Chemical Bonds - Cloudfront.net
advertisement

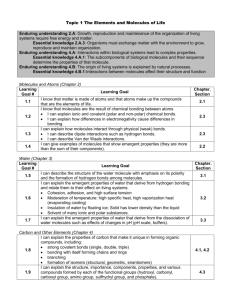
Chemical Bonds 2. Biological, chemical, and physical properties of matter result from the ability of atoms to form bonds from electrostatic forces between electrons and protons and between atoms and molecules. As a basis for understanding this concept: a. Students know atoms combine to form molecules by sharing electrons to form covalent or metallic bonds or by exchanging electrons to form ionic bonds. b. Students know chemical bonds between atoms in molecules such as H2 , CH4 , NH3 , H2 CCH2 , N2 , Cl2 , and many large biological molecules are covalent. c. Students know salt crystals, such as NaCl, are repeating patterns of positive and negative ions held together by electrostatic attraction. d. Students know the atoms and molecules in liquids move in a random pattern relative to one another because the intermolecular forces are too weak to hold the atoms or molecules in a solid form. e. Students know how to draw Lewis dot structures. Sunday 7 Monday 8 Begin Standard 2 Tuesday 9 Wednesday Thursday 10 11 Friday 12 DUE Pg 193-199 #’s 1-22 7.1 & 7.2 quiz 19 Saturday 13 14 15 16 17 18 DUE Pg 203 #’s 23-29 7.3 quiz 21 22 23 DUE Pg 216 #’s 1-6 8.1 quiz 24 25 26 27 28 29 DUE 30 Pg 220-229 #’s 7-22 8.2 quiz 31 1 November 2 3 4 5 6 7 DUE 8 Pg 239-244 #’s 30-38 8.4 quiz 9 10 11 12 13 Standard 2 TEST 20