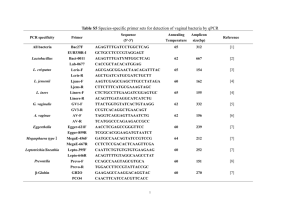
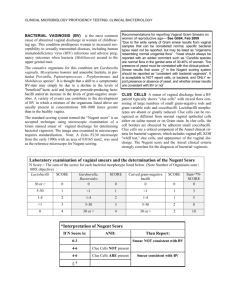
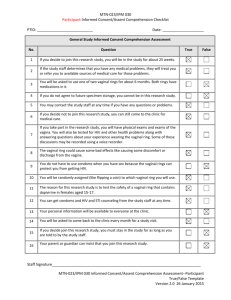
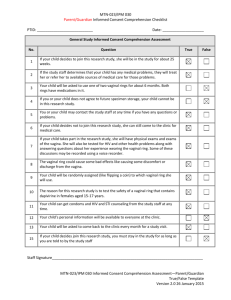
chapter three
advertisement

Effects of Bacterial Vaginosis Associated Bacteria, Endogenous Lactobacillus and Sociodemographic Factors on Vaginal Colonization with the Probiotic Lactobacillus crispatus CTV-05: A Study of the Intervention Cohort in a Phase IІA Clinical Trial of LACTIN-V® in San Francisco, USA Benjamin Mbiyu Ngugi A Thesis Submitted in Fulfilment for the Award of the Degree of Doctor of Philosophy in Epidemiology at the Jomo Kenyatta University of Agriculture and Technology 2012 i DECLARATION This thesis is my original work and has not been presented for a degree in any other University. Signature…………………………….......................... Date............................. Benjamin Mbiyu Ngugi This thesis has been submitted for examination with our approval as University Supervisors: 1. Signature…………………………….......................... Date............................. Dr. Craig Cohen University of California, San Francisco, USA 2. Signature…………………………….......................... Date............................. Dr. Elizabeth Bukusi KEMRI, Kenya 3. Signature…………………………….......................... Date............................. Dr. Gideon Kikuvi JKUAT, Kenya 4. Signature…………………………….......................... Dr. Joseph Gikunju JKUAT, Kenya ii Date............................. DEDICATION I dedicate this thesis to my family whose patience, endurance, understanding and encouragement made the completion of the project a reality. iii ACKNOWLEDGEMENTS I would like to sincerely thank my supervisors, Dr. Craig Cohen, Dr. Elizabeth Bukusi, Dr. Joseph Gikunju and Dr. Gideon Kikuvi, for their tireless efforts in advising, guiding and supervising this project. I would also like to thank the following institutions and programs: Osel Inc. Of Santa Clara, California, USA, for funding this study (through grant UL1 RR024131-01 from the National Centre for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research); the Infectious Disease Research Training Program (IDRTP) for the scholarship which enabled me complete my studies at JKUAT and also enabled me to travel to San Francisco, USA, where this study was undertaken; University of California, San Francisco, USA, for the permission to use their facilities for this study; the Program in Infectious Diseases, Fred Hutchinson Cancer Research Centre laboratories in Seattle, Washington, USA for helping me to carry out the bacteria identification and quantification through qPCR assays. Most importantly, I want to thank all the women who responded to our call and agreed to be part of the LACTIN-V® study. Last but not least, thanks be to God, for without Him nothing is possible. iv TABLE OF CONTENTS DECLARATION ................................................................................................................ ii DEDICATION ................................................................................................................... iii ACKNOWLEDGEMENTS .............................................................................................. iv TABLE OF CONTENTS ................................................................................................... v LIST OF TABLES .............................................................................................................. x LIST OF FIGURES .......................................................................................................... xii LIST OF PLATES ........................................................................................................... xiii LIST OF APPENDICES ................................................................................................. xiv LIST OF ABBREVIATIONS AND ACRONYMS ........................................................ xv ABSTRACT ................................................................................................................... xviii CHAPTER ONE ................................................................................................................. 1 1.0 INTRODUCTION .................................................................................................. 1 1.1 Background........................................................................................................... 1 1.2 Research Question ................................................................................................ 4 1.3 Statement of the problem...................................................................................... 4 1.4 Study Justification ................................................................................................ 6 1.5 Hypothesis ............................................................................................................ 6 v 1.6 Objectives ............................................................................................................. 7 1.6.1 General objective .............................................................................................. 7 1.6.2 Specific objectives ............................................................................................ 7 CHAPTER TWO ................................................................................................................ 