Patients with psoriasis do not have detectable T cells to psoriasin
advertisement

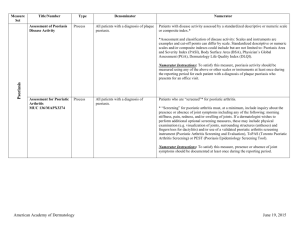
CLINICAL AND LABORATORY INVESTIGATIONS Detection of psoriasin/S100A7 in the sera of patients with psoriasis K.S. Anderson*†, J. Wong*, K. Polyak†, D. Aronzon* and C. Enerbäck‡ *Cancer Vaccine Center and †Department of Medical Oncology, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA 02115, U.S.A. ‡Department of Clinical Genetics and Department of Dermatology, Sahlgrenska University Hospital, S-41345 Göteborg, Sweden Correspondence to Karen S. Anderson. E-mail: kanderson@partners.org Conflicts of interest None declared. Copyright Journal Compilation © 2009 British Association of Dermatologists KEYWORDS autoantibodies • biomarker • psoriasis • S100A7 ABSTRACT Background Psoriasis is a disease of dysregulated inflammation and epithelial hyperproliferation in the skin, involving both the innate and adaptive immune system. Psoriatic keratinocytes express high levels of psoriasin (S100A7), a small calciumbinding protein. Objectives To determine if patients with active psoriasis have elevated serum levels of psoriasin and psoriasin-specific autoantibodies. Methods Blood was collected from 14 patients with psoriasis vulgaris at the start of narrowband ultraviolet (UV) B therapy and from 11 of these patients every 2 weeks during the course of the UVB treatment. Patient and control sera were tested for psoriasin antigen levels by sandwich enzyme-linked immunosorbent assay, and for psoriasin autoantibody titres using recombinant purified psoriasin and overlapping peptides. Results We confirmed strong and specific expression of psoriasin in psoriatic epidermis by immunohistochemistry. Systemic psoriasin antigen levels tended to be lower in patients (mean 213 ng mL−1) than in controls (mean 331 ng mL−1, P = 0·308) and decreased with increasing disease severity. Psoriasin-specific autoantibodies were detected in a subset of patients with psoriasis and healthy normal donors (mean 0·347 vs. 0·255 units, P = 0·246). The epitopes recognized by the autoantibodies were mapped to an external loop domain of the molecule but did not show corresponding Tcell immunogenicity. Conclusions Although psoriasin is overexpressed in psoriatic skin lesions, systemic levels of psoriasin tended to be lower with increasing disease severity, which may be due to the presence of psoriasin-specific autoantibodies. Neither psoriasin nor psoriasin-specific autoantibodies appear to be promising serum biomarkers for clinical psoriasis. Accepted for publication 11 August 2008 DIGITAL OBJECT IDENTIFIER (DOI) 10.1111/j.1365-2133.2008.08904.x About DOI Article Text Psoriasis is considered to be a genetically determined disease of dysregulated epidermal hyperproliferation and inflammation, which is initiated and maintained by multiple mechanisms of the immune system. Recent findings suggest a pathological collaboration between innate immunity (mediated by antigen-presenting cells and natural killer T lymphocytes) and acquired immunity (mediated by T lymphocytes) that results in the inflammatory infiltrate seen in psoriatic plaques.1 Hyperproliferation of the keratinocytes may be induced by the chemokines and cytokines secreted by inflammatory cells. Calcium-binding S100 proteins are involved in many biological processes.2 Several S100 proteins, specifically S100A7 (psoriasin), S100A8 (calgranulin A) and S100A9 (calgranulin B) have been found to be markedly upregulated in psoriatic skin lesions.3,4 The expression of all three S100 proteins was induced by the proinflammatory cytokine interleukin (IL)-22 in psoriatic skin, and the serum level of IL-22 correlated with psoriasis disease severity.5 We and others have previously demonstrated a strong correlation between S100A7 and S100A9 expression and keratinocyte differentiation in normal and psoriatic skin.4,6 Psoriasin expression is also regulated by interferon (IFN)-γ,7 it has been proposed to be a chemoattractant of CD4+ lymphocytes in the skin,8 and it upregulates vascular endothelial growth factor.9 These findings suggest a potential role for psoriasin in the dysregulated differentiation, increased angiogenesis and intense inflammatory reaction characteristic for psoriatic lesions. Elevated antigen levels of S100A8/S100A9 