Unit 2 Practice Test
Multiple Choice
Identify the choice that best completes the statement or answers the question.
____
____
____
____
____
____
____
____
____
____
____
____
____
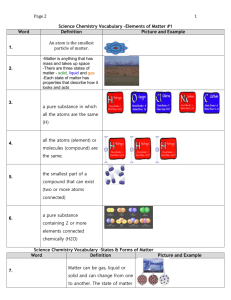
1. Matter that has a definite volume and a definite shape is a ____.
a. gas
c. plasma
b. liquid
d. solid
2. Matter in which the particles are free to move in all directions until they have spread evenly throughout their
container is a ____.
a. gas
c. plasma
b. liquid
d. solid
3. Three examples of physical change are ____.
a. boiling of water, bursting of a balloon, and crumpling a piece of paper
b. burning of gasoline, rotting of an egg, and exploding fireworks
c. freezing of water, evaporation of gasoline, and rusting a nail
d. sawing of wood, crushing a can, and toasting a marshmallow
4. Fog is an example of a ____.
a. colloid
c. solution
b. compound
d. substance
5. When two or more substances are combined so each substance can be separated by physical means, the result
is a(n)____.
a. chemical change
c. element
b. compound
d. mixture
6. The scattering of light by colloids is called ____.
a. air pollution
c. suspension
b. conservation
d. The Tyndall effect
7. The oceans are an example of a(n) ____ solution.
a. alloy
c. liquid
b. gaseous
d. solid
8. Increasing the surface area of a solid ____.
a. causes the solid to ionize
c. slows the rate of solution
b. has no effect on the rate of solution
d. speeds the rate of solution
9. The concentration of solution that contains a large amount of solute in the solvent could be described as ____.
a. concentrated
c. polar
b. diluted
d. unsaturated
10. The amount of solute that can be dissolved in a specific amount of solvent at a given temperature is its ____.
a. concentration
c. dilution
b. density
d. solubility
11. Which of the following will speed up the dissolving of a solid solute in water?
a. Cool the solution.
c. Grind up the solvent.
b. Freeze the solute.
d. Stir the solution.
12. A(n) ____ is a substance that produces OH ions in a solution.
a. acid
c. salt
b. base
d. alcohol
13. ____ change color in the presence of an acid or a base.
a. Acids
c. Buffers
b. Glycerins
d. Indicators
____ 14. A bitter taste and a slippery feel are clues that a solution is probably a(n) ____.
a. acid
c. hydrate
b. base
d. salt
____ 15. A base that only partly ionizes in a solution is a ____ base.
a. concentrated
c. strong
b. dilute
d. weak
____ 16. Coffee has a pH of about 5. Coffee is ____.
a. extremely acidic
c. somewhat acidic
b. extremely basic
d. somewhat basic
Matching
Match each term with the correct definitions below.
a. element
d. compound
b. mixture
e. matter
c. substance
____
____
____
____
____
17.
18.
19.
20.
21.
composition variable
has mass, takes up space
two or more elements combined
all atoms the same
composition definite
Completion
Complete each statement.
22. ____________________ is the total energy of the molecules in a material.
23. Specific heat is the amount of ____________________ required to raise the temperature of 1 kg of a material
1 K.
24. In comparing plasma to a solid, a liquid, and a gas, plasma is most similar to ____________________.
25. The energy needed to change a liquid to a gas is the _________________________.
26. The energy needed to change a solid to a liquid is the _________________________.
27. Matter is classified as ____________________ and mixtures.
28. A crystal of solute was dropped into a solution, and it dissolved. The original solution was
____________________.
29. If a crystal of solute is dropped into a solution and other crystals appear, the solution was
_________________________.
30. Stirring ____________________ the speed of dissolving of a solid in a liquid.
31. Decreasing temperature ____________________ the speed of dissolving for a gas in a liquid.
Short Answer
32. Which of the states of matter can you force into a smaller volume? Explain.
33. Why is saltwater a mixture, not a substance?
34. Iron and oxygen combine to form rust. Compare the mass of iron and oxygen before the reaction with the
mass of the rust after the reaction.
35. Fresh pickle juice has a light green color. Is it a colloid, a solution, or a suspension?
36. Explain why air is considered a solution.
SOLUBILITY OF SUBSTANCES IN WATER AT 20°C
Solid Substances
Solubility in g/100 g of Water
Barium sulfate
Lithium carbonate
Potassium chloride
Sodium nitrate
Lithium bromide
0.00025
1.3
34.0
87.6
166.0
Table 22-1
37. According to Table 22-1, how would you classify a solution that contains 1.5 g of lithium carbonate in 100 g
of water at 20°C.
38. According to Table 22-1, how much potassium chloride dissolves in 250 g of water to form a saturated
solution at 20°C?
39. What factor determines the strength of an acid or a base?
40. List two properties that acids and bases have in common.
Modified True/False
Indicate whether the statement is true or false. If false, change the identified word or phrase to make the statement true.
____ 41. The melting point is the same temperature as the freezing point for a substance.
_________________________
____ 42. Substances are either elements or mixtures. _________________________
____ 43. Smoke is a compound. _________________________
____ 44. The law of conservation of mass states that the mass of all substances before a physical change equals the
mass of all the substances after the change. _________________________
____ 45. The freezing of water is a chemical change. _________________________
____ 46. When lightning converts oxygen gas, O2, into ozone, O3, it is a physical change.
_________________________
____ 47. A fruit salad is a homogeneous mixture. _________________________
____ 48. Flammability is a physical property. _________________________
____ 49. A suspension is a homogeneous mixture. _________________________
____ 50. The breaking of a glass window is a physical change. _________________________
Unit 2 Practice Test
Answer Section
MULTIPLE CHOICE
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
D
A
A
A
D
D
C
D
A
D
D
B
D
B
D
C
MATCHING
17.
18.
19.
20.
21.
B
E
D
A
C
COMPLETION
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
Thermal energy
energy
a gas
heat of vaporization
heat of fusion
substances
unsaturated
supersaturated
increases
increases
SHORT ANSWER
32.
33.
34.
35.
36.
37.
38.
39.
40.
Gas and plasma; there is still room between the particles.
The salt and the water can be separated by physical means. Distillation separates dissolved solids from liquids.
masses are equal; law of conservation of mass
It is a solution because it does not scatter light.
Air contains several dissolved gases.
supersaturated
85 g
how completely a compound separates into ions when dissolved in water
form solutions, are corrosive, can be detected with a pH indicator
MODIFIED TRUE/FALSE
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
T
F, compounds
F, colloid
F, chemical
F, physical
F, chemical
F, heterogeneous
F, chemical
F, heterogeneous
T
 0
0





![Chem 3 Elements, Compounds, and Mixtures PPT [11/15/2013]](http://s2.studylib.net/store/data/005796663_1-9247eb9205249afa1115390decddad23-300x300.png)