8 2.0 LITERATURE REVIEW ...................................................................................... 8 2.1 Bacterial Vaginosis ............................................................................................... 8 2.1.1 Definition of bacterial vaginosis ...................................................................... 8 2.1.2 Epidemiology of bacterial vaginosis ................................................................ 9 2.1.3 Aetiology of bacterial vaginosis ..................................................................... 11 2.1.4 Clinical presentation and diagnosis of bacterial vaginosis ............................. 13 2.1.5 Adverse outcomes and sequelae of bacterial vaginosis .................................. 15 2.2 Treatment of Bacterial Vaginosis ....................................................................... 17 2.2.1 Anti-microbial treatment ................................................................................ 17 2.2.2 Recurrence of BV after anti-microbial treatment ........................................... 18 2.2.3 Vaginal lactobacilli ......................................................................................... 19 2.2.4 Interaction between vaginal lactobacilli and BV-associated bacteria ............ 20 2.2.5 Use of Lactobacillus probiotics in BV treatment ........................................... 24 Probiotic Lactobacillus Crispatus CTV-05 (LACTIN-V®) ............................... 28 2.3 2.3.1 Suitability of Lactobacillus crispatus CTV-05 as a vaginal probiotic ........... 28 2.3.2 Randomised Controlled Trials with LACTIN-V® ......................................... 29 vi 2.3.3 Factors that may affect vaginal colonization with probiotic Lactobacillus ............crispatus CTV-05 ........................................................................................... 30 CHAPTER THREE.......................................................................................................... 35 3.0 MATERIALS AND METHODS ......................................................................... 35 3.1 Study Materials ................................................................................................... 35 3.1.1 MetroGel®....................................................................................................... 35 3.1.2 LACTIN-V® applicator .................................................................................. 35 3.1.3 LACTIN-V® ................................................................................................... 36 3.2 Study Methods .................................................................................................... 36 3.2.1 Study design ................................................................................................... 36 3.2.2 Study site ........................................................................................................ 37 3.2.3 Study population and participant recruitment ................................................ 37 3.2.4 Study procedures ............................................................................................ 41 3.2.5 Study measurements ....................................................................................... 44 3.3 Data management and analysis .......................................................................... 52 3.4 Ethical Considerations ........................................................................................ 53 CHAPTER FOUR ............................................................................................................ 55 4.0 4.1 RESULTS .............................................................................................................. 55 Participants’ socio-demographic and sexual behaviour characteristics ............. 55 vii 4.2 L. crispatus CTV-05 vaginal colonization ......................................................... 58 4.3 BV-associated Bacteria qPCR Amplification Curves ........................................ 62 4.4 Baseline concentrations of BV-Associated Bacteria .......................................... 65 4.5 Effects of BV-associated bacteria on L. crispatus CTV-05 colonization .......... 67 4.6 Factors associated with L. crispatus CTV-05 vaginal non-colonization ............ 73 4.7 Correlation between post-treatment vaginal levels of L. crispatus species and ............ pretreatment levels of BV-associated bacteria .................................................. 75 4.8 The effects of the concentration of BV-associated bacteria on BV persistence ............ (Nugent score ≥7) .............................................................................................. 79 CHAPTER FIVE .............................................................................................................. 83 5.0 DISCUSSION, CONCLUSION AND RECOMMENDATIONS ..................... 83 5.1 Discussion........................................................................................................... 83 5.1.1 Proportion of Women Successfully Colonized with L. Crispatus CTV-05 .............. 83 5.1.2 Effects of BV-Associated Bacteria on Vaginal Colonization with L. Crispatus ............CTV-05 ....................................................................................................................... 