molecules have been detected in the sera of patients with psoriasis and were found to correlate with disease activity.10 As S100A7, S100A8 and S100A9 are coexpressed in psoriatic skin keratinocytes and are implicated in the pathogenesis of psoriasis,11,12 we hypothesized that psoriasin/S100A7 may also be detected in the sera of patients with psoriasis and may be a potential biomarker of disease severity. However, we found that the level of serum psoriasin was decreased in patients with psoriasis and decreased with increasing disease severity. Because psoriasin is partially secreted, we hypothesized that psoriasin may induce psoriasin-specific autoantibodies that could be used as surrogate biomarkers in patients with psoriasis. Secreted antigens, including the extracellular domain of Her2 in breast cancer, can induce autoantibodies that are highly specific and sensitive biomarkers for breast cancer.13 To test this hypothesis, we developed a highly sensitive enzyme-linked immunosorbent assay (ELISA) for the detection of IgG antipsoriasin autoantibodies, confirmed the specificity of these antibodies by Western blotting, and mapped the interactive epitopes using overlapping peptides. A subset of patients with psoriasis and healthy donors had detectable autoantibodies to psoriasin, but this was independent of disease severity and serum psoriasin levels. Although the number of patients analysed was fairly small, based on our results neither psoriasin antigen nor antipsoriasin autoantibodies appear to be promising serum biomarkers for psoriasis. Material and methods Patients and serum samples Patients with psoriasis vulgaris treated at the psoriasis dermatological clinics at the Department of Dermatology, Sahlgrenska University Hospital, Göteborg, Sweden, who had not received systemic (e.g. methotrexate or ciclosporin) or topical (e.g. local glucocorticoids) treatment for at least 4 weeks prior to sample collection, were selected for the study. The study was performed in the autumn and the patients had not received strong summer sun exposure just before the study. Patients with other types of psoriasis (guttate psoriasis and psoriatic arthritis) were not included. Overall 14 patients were analysed, four women and 10 men. The mean age of the patients was 47 years (range 21–64). Patients received narrowband ultraviolet (UV) B treatment 3 days a week for 8–10 weeks. Patients were treated by whole-body exposure using Philips TL-01 lamps. Increments were according to standard procedures. Peripheral blood was collected at the start of and every 2 weeks during the course of UVB treatment. Serum was immediately frozen on liquid nitrogen and stored at −80 °C. Peripheral blood mononuclear cells (PBMC) were isolated using ACCUSPIN™ (Sigma-Aldrich, St Louis, MO, U.S.A.) and cellular viability was confirmed by trypan blue exclusion assay. The study was approved by the ethics committee of Göteborg University and all patients gave written informed consent. Control serum was obtained with informed consent from healthy blood donors at the Dana-Farber Cancer Institute. Immunohistochemistry Tissue samples for immunohistochemical studies were selected from the files of the Departments of Dermatology and Pathology at Sahlgrenska University Hospital. Informed consent was requested from the patients under approval by the ethics committee of the University of Göteborg. Tissue samples were de-identified prior to the study. Immunohistochemical analysis was performed essentially as described.14 S100A7 protein purification and peptides cDNA encoding for full-length human S100A714 was subcloned as an N-terminal Histagged fusion molecule in pQE30 (Qiagen, Valencia, CA, U.S.A.) and expressed in bacteria. Protein was purified under denaturing conditions in 6 mol L−1 guanidine-HCl with 100 mmol L−1 NaCl and 10 mmol L−1β-mercaptoethanol. Protein was purified using a nickel NTA column (Qiagen) using a slow urea step gradient with 0·5 mol L−1 NaCl and 20% glycerol. Protein purity was confirmed by sodium dodecyl sulphate– polyacrylamide gel electrophoresis (SDS-PAGE), and concentration determined by bicinchoninic assay (Pierce Biotechnology, Rockford, IL, U.S.A.). Overlapping peptides (15-mer, overlapping by 10) spanning the full-length molecule were obtained from Invitrogen (Carlsbad, CA, U.S.A.). S100A7–glutathione-S-transferase (GST) fusion proteins and GST control protein were expressed from pGEX4T-1 (Amersham, Piscataway, NJ, U.S.A.) and purified as described.15 Sandwich enzyme-linked immunosorbent assay for the detection of S100A7 in serum Monoclonal antibodies 817, 1068 and 396 were generated by standard immunization with recombinant psoriasin.14 A mixture of two capture antibodies, clones 817 and 1068, each at 2 μg mL−1 in carbonate binding buffer pH 9·6, were coated overnight at 4 °C on to 96-well Nunc-Immuno plates (Nalge Nunc, Rochester, NY, U.S.A.). The wells were blocked with 1% bovine serum albumin, then 50 μL of human serum was added in duplicate to each well at room temperature for 1 h. The detection antibody 396 was biotinylated using an EZ-Link biotinylation kit (Pierce Biotechnology), used at 2 μg mL−1, and detected with streptavidin–horseradish peroxidase (HRP) and SuperSignal ELISA Substrate (Pierce Biotechnology) using a Victor3 plate reader (PerkinElmer, Waltham, MA, U.S.A.). Enzyme-linked immunosorbent assay and Western blot for the detection of S100A7-specific autoantibodies His-tagged psoriasin antigen and peptides were diluted to 5 μg mL−1 in binding buffer and coated overnight at 4 °C on to 96-well Nunc-Immuno plates (Nalge Nunc) as described.15 Plates were blocked with 2% nonfat milk and 50 μL of sera (1 : 300 dilution) was added in duplicates followed by incubation with secondary antibody (goat antihuman IgG-HRP, 1 : 5000; Jackson ImmunoResearch, West Grove, PA, U.S.A.) and detection with TMB+ Substrate (Dako, Carpinteria, CA, U.S.A.). For Western blotting, 10 μg of S100A7-GST or GST antigen was separated by SDSPAGE, transferred to nitrocellulose membrane, immunoblotted with patient sera at 1 : 500 and goat antihuman IgG-HRP (Jackson ImmunoResearch) and detected with ECL reagent per manufacturer's instructions (NEN, Boston, MA, U.S.A.). In vitro stimulation of peripheral blood mononuclear cells and interferon-γ enzyme-linked immunospot assay PBMC from patients with psoriasis were stimulated in cell culture for 1 week in the presence of 10 ng mL−1 IL-7 (Endogen Inc., Woburn, MA, U.S.A.) from day 0 and 20 IU mL−1 IL-2 (Chiron Corp., Emeryville, CA, U.S.A.) added on days 1 and 4. Peptides were added on day 0 at 10 μg mL−1. On day 7, 50 000 PBMC per well were coincubated with peptide pools corresponding to final concentrations of 4 μg mL−1 of each individual peptide in triplicate in enzyme-linked immunospot (ELISPOT) plates (Millipore, Bedford, MA, U.S.A.) for 24 h. IFN-γ secretion was detected following the manufacturer's instructions (Mabtech AB, Nacka Strand, Sweden) and imaged using an ImmunoSpot Series Analyzer (Cellular Technology Ltd, Cleveland, OH, U.S.A.). Statistical analysis Psoriasis Area and Severity Index (PASI) score was confirmed with a self-reported patient opinion index of disease severity (a scale of 0–10 with 10 at start of treatment). Patients were considered responders if their PASI score improved by at least 75% compared with baseline or their self-opinion score improved to below 4. Three patients who withdrew before at least three samples were collected were not included in the analysis. Kappa statistic was used to analyse associations among the outcome of responder status using the PASI and patient opinion at the last available time point. The statistical significance of the differences between patients and normals in ELISA was analysed using two-sided Student's t-test. Results Clinical response to ultraviolet B treatment In order to determine if psoriasin may be used as a serological biomarker of psoriasis, 14 patients with active psoriasis were enrolled in a clinical trial through the dermatology clinic in Göteborg, Sweden. Individual PASI scores were recorded over time (Fig. 1). Baseline PASI scores ranged from 2·0 to 25·3 (mean 8·5). Patients received standard UVB therapy and serum samples were collected at weeks 0, 2, 4, 6 and 10 weeks follow-up. Patients were deemed eligible for the analysis if assessed at 4 weeks or later after therapy was initiated (n = 11) and the last time point was used for evaluation. Nine of 11 eligible patients (82%) had at least a 75% improvement in PASI score and all patients had a decrease in PASI score with UVB therapy (P < 0·003). This is consistent