your answer. 22.10. Give the oxidation number of the metal ion in each of the following complexes. 22.20. In which of the following complexes are geometric isomers possible? If isomers are possible, draw their structures and label them as cis or trans, or as fac or mer.