85 5.1.3 Effects of other Vaginal Lactobacilli on Colonization with L. Crispatus CTV-05 ... 88 5.1.4 Effects of Socio-demographic and Sexual Factors on Colonization with L. Crispatus ............CTV-05 ....................................................................................................................... 91 5.1.5 5.2 Limitations ................................................................................................................. 93 Conclusion .......................................................................................................... 94 viii 5.3 Recommendations .............................................................................................. 96 REFERENCES ................................................................................................................. 98 APPENDICES................................................................................................................. 120 ix LIST OF TABLES Table 2.1: Classification of commonly isolated and some recently described BV..................associated bacteria species ............................................................................... 11 Table 2.2: Bacterial vaginosis recurrence in clinical trials which used antibiotics and ..................probiotics for the treatment of BV.................................................................... 18 Table 2.3: Classification of selected lactobacilli for probiotic use based on metabolic ..................production of lactic acid from sugars ............................................................... 25 Table 2.4: Summary of Randomized Clinical Trials (RCT) using probiotics in women ..................with bacterial vaginosis .................................................................................... 26 Table 3.1: Primer and probe sequences used in qPCR assaysα ......................................... 49 Table 4.1: Socio-demographic characteristics of the study participants. .......................... 56 Table 4.2: Baseline sexual risk factors among the study participants ............................... 57 Table 4.3: Vaginal colonization with any Lactobacillus species at enrolment and ..................subsequent Lactobacillus crispatus CTV-05 colonization at Day 28 .............. 60 Table 4.4: Vaginal colonization with any Lactobacillus species and Lactobacillus ..................crispatus CTV-05 at Day 28............................................................................. 60 Table 4.5: Pretreatment vaginal concentration of various bacteria† .................................. 65 Table 4.6: Pretreatment vaginal concentration of various bacteria just before LACTIN-V® ..................treatment (enrolment visit) † ............................................................................. 66 Table 4.7: Concentrations of bacteria in vaginal fluid just before LACTIN-V® treatment .................. (enrolment visit) grouped by Lactobacillus species colonization status in the ..................same visit .......................................................................................................... 67 x Table 4.8: Vaginal concentration of various bacteria at screening and day 28 in women ..................grouped according to their post-treatment Lactobacillus crispatus CTV-05 ..................colonization status. ........................................................................................... 70 Table 4.9: Vaginal concentration of various bacteria at enrollment and day 28 in women ..................grouped according to their post-treatment Lactobacillus crispatus CTV-05 ..................colonization status. ........................................................................................... 72 Table 4.10: Un-adjusted risks of colonization with Lactobacillus crispatus CTV-05 at the ..................Day 28 visit (using exact logistic regression)................................................... 74 Table 4.11: Pearson correlation coefficients of bacteria vaginal concentrations at the ..................screening (pretreatment) visit correlated with bacterial concentrations at the ..................day 28 visit (post-treatment) ............................................................................. 76 Table 4.12: Pearson correlation coefficients of bacteria vaginal concentrations at the ..................enrolment visit correlated with bacterial concentrations at the day 28 visit .................. (post-treatment) ............................................................................................... 78 Table 4.13: Concentration of bacteria in vaginal fluid at screening and 28 days after ..................LACTIN-V treatment in women with Nugent score <7 and those with Nugent ..................score ≥7 ............................................................................................................ 82 xi LIST OF FIGURES Figure 2.1: Competing models for the pathogenesis of bacterial vaginosis. ..................... 13 Figure 3.1: Chart of the clinical visits and study product administration ......................... 42 Figure 4.1: Proportion of participants colonized by Lactobacillus crispatus CTV-05 study ...................strain amongst those colonized by any Lactobacillus species on follow-up ................... (n=18) ............................................................................................................. 