with published data on the rate of response to UVB therapy.16 Two patients (patients 8 and 14) had a transient increase in PASI score at week 6 after initiation of treatment, but they subsequently improved. PASI score was confirmed with a self-reported patient opinion index of disease severity shown in Table 1. Ten of 11 (91%) patients had a severity index of improvement < 5. Kappa statistic of 0·62 (n = 11) showed significant agreement between the two scoring systems. Fig 1. Decrease in Psoriasis Area and Severity Index (PASI) score with ultraviolet (UV) B treatment of psoriasis. Fourteen patients with moderate to severe psoriasis vulgaris at baseline underwent UVB treatment and evaluation every 2 weeks until study completion. Individual PASI scores at 2-week intervals are shown, and decreased in all patients with treatment. [Normal View ] Table 1 Decrease in clinical disease severity with time (A–E) on ultraviolet (UV) B treatment PASI score Patient opinion Patient A B C D E A B C D E 1 2 3 4 5 6 7 8 9 10 11 12 8·4 8·3 3·9 25·3 3·3 2 15·6 9 4·2 5·4 4·2 8·2 8·4 5·4 1·8 15·7 3·3 2 2·7 1·3 12·9 0·7 0·5 1·6 0·8 6·4 0 1·5 0·9 10 7 6 6 10 4 5 4 5 5 2 3 2 3 0 4 2 7 3·4 4·5 2·5 5·7 5 1·9 2·7 6·3 0·4 1·35 2·2 0 0 8 3 4·25 3 0 3 0·2 0 7 9 8 6·5 6 6 7 5·5 0·4 10 10 10 10 10 10 10 10 10 10 10 10 0 0 0 13 14 3·6 17·2 2·8 14·4 1·6 11·2 0·4 12·8 0 4·8 10 10 8·5 8 6 7 3 8 0 5 Fourteen patients with moderate to severe psoriasis vulgaris at baseline (A) underwent UVB treatment and evaluation every 2 weeks (B, C and D) until study completion (E). Clinical improvement after UVB treatment is shown by Psoriasis Area and Severity Index (PASI) score and self-reported patient opinion of severity (range 1–10, with 10 at baseline). Psoriasin is specifically overexpressed in psoriatic skin lesions Elevated antigen levels of S100A8/S100A9 molecules have been detected in the sera of patients with psoriasis, and they correlated with disease activity.10 As psoriasin/S100A7 expression is also upregulated in psoriatic skin6 and it is a partially secreted protein,14 we hypothesized that psoriasin would also be elevated in the serum. To develop a highly sensitive sandwich ELISA for psoriasin, three distinct monoclonal antibodies were raised in mice by S100A7 injection.14 Immunohistochemical analysis of psoriatic skin lesions using the monoclonal antibody clone 1068 demonstrated high psoriasin expression in psoriatic keratinocytes and in the epithelial cells of normal hair follicles (Fig. 2). Psoriasin expression was often stronger in keratinocytes in the superficial parts of the hyperplastic epithelium and absent in the basal layers. In the keratinocytes, strong staining was often observed in the peripheral parts of the individual cells and faint staining could sometimes be observed in apparent intercellular bridges (desmosomes). Low expression of psoriasin was observed in keratinocytes of normal skin. In some cases, the epithelium was completely negative, but it often showed faint staining in superficial parts, which represent the more differentiated cells (data not shown). Fig 2. S100A7 (psoriasin) is strongly overexpressed in psoriatic skin. Immunostaining using monoclonal antibody 1068 directed against psoriasin at low (a) and high (b) power. There is preferential staining in differentiated spinous layer cells of the central and superficial parts of the hyperplastic and elongated rete ridges. [Normal View ] Serum levels of S100A7 antigen are decreased in the sera of patients with psoriasis We developed a sandwich ELISA for the detection of S100A7 protein in the sera of patients with psoriasis. Detection limits were calibrated using recombinant purified His-tagged S100A7 and S100A7-GST (Fig. 3 and data not shown). Both His-S100A7 and S100A7-GST had comparable limits of detection. GST alone was not detected and the data depicted in Figure 3 are representative of more than five replicate assays. The ELISA had a detection limit of 20 ng mL−1, which is significantly below the 365 ng mL−1 reported mean serum levels of S100A8/S100A9 in normal individuals.10 The range of detection was 20–2000 ng mL−1 and titration curves of recombinant S100A7 protein were included with every assay. ELISA analysis of serum protein concentration of psoriasin was performed in duplicate on all 14 patients