Plans UNIT 6
advertisement

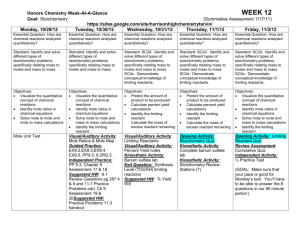
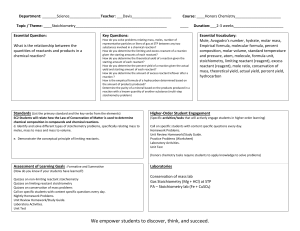
Unit 6 Lesson Plans- Moles & Stoichiometry 2015-2016 Honors Lesson 71 72 73 74 75 Objective Notes- Measuring Matter Elements - How do scientists count the number of atoms, molecules, and formula units? - What is Avogadro’s number? - How can particles be converted to moles and how can moles be converted to particles? - What is molar mass? What are the units for molar mass? - How can moles be converted to mass and how can moles be converted to moles? Notes- Particle to Mass Conversions - How can a particle be converted to mass? Can that conversion be done in one step? - Can a mass be converted to a particle in one step? How many steps does it take? - How can the molar mass be calculated for compounds? - Do the conversion equations change when calculated for a compound versus an element? Mole Activity - How can the mass of a substance be converted first into moles and then into atoms? - What is the molar mass of an element versus the molar mass of a compound? - What units must accompany mole conversions? Quiz- Molar Mass Conversions - How can moles be converted to mass of a compound? - How can mass be converted to particles of a compound? Moles in a Burning Candle Activity - How can you determine the moles of wax used in a burning candle? - How can you calculate the particles of wax used? - Can you determine how long it would take the candle to burn completely? Notes of Formula of a Hydrate - How are hydrates named? - What is a hydrate and how can the number of waters in a hydrate be calculated? - What are the steps to solving hydrate problems? Lab # 14- Formula of a Hydrate Lab - Can you determine the moles of water in a mole of hydrated compound? - How many waters are contained in hydrated Magnesium sulfate? - How can a hydrate be anhydrated? Homework (due dates) - Mole Conversion WS Per 4- in class Per 6- 2/8 Per 8- in class - Particle Mass/Molar Mass WS Per 4- 2/5 Per 6- 2/9 Per 8- 2/8 - Quiz Moles Per 4- 2/9 Per 6- 2/11 Per 8- 2/9 - Finish Mole Activity Per 4- 2/9 Per 6- 2/11 Per 8- 2/9 - Finish Candle Activity Per 4- 2/10 Per 6- 2/12 Per 8- 2/10 - Finish Lab #14 Per 4- 2/11 Per 6- 2/17 Per 8- 2/12 76 77 78 79 80 81 Notes- Percent Composition, Empirical and Molecular Formulas -What is percent composition? What is the formula for percent composition? -What is an empirical formula? -How can an empirical formula be calculated? -What is the difference between an empirical and a molecular formula? -How can a molecular formula be calculated? Empirical Formula Activity - Can an empirical formula of a compound be determined in the lab? - If the empirical formula is calculated in the lab will the mole ratio come out to be a whole number? Why not? - Can the percent of each element in that compound be determined? If the percent is not what it should be could a percent error be determined? What could cause the percent to be off? Moles Review - What is percent composition and how is it calculated to equal exactly 100%? - How can moles be converted to atoms and how can moles be converted to mass? - What is an empirical formula? What is a molecular formula? How can both be calculated from percents? - What is a hydrate and how can hydrates be calculated? - Mini Mole Test - What is a hydrate and how can a hydrate be calculated and named? - How are empirical and molecular formulas related? - How are mole conversions done? - Notes- Stoichiometry - What is Stoichiometry? - How are moles and mass related in stoichiometry? - What is a mole ratio and how are they determined? - What is a mole to mole conversion? How many steps is that type of conversion? - What is a mass to mole conversion and how is it different than a mole to mole? - What is a mass to mass and what are the three steps required to calculate this type of problem? - What is the most important step for all three of these types of problems? - Stoichiometry Practice- Practice WS 2 Go