Light - Cloudfront.net
advertisement

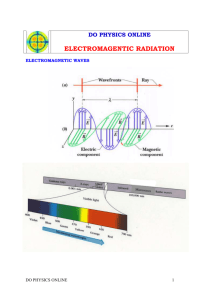
LIGHT (AND THE ELECTROMAGNETIC SPECTRUM) ELECTROMAGNETIC WAVES Electromagnetic Radiation is just a name for the range of radiation….(feel free to start singing) Light is both a particle and a wave Light particles are called photons (mass-less bundles of concentrated electromagnetic energy) We only see light in the range of 400-700 nm speed of light = c = 3.0*108 m/s c = λ*f WHY ARE SOME THINGS SEE-THROUGH? Light is energy carried in an electromagnetic wave that is generated by vibrating electric charges. Visualize the electrons in an atom as connected by imaginary springs. When light hits the electrons, they vibrate. The natural vibration frequencies of an electron depend on how strongly it is attached to a nearby nucleus. TRANSPARENCY If frequency of the light ≠ natural frequency of the material electrons forced into vibration with small amplitudes The atom holds the energy for less time less chance of collision with neighboring atoms less energy transferred as heat Energy of the vibrating electrons reemitted as transmitted light Materials that transmit light are transparent. OPACITY If frequency of the light = natural frequency of the material electrons forced into vibration with large amplitudes The atom holds the energy for more time more chance of collision with neighboring atoms more energy transferred as heat Materials that absorb light without reemission and thus allow no light through them are opaque. GLASS: AN EXAMPLE Glass is _______________ to visible light. BUT…Electrons in glass have a natural vibration frequency in the ultraviolet range UV light shines on glass resonance occurs e- forced into vibration with higher amplitudes atoms hold energy for more time collide with neighboring atoms energy LOST to heat UV light does not pass through glass Glass is ____________ to UV light. GLASS: EXAMPLE CONTINUED When the EM wave has a lower frequency than UV, as visible light does, e- are forced into vibration with smaller amplitudes atom holds the energy for less time less chance of collision with neighboring atoms less energy is transferred as heat Energy of the vibrating electrons is reemitted as transmitted light. SPEED OF LIGHT IN MATERIALS In a vacuum, the speed of light is a constant 3 x 108 m/s Atmosphere: very close to c Water: 0.75c Glass: 0.67c Diamond: 0.40c When light emerges from these materials into the air, it travels at its original speed, c SHADOWS A thin beam of light is called a ray When light shines on an object, some of the rays may be stopped while others pass on in a straight line path A shadow is formed where light rays cannot reach Fuzzy part around the edges of the shadow happens when: Light from one source is blocked but where other light fills in or Where light from a source is only partially blocked POLARIZATION When light from a lamp or the sun shines on a polarizing filter, the light that is transmitted is polarized Light will pass through a pair of polarizing filters when their polarization axes are aligned, but not when they are crossed at right angles. 3D Hold an upright finger at arm’s length and see how it switches position relative to the background as you alternately close each eye. Vision in three dimensions depends on the fact that both eyes give impressions simultaneously, each eye viewing a scene from a slightly different angle. The view seen by each eye is different. The combination of views in the eye-brain system gives depth. A pair of photographs or movie frames, taken a short distance apart (about average eye spacing), can be seen in 3-D when the left eye sees only the left view and the right eye sees only the right view. 3D MOVIES! Movies project the pair of views through polarization filters onto a screen. Their polarization axes are at right angles to each other, so the right eye sees only the right view and the left eye sees only the left view. Overlapping pictures look blurry to the naked eye To see in 3-D, the viewer wears polarizing eyeglasses with the lens axes also at right angles Each eye sees a separate picture, just as in real life. The brain interprets the two pictures as a single picture with a feeling of depth LIGHT PART II: COLOR (AND THE ELECTROMAGNETIC SPECTRUM) AND ON TO….COLOR: THE SPECTRUM By passing a narrow beam of sunlight through a triangular-shaped glass prism, Newton showed that sunlight is composed of a mixture of all the colors of the rainbow spectrum = spread of colors ROYGBV white light = combo of all the colors black = absence of light WHY DO WE SEE DIFFERENT COLORS? The color of an opaque object is the color of the light it reflects The color of a transparent object is the color of the light it transmits Most materials absorb light of some frequencies and reflect the rest. Material absorbs light of most visible frequencies & reflects red the material appears red Reflects light of all the visible frequencies it will be the same color as the light that shines on it Absorbs all the light that shines on it it reflects none and is black SUNLIGHT White light from the sun is a composite of all the visible frequencies. The brightness of solar frequencies is uneven Lowest frequencies of sunlight in the red region Not as bright as those in the middle-range yellow and green region Yellow-green light is the brightest part of sunlight. COLORED LIGHT Light of all the visible frequencies mixed together produces white White also results from the combination of only red, green, and blue light R, G, B called additive primary colors Red + Green light = Yellow Red + Blue light = Magenta Green + Blue light = Cyan COLORED PIGMENTS Red + green + blue paint muddy dark brown Not white! The mixing of paints and dyes is an entirely different process from the mixing of colored light. When paints or dyes are mixed, the mixture absorbs all the frequencies each paint or dye in it absorbs Example: Blue paint reflects mostly blue light, but also violet and green Yellow paint reflects mostly yellow light, but also red, orange, and green It absorbs red, orange, and yellow light. It absorbs blue and violet light. When blue and yellow paints are mixed, between them they absorb all the colors except green FUN THINGS COLOR HELPS US UNDERSTAND How do color television sets work? How is color printing done? Why is the sky blue? Why are sunsets red? Why is water greenish blue? ATOMIC SPECTRA Electrons surrounding the atomic nucleus have welldefined orbits Atom absorbs external energy one or more of its electrons is boosted to a higher energy level AKA – well defined energy levels—lower energy near the atomic nucleus and higher energy farther from the nucleus ‘excited state’ Electron quickly drawn back to its original or a lower level atom emits a pulse of light called a photon When made to emit light, every element has its own characteristic color (the “fingerprints” of the elements) The light is analyzed using a spectroscope The spectrum of an element appears not as a continuous band of color but as a series of lines called a line spectrum Each line corresponds to a frequency of light Spectral lines seen in the spectroscope are images of the slit through which the light passes Much of the information that physicists have about atomic structure is from the study of atomic spectra