with psoriasis at baseline and run concurrently with sera from 14 healthy normal volunteers twice. In contrast to S100A8/S100A9, the mean serum levels of S100A7 antigen were decreased in patients with psoriasis (Fig. 4a, mean 213 ng mL−1) compared with normals (mean 331 ng mL−1) but this was not statistically significant (P = 0·308). S100A7 serum protein levels were comparable with the reported mean serum levels of S100A8/S100A9 in normals (365 ng mL−1).10 Fig 3. Development of an enzyme-linked immunosorbent assay (ELISA) for the detection of psoriasin antigen. Recombinant His-tagged psoriasin protein was purified and titrated in a sandwich ELISA to determine the level of sensitivity of detection of psoriasin protein. The assay consisted of two capture monoclonal antibodies (mAbs) (including mAb 1068 shown in Figure 2) and an independent biotinylated mAb detector antibody. The assay shown here was representative of over five different quantitative assays. Both His-tagged and glutathione-Stransferase (GST)-tagged psoriasin protein showed similar limits of detection to 20 ng mL−1 psoriasin, and GST protein alone was not detected (not shown). [Normal View ] Fig 4. Serum levels of psoriasin/S100A7 are decreased in patients with psoriasis. (a) Serum levels of psoriasin were assayed using the psoriasin enzyme-linked immunosorbent assay in sera from all 14 patients with psoriasis at baseline, and in sera from 14 healthy volunteers. Levels of psoriasin detected in the sera were lower in patients (mean 213 ng mL−1) vs. healthy donors (mean 331 ng mL−1) but this difference was not significant (P = 0·308). (b) Psoriasin levels do not increase with increased disease severity as measured by Psoriasis Area and Severity Index (PASI) score. The patients with psoriasis shown in (a) were separated into two groups, those with lower PASI score (n = 7, mean psoriasin level 239 ng mL−1) and those with higher PASI score (n = 7, mean psoriasin level 188 ng mL−1): differences in psoriasin level between the groups were not significant (P = 0·25). [Normal View ] The patients assayed in Figure 4(a) were divided into two groups by disease severity as measured by PASI score. The mean levels of psoriasin detected in the sera of patients with low PASI score (mean 239 ng mL−1) decreased with increased severity of the disease (188 ng mL−1, P = 0·504; Fig. 4b). This result is in contrast to the correlation of S100A8/S100A9 level with disease severity, where mean levels of 1315 ng mL−1 were observed in patients with high PASI score.10 UVB therapy did not significantly affect S100A7 antigen levels in the 11 patients included in the study (data not shown, P = 0·56). Autoantibodies to S100A7 are not elevated in the serum of patients with psoriasis We hypothesized that the lack of increased psoriasin protein levels in the serum of patients with psoriasis could be due to autoantibodies to S100A7 that may result in the clearance of antigen from the serum and could themselves be potential biomarkers of disease. To detect these autoantibodies we developed a sandwich ELISA using purified recombinant His-tagged S100A7 antigen refolded in urea following denaturation with guanidium chloride. Sera from the 14 patients with active psoriasis and from 14 healthy controls were assayed in duplicate, repeated twice (Fig. 5a). We determined that patients with active psoriasis had slightly increased autoantibodies to S100A7 compared with healthy controls; however, these differences were not statistically significant (mean 0·347 vs. 0·255 units, P = 0·246). Antipsoriasin autoantibody levels also did not correlate with disease severity and serum S100A7 protein levels (data not shown). The half-life of IgG in serum is 23 days;17 thus, IgG autoantibody levels might decrease following 4–6 weeks of therapy. Correlating with this, autoantibodies to p53 antigen, a biomarker of breast cancer, decrease rapidly and specifically after induction chemotherapy.15 In the 11 patients analysed S100A7 autoantibody levels did not significantly change after UVB treatment (P = 0·83). The specificity of the autoantibody identified by ELISA for psoriasin was confirmed by Western blotting sera from a patient with breast cancer (a disease also associated with S100A7 overexpression14) that had elevated S100A7 autoantibodies detected