Over Stoichiometry WS - How is a mole to mole calculated and how is it different from a mole to mass? - Why is labeling so important in these types of problems? - How can these problems be done without memorization of equations but rather with logic? - Empirical & Molecular WS Per 4- 2/17 Per 6- 2/19 Per 8- 2/17 Finish Empirical Activity Per 4- 2/18 Per 6Per 8- 2/18 - Study Mini Mole Test Per 4- 2/19 Per 6- 2/22 Per 8- 2/22 - Stoichiometry WS Per 4- 2/23 Per 6- 2/23 Per 8- 2/23 - Stoichiometry WS 3 Per 4- 2/24 Per 6- 2/25 Per 8- 2/24 Quiz Stoich Per 4- 2/24 Per 6- 2/25 Per 8- 2/24 82 83 84 85 Go over Stoich WS 3 A- Stoichiometry Quiz - How can mole ratios be determined for a balanced chemical equation? - Can the three types of stoichiometry problems be distinguished and calculated? - Can a stoichiometry problem be solved without first writing a balanced chemical equation? - What is a hydrocarbon combustion reaction? B- S’more Activity - What is stoichiometry and can it be applied to calculate how many S’mores can be made from a set amount of materials? - What is a limiting reactant? Can students learn what the concept of a limiting reactant is by making S’mores? - What is an excess reactant and was is the excess in this S’mores making activity? Notes- Limiting Reactants - What is a limiting reactant? What is an excess reactant? - What are the steps needed to calculate the limiting reactant? - Once the limiting reactant is found what other calculations can be done? - How can the left over excess reactant be calculated? - How are these calculations related to real life chemistry? Notes- Percent Yield - How is percent yield calculated and how is it different from percent error? - How is percent yield used in limiting reactant equations? - How can the theoretical be calculated from a basic stoichiometry and then be plugged into a percent yield problem? Start Percent Yield WS A- Lab #15- Aluminum Foil Lab - What products result when a single replacement reaction occurs between aluminum foil and copper (II) chloride? - Is there another sign of a chemical reaction besides a temperature change? - How is it possible to calculate the mass that should have resulted from this experiment? - How close was the results achieved to the results that should have been achieved? B- Smarties Percent Composition Activity - Can students find the percent composition of each color candy in a Smarties package? - How do students find the correct masses for the “Smarties” compound? - How can the correct molecular formula be found for the “Smarties” compound? - Can students apply the percent composition concept to all compounds? - Finish Activity Per 4- 2/25 Per 6- 2/26 Per 8- 2/26 - Limiting Reactant WS#1 Per 4- 2/29 Per 6- 2/29 Per 8- 2/29 Finish Percent Yield WS Per 4- 3/1 Per 6- 3/2 Per 8- 3/1 - Finish Lab #15 Per 4Per 6on test Per 8- -Smarties Activity Per 4- 3/2 Per 6- 3/3 Per 8- 3/3 86 87 88 Stoichiometry Review - What are the three types of stoichiometry and how can they can be calculated? - What is a limiting reactant and how can it be calculated? - How can the theoretical yield of a product be calculated once the limiting is determined? - How can the theoretical yield be applied to the experimental and what does that number imply about the lab results? Mini Test Stoichiometry - How can the type of stoichiometry problem be determined? - How can percent yield be calculated without the theoretical yield being given? - How can a limiting reactant problem be determined without the problem specifically saying find the limiting reactant? - How can a mole ratio be determined? Egg Activity -What happens when an egg white is mixed with sodium bicarbonate and citric acid? - What real world application are we testing? - How can calculate the moles of material used? The percent composition of elements in the materials used? - How can you graph your results? - Study Stoichiometry Test Per 4- 3/4 Per 6- 3/4 Per 8- 3/4 - Finish Activity Per 4- 3/8 Per 6- 3/9 Per 8- 3/9