61 Figure 4.2: Gardnerella vaginalis standard curves (a) and target amplification curves at ...................screening, enrolment and day 28 for subject 1606 (b). ................................... 63 Figure 4.3: Lactobacillus crispatus standard curves (c) and target amplification curves at ...................screening, enrolment and day 28 for subject 0106 (d). ................................... 64 Figure 4.4: Variation in the vaginal concentration of various bacterial species with time in ...................two selected subjects ....................................................................................... 69 Figure 4.5: A scatter plot of post-treatment Lactobacillus crispatus species concentration ...................in vaginal fluid versus that of Gardnerella vaginalis with a Lowess smoother ...................plot. .................................................................................................................. 79 Figure 4.6: A scatter plot of post-treatment Lactobacillus crispatus species concentration ...................in vaginal fluid versus that of Lactobacillus iners with a Lowess smoother ...................plot. .................................................................................................................. 80 xii LIST OF PLATES Plate 2.1: Gram stains of vaginal fluid from a woman without BV (A) and from a woman ...................with BV (B). .................................................................................................... 16 Plate 2.2: Vaginal biopsy from a subject with BV. ........................................................... 24 Plate 3.1: The Study Site: San Francisco General Hospital (SFGH) and University of ...................California, San Francisco (UCSF), USA. ........................................................ 38 Plate 3.2: rep-PCR DNA fingerprints generated from different production lots of ...................Lactobacillus crispatus CTV-05 gelatin capsules produced from 1995 to 2001 ................... ......................................................................................................................... 47 Plate 4.1: Polaroid picture of one the repPCR DNA fingerprints generated from ...................Lactobacillus crispatus CTV-05 in this study.. ............................................... 59 xiii LIST OF APPENDICES Appendix 1: Enrolment Case Review Form (CRF) ........................................................ 120 Appendix 2: Eligibility Criteria ....................................................................................... 125 Appendix 3: Eligibility Criteria (Form 2) ....................................................................... 126 Appendix 4: Final Visit Case Review Form (CRF) ........................................................ 127 Appendix 5: Consent Form ............................................................................................. 130 Appendix 6: Kemri Scientific Steering Committee Approval ........................................ 143 Appendix 7: Kemri Ethical Review Committee Approval ............................................. 144 Appendix 8: UCSF Committee On Human Research Approval ..................................... 145 xiv LIST OF ABBREVIATIONS AND ACRONYMS β-hCG beta-human chorionic gonadotropin BLD below limits of detection bp base pairs BV bacterial vaginosis BVAB 1, 2, 3 BV-associated bacteria species 1, 2 and 3 CFU colony forming units CCRC CTSI Clinical Research Centre CI confidence interval CRF clinical review form/s CTSI Clinical Translational Science Institute CTV-05 Lactobacillus crispatus CTV-05 strain DGGE denaturing gradient gel electrophoresis DNA deoxyribonucleic acid ELISA enzyme-linked immunosorbent assay FAO Food and Agriculture Organization FDA Food and Drug Administration HIPAA Health Insurance Portability and Accountability Act HIV Human Immunodeficiency Virus HSV-2 Herpes Simplex Virus type 2 H2O2 hydrogen peroxide IDRTP Infectious Diseases Research Training Program xv IQR interquartile range IRB institutional review board ITT intent-to-treat IUD intra-uterine device KEMRI Kenya Medical Research Institute KOH potassium hydroxide LACTIN-V® probiotic Lactobacillus crispatus CTV-05 formulation LMP last menstrual period Log10 logarithm to base 10 MetroGel® metronidazole gel mg milligrams ml millilitres NaOH sodium hydroxide NIH National Institutes of Health nm nanometres NS Nugent score OR odds ratio PAP smear Papanikolaou smear for cervical cancer screening qPCR quantitative polymerase chain reaction RCT randomized controlled trial rep-PCR repetitive sequence-based polymerase chain reaction rRNA ribosomal ribonucleic acid xvi rpm revolutions per minute SFGH San Francisco General Hospital SOP standard operating procedures Study ID study identification number STI sexually transmitted infection UCSF University of California, San Francisco µl microlitre µm micrometre UK United Kingdom USA United States of America UTI urinary tract infection WHO World Health Organization xvii ABSTRACT Several fastidious bacteria have been associated with bacterial vaginosis (BV), but their role in lactobacilli recolonization failure is unknown. This study examined the effect of seven BV-associated bacterial species and two Lactobacillus species on