by the ELISA (Fig. 5b). Fig 5. Detection of S100A7-specific antibodies in sera. (a) Detection of S100A7-specific antibodies by enzyme-linked immunosorbent assay (ELISA). Recombinant His-tagged S100A7 protein was adhered to 96-well plates, and detected with sera from healthy donors (left) and patients with psoriasis (right). No detectable difference in antibody levels was observed (mean 0·347 vs. 0·255 units, P = 0·246). O.D., optical density. (b) S100A7 autoantibodies detected by ELISA are specific to S100A7 protein. Recombinant glutathione-S-transferase (GST) and S100A7-GST fusion proteins were purified and separated by sodium dodecyl sulphate–polyacrylamide gel electrophoresis and detected by Coomassie staining (left). The antigens were immunoblotted using 1 : 500 dilution of sera from a patient with breast cancer that had elevated levels of S100A7 autoantibodies by the ELISA shown in (a). The S100A7-GST fusion protein is specifically detected (arrow), while the control GST protein is not (right). [Normal View ] Epitope mapping of anti-S100A7 antibodies S100A7 molecules are small, homodimeric Zn- and Ca-binding molecules. To confirm the specificity of the anti-S100A7 autoantibodies that we identified using ELISA, 15mer overlapping peptides spanning the full-length psoriasin protein were coated on to 96-well plates and assayed with pretreatment patient sera from patients 6, 9 and 12 (Fig. 6). The immunodominant regions of the S100A7 protein included the N-terminal portion and peptides 8, 9 and 17. Using the crystal structure of S100A7,18 we determined that these peptides 8, 9 and 17 are located on the same external region of the molecule (Fig. 7). Fig 6. Mapping of the immunogenic regions of S100A7. Overlapping peptides (15-mers) were adhered to 96-well plates, and the antibody-positive psoriasis sera from Figure 5 were added at 1 : 300 dilution. Peptide-specific autoantibodies to the peptides shown were detected. [Normal View ] Fig 7. Structural mapping of the immunodominant epitopes of S100A7. A ribbon diagram of the homodimeric S100A7 molecules is shown, with the immunogenic peptides 8 (blue), 9 (red) and 17 (green) outlined in colour. [Normal View ] Patients with psoriasis do not have detectable T cells to psoriasin peptides The data from Figures 5(a) and 6 suggested that a subset of patients develops active immunity to psoriasin. Because antibody responses to proteins can be associated with measurable T-cell responses and psoriasis is associated with a T-cell inflammatory process, we examined PBMC from the patients at baseline for the presence of IFN-γsecreting T cells to psoriasin-derived peptides. Using the set of 18 overlapping 15-mer peptides spanning the entire molecule, we created three pools of six peptides, and stimulated PBMC for 1 week with the peptides. PBMC were also stimulated with no peptide control and with phytohaemagglutinin (PHA; positive control). After 1 week of stimulation, the cells were restimulated on ELISPOT plates with the peptides, and IFN-γ-secreting cells were quantitated by standard ELISPOT analysis (Fig. 8). T-cell function was confirmed with PHA stimulation. No psoriasin-specific T cells were detected in the PBMC of the 14 patients at baseline. Fig 8. Interferon (IFN)-γ-secreting T cells to psoriasin peptides are not detected in patients with psoriasis. Peripheral blood mononuclear cells (PBMC; 50 000 cells per well) were stimulated in vitro for 1 week with three separate pools (pools 1, 2 or 3) each containing six overlapping peptides of psoriasin, or no peptide. IFN-γ secretion of activated T cells was measured by enzyme-linked immunospot analysis, and a representative patient sample is shown. No psoriasin-specific T cells were detected in the PBMC of the 14 patients at baseline. The cells could be readily stimulated to secrete IFNγ with the mitogen phytohaemagglutinin (PHA). [Normal View ] Discussion Active psoriatic skin lesions are associated with increased serum levels of S100A8 (calgranulin A), S100A9 (calgranulin B) as well as that of several cytokines including IFN-γ, IL-12, IL-17, IL-18 and IL-22.19 Based on these prior data, we hypothesized that, similar to S100A8/S100A9, elevated levels of S100A7 protein would be detected in the sera of patients with active psoriasis, and that these levels would be