CHAPTER 12
advertisement
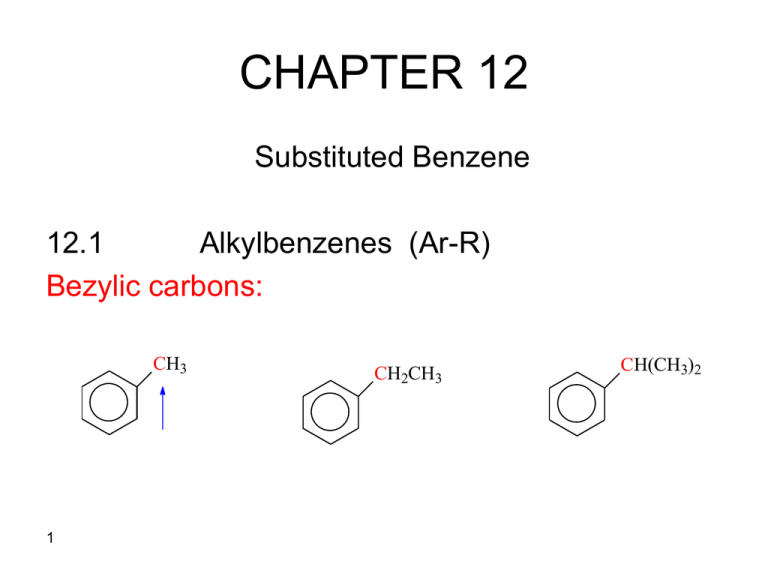
CHAPTER 12 Substituted Benzene 12.1 Alkylbenzenes (Ar-R) Bezylic carbons: CH3 1 CH2CH3 CH(CH3)2 Reactions of Alkylbenzens 1. Free Radical Halogenation CH2CH3 Br2 hv 2 Br CHCH3 2- Oxidation CH3 KMnO4 hot CH2CH3 MnO4hot R CO2H KMnO4 hot CO2H CO2H CH2CH3 CHCH3 3 MnO4hot CO2H CH3 Notice: CCH3 CH3 MnO4hot No Reaction Benzylic carbon has no hydrogen CH3 CH3 MnO4hot MnO4hot CO2H HO2C CO2H CO2H 4 Phenols (ArOH) A phenol has a hydroxyl group directly attached to the benzene ring SP2- hybridized carbon OH C---O bond is not easily broken R-OH + HBr 5 No substitution or elimination reaction RBr + H2O Ar-OH + HBr No reaction SN1 or SN2 Acidity of Phenols CH3CH2O- Na+ + NaOH CH3CH2OH pKa= 16 H2O Not favored pKa=15.7 OH + NaOH O- Na+ + H2O favored pKa= 15.7 pKa= 10 O- + R X 6 + a phenoxide ion SN2 OR + X- Esterfication of phenols O OH + CH3C O O OCCH3 OCCH3 + CH3CO2H 7 The Kolbe Reaction OH 1) OH2) CO2 OH 3) H+ CO2H Mechanism OH OH- O- + O C O O H C O O- OH H+ OH 8 Salicylic acid CO2H CO2- Importance of Kolbe Reaction To Synthesize aspirin OH + CO2H O O CH3COCCH3 O OCCH3 CO2H Acetylsalicylic acid (Aspirin) 9 The Reimer-Tiemann Reaction The reaction of phenol with chloroform in the presence of base. OH 10 OH- , 1) CHCl3, 2) H2O, H+ 70 C OH CH O Salicylaldehyde Mechanism: OH- H CCl3 + - H2O H O- CCl3 CCl2 CCl2 dichlorocarbene ..CCl 2 + Cl- CHCl2 O OOHO O H 11 H OH H H+ O- O CHCl O- Oxidation of phenols: phenol resists oxidation Oxidation of hydroquinones (1,4-dimethoxy benzenes) O OH O + 2 H+ + 2e - H O OH hydroquinone 1.4-benzoquinone OH O (quinone) O + OH H O 1.2-benzoquinone 12 2 H+ + 2e Preparation of Aniline HNO3 H2SO4 NO2 Fe, HCl NO2 N+H3Cl- H2, Pd/C or Pt 13 OH- NH2 Benzenediazonium Salts NH2 NaNO2, HCl, 0O C + N ClN + H2O benzenediazonium chloride Excellent leaving abilty of N2 gas very reactive + N N 14 _ N 2 + Nu - Nu Reactions of benzenediazinium Salts Br Cl CuCl CuBr 1) HNO3, H2SO4 2) H2, Pt NH2 NaNO 2, HCl, O CuCN, KCN + N N Cl- KI 0C H3PO2 H2O, 1) H+ 2) H OH 15 CN I HBF4 heat F Coupling reactions + N + N N N + N N + OH CO2H N N OH Azo compound NH2 O NaNO2, HCl, 0 C CO2H 16 + N N + NHCH3 N N NHCH3 Synthesis involving diazonium salts ? CH3 ? ? ? CH3 HNO3, H2SO4 CH3 CuCN, KCN NO2 1) C N Fe, HCl 2) OH N2+ Cl- CH3 NaNO2 , O CH3 17 NH2 HCl, O C Nucleophilic Aromatic Substitution X But + X Nu + - No SN1 or SN2 reaction Nu - Nucleophilic Aromatic Substitution 18 Nu + X- Examples 10% Cl