vaginal colonization with L. crispatus CTV-05 (LACTIN-V). Twenty four women with BV were treated with metronidazole vaginal gel and then randomized 3:1 to receive either LACTIN-V or placebo. Vaginal swabs for L. crispatus CTV-05 culture and 9-bacterium specific 16S rRNA gene quantitative PCR assays were analyzed on several study visits for the 18 women receiving LACTIN-V. Vaginal colonization with CTV-05 was achieved in 61% of the participants receiving LACTIN-V at either the day 10 or the 28 visit and 44% at day 28. Participants not colonized with CTV-05 had generally higher median concentrations of BV-associated bacteria compared to those who colonized. Between enrollment and day 28, the median concentration of Gardnerella vaginalis minimally reduced from 104.5 to 104.3 16S rRNA gene copies per swab in women who colonized with CTV-05 but increased from 105.7 to 107.3 in those who failed to colonize (p=0.19). Similarly, the median concentration of Atopobium spp. reduced from 102.7 16S rRNA gene copies per swab to below limit of detection (375 rRNA gene copies per swab) in women who colonized with CTV-05 but increased from 102.7 to 106.6 in those who failed to colonize (p=0.04). The presence of endogenous L. crispatus at enrollment was found to be significantly associated with a reduced odds of colonization with CTV-05 on day 28 (p=0.003) and vaginal intercourse during the study significantly impaired successful CTV05 colonization (p=0.018). These results suggest that vaginal concentration of certain BVxviii associated bacteria, vaginal intercourse during treatment and presence of endogenous L. crispatus at enrollment predict failure of colonization with probiotic lactobacilli. xix CHAPTER ONE 1.0 INTRODUCTION 1.1 Background Bacterial Vaginosis (BV) is a common polymicrobial disorder characterized by an overgrowth of anaerobic or facultative bacteria and a reduction or absence of lactobacilli. BV accounts for 40% to 50% of all cases of vaginitis (Demba et al., 2005; Mitchell, 2004) and is associated with numerous disorders of the female genito-urinary tract including adverse pregnancy outcomes (Jacobsson et al., 2002; Karaer et al., 2005; Schwebke 2003; Svare et al., 2006) sexually transmitted infections (STI) (Bradshaw et al., 2005; Demirezen et al., 2005; Ness et al., 2005; Schwebke 2003) and human immunodeficiency virus (HIV) acquisition (Taha et al., 1998). The vaginal ecology is dynamic, where a Lactobacillus-dominant flora maintains an optimum acidic pH, which suppresses BV-associated bacteria. A change in normal vaginal flora including the reduction of Lactobacillus allows pathogenic bacteria to multiply (Taha et al., 1998). The microorganisms involved in the BV pathogenesis are diverse, but most frequently include Gram-negative or Gram-variable coccobacilli (Gardnerella vaginalis and Bacteroides species (spp.)), curved Gram-variable anaerobic bacteria, other anaerobic organisms (Prevotella spp., Peptostreptococcus spp., Fusobacterium spp.) and genital Mycoplasmas (Mycoplasma hominis and Ureaplasma urealyticum) (Bradshaw et al., 2005). Recently, several fastidious vaginal bacteria have been identified using sequence1 based detection methods and reported to be associated with BV. These include Atopobium vaginae, Megasphaera species, Leptotrichia/Sneathia species and Bacterial Vaginosis Associated Bacteria species (BVAB1, BVAB2 and BVAB3) (Ferris et al., 2007; Fredricks et al., 2005a; Fredricks et al., 2007). BV treatment traditionally aimed at the eradication of anaerobic bacteria by use of antibiotics like clindamycin or metronidazole. While cure rates are comparable between these two drugs, they are unacceptably low (50%–80%) (Ferris et al., 2007) and are often unable to prevent BV recurrence (Carey et al., 2000), making the use of Lactobacillus probiotics a promising treatment and prevention strategy. Probiotics are live microorganisms that are an intrinsic part of the normal flora. When administered in adequate amounts they confer a health benefit on the host (Waigankar and Patel, 2011). They generally increase the normal flora by replenishing suppressed bacteria and inhibiting the growth of pathogenic flora mainly dominated by anaerobic bacteria (Cribby et al., 2008). The term probiotic was derived from a Greek word, meaning “for life” (FAO and WHO, 2001). The World Health Organization (FAO & WHO, 2001) has stated that there is adequate scientific evidence to suggest that there is potential for probiotic foods to provide health benefits. In developed countries over-the-counter probiotics have been in use for sometime albeit without sufficient scientific evidence of their effectiveness (Chondros et al., 2004). 