correlated with treatment and disease severity. However, we found slightly decreased levels of S100A7 in the sera of patients with active psoriasis, and these levels decreased with increasing disease severity. The absolute levels detected in psoriasis were markedly lower than in a published report on the S100A8/S100A9 levels in serum.10 Whereas this may reflect differences in the relative expression or secretion of the different S100 proteins in active psoriasis, we confirmed by immunohistochemistry that S100A7 is strongly expressed in psoriatic lesions and that it can be detected by the antibody used in our ELISA system. This led us to hypothesize that psoriasin/S100A7 antigen may be cleared by the systemic circulation. This could occur by rapid protein binding or by induction of autoantibodies that enhance clearance of the complex. Psoriasin/S100A7 forms a complex with fatty acid-binding protein in keratinocytes20 and both proteins can be detected by Western blotting in the urine of patients with melanoma.21 Numerous selfantigens have been identified that induce autoantibodies that can be detected in the sera of patients.13 Certain antigens, such as Ro/SS-A, La/SS-B and others22,23 that are associated with autoimmune disease, are used as biomarkers of disease severity. Other cancer autoantibodies are induced by strong overexpression of the antigen, such as mutant p53 antigen,24 her2/neu,25 MUC126 and NY-ESO-1.27 We reasoned that the high expression and partial secretion of psoriasin made it a good candidate autoantigen. We detected psoriasin-specific antibodies in a subset of patients with psoriasis, but this did not correlate with the presence of disease, disease severity, antigen levels, or presence of T-cell immunity. As psoriasin is expressed at low levels in healthy skin, this may stimulate the development of low levels of autoantibodies to psoriasin in a subset of healthy donors. It remains to be determined what impact, if any, autoantibodies to psoriasin have on the clinical outcome of psoriasis. Psoriasinspecific antibodies were also recently detected in patients with ovarian cancer.28 It is not surprising that psoriasin induces a specific immune response detectable in the systemic circulation. Psoriasis was originally considered to be primarily a keratinocyte proliferation disorder based on the clinical signs of epidermal hyperplasia. The persistent inflammatory infiltrate in psoriasis led to the proposal that the epidermal abnormalities were driven by mediators of T-cell activation. This was initially suggested by the therapeutic effect of T-cell inhibitors, such as ciclosporin.29 Mediators of adaptive immunity, including Th1 cells, antigen-presenting cells and IFN-γ, are implicated in the pathogenesis of psoriasis,30 as are mediators of innate immunity, such as tumour necrosis factor-α.31 Despite the intense inflammatory infiltrate, we did not detect psoriasin-specific IFN-γ-secreting T cells in the peripheral blood of patients with active psoriatic lesions, suggesting that the T-cell infiltration is not specific to psoriasin. Acknowledgments We thank Imgenex for providing monoclonal antibodies for these studies. This study was supported by a research grant from the Swedish Psoriasis Foundation and the Welander Foundation. References 1 Gaspari AA. Innate and adaptive immunity and the pathophysiology of psoriasis. J Am Acad Dermatol 2006; 54:S67–80. Links 2 Donato R. Intracellular and extracellular roles of S100 proteins. Microsc Res Tech 2003; 60:540–51. Links 3 Madsen P, Rasmussen HH, Leffers H et al. Molecular cloning, occurrence, and expression of a novel partially secreted protein 'psoriasin' that is highly upregulated in psoriatic skin. J Invest Dermatol 1991; 97:701–12. Links 4 Broome AM, Ryan D, Eckert RL. S100 protein subcellular localization during epidermal differentiation and psoriasis. J Histochem Cytochem 2003; 51:675– 85. Links 5 Wolk K, Witte E, Wallace E et al. IL-22 regulates the expression of genes responsible for antimicrobial defense, cellular differentiation, and mobility in keratinocytes: a potential role in psoriasis. Eur J Immunol 2006; 36:1309–23. Links 6 Martinsson H, Yhr M, Enerbäck C. Expression patterns of S100A7 (psoriasin) and S100A9 (calgranulin-B) in keratinocyte differentiation. Exp Dermatol 2005; 14:161–8. Links 