2.1 and 2.2 outline for notes
advertisement

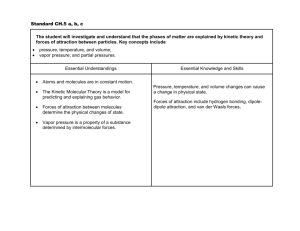
Notes Chapter 2 Chemistry of Life I. Nature of Matter a. Matter is made of Atoms i. All matter consists of _________. An __________ is the smallest unit of matter that cannot be broken down by chemical means. ii. The three particles of an atom are the _________________, _____________________, and ____________________________. b. Elements are the simplest pure substances. i. An ______________ is a substance made of only one kind of _______, and it is therefore a pure substance. ii. There are over _____ known elements. iii. Examples of elements are ________ and ___________. iv. Elements differ in the number of ______________ their elements contain. c. Atoms can bond together. i. A _________________________is a substance made of the joined atoms of _____ or more different _________________________ ii. Covalent bonds 1. Atoms ________________ electrons. 2. A ____________________ is a group of atoms held together by covalent bonds. 3. An example of a molecule is _______________ iii. Hydrogen Bonds 1. A Hydrogen bond is a ________________ chemical attraction between ______________ molecules. 2. Polar molecules have an ___________________ distribution of electrical charge (where one end is positive and the other is negative like with water). 3. Non-polar molecules have an ______________distribution of electrical charge. iv. Ionic Bonds 1. Atoms or molecules ___________ or _____________ electrons. 2. An ___________ is an atom that has gained or lost one or more electrons. 3. Ions of ________________ charge may interact to form an Ionic Bond 4. An example is _____________________________ II. Water and Solutions – Nearly _______ % of your body is made of water. a. Water stores heat efficiently i. Water heats more ______________ and retains heat _________________ than many other substances. ii. Many organisms release excess heat through water ______________________. iii. How do you release excess heat???? iv. Water helps cells maintain ________________________ because its ability to control temperature enables cells to maintain constant internal temperature when the external temperature changes drastically. b. Water Bonds to itself and other substances. i. ________________ is the attraction between substances of the same kind. ii. Hydrogen bonds between water molecules cause the ________________ of liquid water. iii. The attraction between water molecules causes a condition known as _____________ _____________________ iv. ______________ is an attraction between different substances. v. Because of adhesion some substances get __________. vi. The process in which water molecules move upward through a narrow tube (such as the stem of a plant is called _________________ action. 1. Explain how capillary action works in a plant – summarize it. c. Water dissolves many substances – many substances dissolve in water A _______________ is a mixture in which one or more substances are evenly distributed in another substance. Because some substances dissolve easily in water, they can more easily move _____________ and ________________ cells. i. Polarity 1. The Polarity of water enables many substances to ____________________ in water. 2. _______________ compounds and ________________ molecules dissolve best in water. a. The ions or molecules become _________________ distributed in the water. 3. _______________ molecules do not dissolve well in water. 4. When nonpolar substances, such as oil, are placed in water, the water molecules are ______________ attracted to themselves than to the nonpolar molecules. ii. Acids and bases 1. The bonds of water are strong but may break forming a hydrogen ion _______ and a hydroxide ion _________. (write the equation below) 2. Pure water always has a low concentration of ______________ ______ and _____________________ _________. 3. ________ are coumpouds that form hydrogen ions when dissolved in water a. When acids are added to water, they concentration of _______________ ions is increased above that of pure water. 4. _______ are compounds that reduce the concentration of hydrogen ions when dissolved in water. This happens because the hydroxide ions react with the hydrogen ions to form _______________ molecules. 5. The ______ scale measures the concentration of _________________ ions in solutions. 6. The scale ranges from 0 - ___. Pure water has a pH value of ____. 7. Acidic solutions pH value is ___________ 7. Basic solutions have pH values _________ 7 A solution with a pH of 5 has ______ times as many hydrogen ions as a solution with a pH of 6.