NaOH OH o 350 300 atm increasing O2N reactivity toward Nu - Cl 10% NaOH O2N o 160 P-nitrophenol NO2 NO2 O2N Cl NO2 19 OH H2O warm O2N OH NO2 Other nucleophiles NO2 NO2 O2N Cl NO2 O2N 20 Cl NH3 heat O2N NH2 NO2 CH3CH2Sheat O2N SCH2CH3 Carbanion Mechanism: If the ring is activated toward nucleophilc substitution by an electron withdrawing group (I,e NO2) Two steps 1) addition of Nu: to form carbanion 2) Loss of the halide ion OHslow O2N Cl OH O2N - Cl Carbanion intermediate 21 fast O2N OH Carbanion intermediate is stabilized by resonance OO- O2 N OH Cl O+ N O- Cl OH Cl O+ N O- 22 OH N+ OH Cl Benzyne Mechanism: If there is no electron withdrawing substituent on the ring. Cl NH2 , NH3 NH2 Mechanism Cl H - HCl NH2- NH2- 23 NH3 Benzyne NH2- NH2 + NH2 Benzyne H H H 24 H Syntheses of substituted benzene compounds Cl • How would you synthesize NO2 HNO3 Cl2 H2SO4 • NO2 FeCl3 NO2 But IF YOU START WITH CHLORINATION Cl Cl2 FeCl3 Cl HNO3 H2SO4 o,p-director 25 Cl Cl NO2 + NO2 Syntheses ? ? Br ? NH2 HNO3 H2SO4 1) Fe, HCl NO2 ) OH 2 Br2, FeBr3 Br NO2 26 Note: Nitro group can be converted to arylamines which in turn can also be converted to aryldiazonium salts,=> making a diversity of substituted products 1) NO2 Fe, HCl NaNO2, HCl 2) OH- m-director NH2 0o o,p-director Nu N2+ Nu easily displaced Notice: Amino group does not undergo Friedel-Crafts reaction because an amino group reacts with Lewis acid since it is basic to form strongly deactivating grouo NH2 27 + AlCl3 Al-Cl3 N+H2 strongly deactivating Nitrobenzene does not undergo Friedel-Crafts reactions NO2 O RX or RCX AlX3 No aromatic substitution Notice: An amino group is o,p-director, but an ammonium group ( -NR3+) is an m-director and deactivating. NH2 o,p-director 28 + HNO3 N+H3 NO3m-director Sulfonic acid group (-SO3H) The sulfonic acid group is easily removed and can be displaced by a variety of reagents. SO3H SO3H 29 H2O, H+ 1) Fused NaOH + ) H O, H 2 2 + H2SO4 OH ? ? ? Br OH Br2, FeBr3 H2O, H+ Br HNO3 H2SO4 Br NO2 30 1) Fe, HCl 2) OH- Br NH2 NaNO2,HCl 0o Br N2+ Br ? ? ? Br HNO3 H2SO4 CuBr NO2 Br2, FeBr3 Br NO2 31 Br Br 1) 2) Fe, HCl OH- NH2 NaNO2,HCl 0 N2+ ? ? ? ? CO2H NH2 CH3CH2Cl, AlCl3 KMnO4 heat CH2CH3 HNO3, H2SO4 CH2CH3 NO2 1) Fe, HCl 2) OH CH2CH3 NH2 32 ? ? ? ? OCH3 CH3I HNO3, H2SO4 O- NO2 NaOH H2,Pd/C NH2 33 OH 1) NaNO2,HCl 2) H2O, H+ Cl ? ? ? ? CH3 CO2H KMno4, heat Cl2, FeCl3 CO2H 34 Cl ? ? ? ? CH3 CH3 HNO3, H2SO4 H3PO2 O2N CH3 H2,Pd/C H2N 1) CH3 2) 35 Cl2, FeCl3 NaNO2, HCl Cl- N2 + Cl CH3