Spectroscopy: Element Identification and Emission Spectra
advertisement

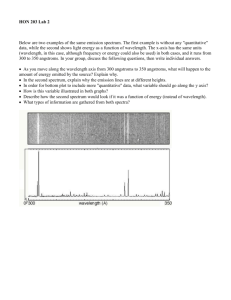
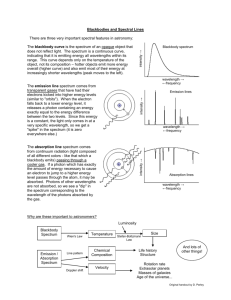
Spectroscopy: Element Identification http://www.800mainstreet.com/spect/emission-flame-exp.html Part 1: Flame Tests & Identification of an Unknown Metal: Use the simulation to view flame tests for some common metallic elements. Complete the data and analysis below. Name of Metallic Observed Flame color ion barium _________________________ calcium _________________________ sodium _________________________ rubidium _________________________ potassium _________________________ lithium _________________________ Unknowns Flame color Identity of metallic ion based on flame test Unknown 1 ____________ __________ Unknown 2 ____________ __________ Part 2: Observing Emission Spectra with the Spectroscope Examine the spectra for the elements Na, Ne, Hg, and He and compare them in terms of colors and numbers of emission line positions: What is similar among them? What is different? Examine the spectra for the elements Na, Ne, Hg, and He and answer the following questions. 1) Which element has the greatest number of visible emission lines: _____ Which element has an emission line with the longest wavelength: ____ What color of light corresponds to this element’s longest emission wavelength: ____ 2) What element has the fewest number of emission lines: ____ What color of light corresponds to this element’s longest emission wavelength: _____ What color of light corresponds to this element’s shoetest emission wavelength: _____ Post-Lab Questions: 1) Both flame testing and spectroscope analysis can be used to identify unknown elements. Would spectroscope analysis or flame testing provide a more accurate method for identification? Why? 2) Many cities have street lights that are filled with sodium vapor. The light appears yellow instead of white. Look at the emission spectrum of sodium and tell why. 3) What would happen to the total size of a hydrogen atom if its electron moves from n=1 to n=4? Why wouldn’t that electron remain at n=4? (Remember, “n” stands for a principal energy level.) Spectroscopy: Emission Spectra 1) Color the space below to correspond to a continuous spectrum. Refer to the electromagnetic spectrum that is being shown in class. Be sure the wavelengths and colors match. Continuous Spectrum: . . . 4000A (400 nm) . . . . . . . . . . 5000A (500 nm) . . . . . . . . . 6000A (600 nm) . . . . . . . . . 7000A (700 nm) Go to the site: http://jersey.uoregon.edu/vlab/elements/Elements.html 2) Toggle between the absorption spectrum and the emission spectrum buttons. How are the absorption spectrum and the emission spectrum of an element related? How is the continuous spectrum similar to an emission spectrum? How is the continuous spectrum different from an emission spectrum? Why are there black spaces in an emission spectrum? What feature of atomic structure relates to these spaces? 3) What is the difference between the ground state and an excited state for an electron? Why is the ground state more stable than an excited state? What makes electrons move to an excited state? What happens when they fall back to ground state? 4) Are electrons in ground state or in excited state when electricity is not passing through them? What color is hydrogen gas when electricity is not passing through it? ________ What happens to hydrogen’s electron when electricity is passed through it? Are electrons in ground state or in excited state when electricity is being passing through them? What happens to hydrogen’s electron after it reaches excited state? Why does the hydrogen produce visible light? What color is the light produced by hydrogen’s electron when it falls back to ground state? 5) Use the periodic table on the website to view the emission spectrum of hydrogen and of helium. Copy the spectra in the spaces provided. Use accurate colors and accurate placements of the bright lines. Record the value of each wavelength. Note: In order to see the wavelength of a line place the cursor on the line and right-click. The wavelength in angstroms will appear. (1 m = 1x1010A = 1x109 nm. So, 10 A = 1 nm) Hydrogen: . . . 4000A . . . . . . . . . 5000A . . . . . . . . . . 6000A . . . . . . . . . 7000A . . . . . . . . . 5000A . . . . . . . . . . 6000A . . . . . . . . . 7000A Helium: . . . 4000A 5) Analysis of the hydrogen spectrum: Text section 5.3 will help with the discussion. a. Review the relationship between wavelength and frequency. Select ONE of the lines on the hydrogen spectrum – be sure you know its wavelength. Use the wavelength to calculate the frequency for that line. Show how the calculation is done – be sure to use unit labels. b. Use the frequency to calculate the energy value for the same line you selected in (5a) above. Show how the calculation is done – be sure to use unit labels. c. What information does this line tell scientists about the most likely placement of energy levels in a hydrogen atom? 6) Electrons can emit electromagnetic radiation that is not visible as a color. For example, ultra-violet light can be emitted by excited electrons. How do we interpret this in terms of the placement of energy levels? 7) Why does each element have a completely unique emission spectrum? 8) Complete this sentence: An element’s emission spectrum provides evidence of energy levels because….