7 Petersson S, Bylander A, Yhr M et al. S100A7 (psoriasin), highly expressed in ductal carcinoma in situ (DCIS), is regulated by IFN-gamma in mammary epithelial cells. BMC Cancer 2007; 7:205. Links 8 Jinquan T, Vorum H, Larsen CG et al. Psoriasin: a novel chemotactic protein. J Invest Dermatol 1996; 107:5–10. Links 9 Krop I, Marz A, Carlsson H et al. A putative role for psoriasin in breast tumor progression. Cancer Res 2005; 65:11326–34. Links 10 Benoit S, Toksoy A, Ahlmann M et al. Elevated serum levels of calciumbinding S100 proteins A8 and A9 reflect disease activity and abnormal differentiation of keratinocytes in psoriasis. Br J Dermatol 2006; 155:62–6. Links 11 Ryckman C, Vandal K, Rouleau P et al. Proinflammatory activities of S100: proteins S100A8, S100A9, and S100A8/A9 induce neutrophil chemotaxis and adhesion. J Immunol 2003; 170:3233–42. Links 12 Nukui T, Ehama R, Sakaguchi M et al. S100A8/A9, a key mediator for positive feedback growth stimulation of normal human keratinocytes. J Cell Biochem 2008; 104:453–64. Links 13 Anderson KS, LaBaer J. The sentinel within: exploiting the immune system for cancer biomarkers. J Proteome Res 2005; 4:1123–33. Links 14 Enerbäck C, Porter DA, Seth P et al. Psoriasin expression in mammary epithelial cells in vitro and in vivo. Cancer Res 2002; 62:43–7. Links 15 Anderson KS, Ramachandran N, Wong J et al. Application of protein microarrays for multiplexed detection of antibodies to tumor antigens in breast cancer. J Proteome Res 2008; 7:1490–9. Links 16 Picot E, Picot-Debeze MC, Meunier L et al. [Narrow-band UVB phototherapy (Philips TL01 lamps) in psoriasis]. Ann Dermatol Venereol 1992; 119:639–42. Links 17 Wochner RD, Drews G, Strober W et al. Accelerated breakdown of immunoglobulin G (IgG) in myotonic dystrophy: a hereditary error of immunoglobulin catabolism. J Clin Invest 1966; 45:321–9. Links 18 Brodersen DE, Nyborg J, Kjeldgaard M. Zinc-binding site of an S100 protein revealed. Two crystal structures of Ca2+-bound human psoriasin (S100A7) in the Zn2+-loaded and Zn2+-free states. Biochemistry 1999; 38:1695–704. Links 19 Arican O, Aral M, Sasmaz S, Ciragil P. Serum levels of TNF-alpha, IFNgamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm 2005; 2005:273–9. Links 20 Ruse M, Broome AM, Eckert RL. S100A7 (psoriasin) interacts with epidermal fatty acid binding protein and localizes in focal adhesion-like structures in cultured keratinocytes. J Invest Dermatol 2003; 121:132–41. Links 21 Brouard MC, Saurat JH, Ghanem G et al. Urinary excretion of epidermal-type fatty acid-binding protein and S100A7 protein in patients with cutaneous melanoma. Melanoma Res 2002; 12:627–31. Links 22 Routsias JG, Tzioufas AG, Moutsopoulos HM. The clinical value of intracellular autoantigens B-cell epitopes in systemic rheumatic diseases. Clin Chim Acta 2004; 340:1–25. Links 23 Lernmark A. Autoimmune diseases: are markers ready for prediction? J Clin Invest 2001; 108:1091–6. Links 24 Soussi T. p53 antibodies in the sera of patients with various types of cancer: a review. Cancer Res 2000; 60:1777–88. Links 25 Disis ML, Pupa SM, Gralow JR et al. High-titer HER-2/neu protein-specific antibody can be detected in patients with early-stage breast cancer. J Clin Oncol 1997; 15:3363–7. Links 26 von Mensdorff-Pouilly S, Petrakou E, Kenemans P et al. Reactivity of natural and induced human antibodies to MUC1 mucin with MUC1 peptides and nacetylgalactosamine (GalNAc) peptides. Int J Cancer 2000; 86:702–12. Links 27 Chen YT, Scanlan MJ, Sahin U et al. A testicular antigen aberrantly expressed in human cancers detected by autologous antibody screening. Proc Natl Acad Sci USA 1997; 94:1914–18. Links 28 Gagnon A, Kim JH, Schorge JO et al. Use of a combination of approaches to identify and validate relevant tumor-associated antigens and their corresponding autoantibodies in ovarian cancer patients. Clin Cancer Res 2008; 14:764–71. Links 29 Griffiths CE, Powles AV, Leonard JN et al. Clearance of psoriasis with low dose cyclosporin. Br Med J (Clin Res Ed) 1986; 293:731–2. Links 30 Sabat R, Sterry W, Philipp S et al. Three decades of psoriasis research: where has it led us? Clin Dermatol 2007; 25:504–9. Links 31 Bos JD, de Rie MA, Teunissen MB et al. Psoriasis: dysregulation of innate immunity. Br J Dermatol 2005; 152:1098–107. Links