2 Several factors compel the examination of probiotics and other alternatives to pharmaceutical remedies. While antibiotics have been shown to be effective in the treatment of most genito-urinary tract infections they are often unable to cure BV or prevent its recurrence (Carey et al., 2000). In addition, antibiotics have many side effects and multi-drug resistant microorganisms are emerging, which suggests that such chemotherapeutic agents ideally should not be used for chemoprophylaxis or health maintenance (Gardiner et al., 2003). Several Lactobacillus species, most of which are part of the healthy vaginal flora, have been tried as probiotics. Although strains of Lactobacillus fermentum and Lactobacillus rhamnosus have been investigated as probiotics to prevent urogenital infections (Anukam et al., 2006b; Martinez et al., 2009; Reid et al., 2003a), focus on Lactobacillus species commonly recovered from the vagina has been recommended (Kiss et al., 2007; Wilks et al., 2004). Lactobacillus crispatus and L. jensenii - both H2O2 producing species - are the predominant vaginal Lactobacillus species colonizing healthy women (including pregnant women) in China (Yan et al., 2009), Japan (Song et al., 1999a), Europe (Kiss et al., 2007; Vasquez et al., 2002), and the USA (Antonio et al., 1999; Fredricks et al., 2005a). L. Crispatus CTV-05, a naturally occurring human vaginal strain of L. crispatus, has thus been formulated as a probiotic, LACTIN-V®, by Osel Inc. (Santa Clara, California, USA). Previous vaginal formulations of L. crispatus CTV-05 at lower concentrations (106 - 5 x 108 colony-forming units (cfu)/capsule) coated by a gelatin capsule appeared to be safe in clinical trials when tested for the prevention of BV and recurrent urinary tract infection 3 (Czaja et al., 2007; Fredricks et al., 2005a). A higher concentration of L. crispatus CTV05 (2 × 109 cfu/dose) administered as a powder using a prefilled tampon-like applicator, in healthy women was found to be safe and acceptable (Hemmerling et al., 2009). In these trials, 40 - 69% of women were successfully colonized with L. crispatus CTV-05. Factors causing a sub-optimal vaginal colonization with exogenous lactobacilli are unknown. 1.2 Research Question What is the effect of BV-associated bacteria, endogenous Lactobacillus species and sociodemographic factors on the colonization of the vagina with probiotic Lactobacillus crispatus CTV-05 in women on treatment for bacterial vaginosis? 1.3 Statement of the problem Bacterial vaginosis accounts for 40% to 50% of all cases of vaginitis and is thought to be more common than either vulvovaginal candidiasis or trichomoniasis (Nyirjesy et al., 2007). BV is common in sub-Saharan Africa where a prevalence of 20–49% has been reported among women presenting to STI clinics with vaginal discharge, 21–52% among pregnant women attending antenatal clinics, and 37–51% in community-based surveys (Demba et al., 2005). BV treatment using antibiotics has been reported to have low cure rates (50%–80%) (Schwebke and Desmond, 2007b) and high recurrence rates up to 60%) one year post- 4 treatment (Bradshaw et al., 2006a). This high prevalence of BV in women especially in Africa, the low antibiotic cure rates, the high recurrence rates and the increased risk that BV places these women to others STIs, particularly HIV, necessitates early trials of promising and safe interventions such as Lactobacillus probiotics. However, there is still insufficient evidence on the effectiveness of Lactobacillus probiotics in the treatment and prevention of recurrence of BV and other urogenital infections. This may be due to the varying rates of Lactobacillus colonization after probiotic treatment. Previous clinical trials using L. crispatus CTV-05 at 1 x 106 to 2 x 109 colony-forming units per dose (formulated as gelatin-coated capsules or administered as a powder using a prefilled tampon-like applicators) reported successful colonization of 40-69% of healthy women with L. crispatus CTV-05 (Antonio et al., 2009; Czaja et al., 2007; Hemmerling et al., 2009; Hemmerling et al., 2010). In another study which used vaginal gelatin capsules containing L. gasseri and L. rhamnosus at 109 cfu per capsule, 35.1% of the lactobacilli treated women were reported to have BV relapse after 6 menstrual cycles (Larsson et al., 2008). Factors causing this sub-optimal vaginal colonization with exogenous lactobacilli and recurrence of BV are unknown, but could include high vaginal levels of BVassociated bacteria, high levels of other lactobacilli and some sociodemographic and sexual factors. This study explored the ability of L. crispatus CTV-05 (LACTIN-V®) to colonize the vagina in premenopausal women treated for BV and sought to determine the impact of BV-associated bacteria, other lactobacilli, sociodemographic and sexual factors on CTV-05 colonization. 5 1.4 Study Justification Elucidating the factors that enhance or inhibit probiotic Lactobacillus colonization of the vagina is vital to ensure their success in the treatment and prevention of BV. If the presence or the vaginal concentration of BV-associated bacteria is found to affect colonization with L. crispatus CTV-05 then more aggressive antibiotic regimens of BV treatment could be sought. This will then improve the chance of Lactobacillus vaginal colonization and reduce the possibility of BV recurrence in these women. If the effectiveness of lactobacilli probiotics in the treatment and prevention of BV is enhanced, they could in future provide a safe and reliable alternative treatment and preventive regimen to antibiotics. They could also play a major role, if widely used, in the reduction of transmission of HIV and other sexually transmitted infections. 1.5 Hypothesis Null Hypothesis (H0): There is no significant association between BV-associated bacteria and vaginal colonization with L. crispatus CTV-05 in premenopausal women diagnosed with BV and treated with topical metronidazole and LACTIN-V®. 6 1.6 Objectives 1.6.1 General objective To determine the effects of BV-associated bacteria, endogenous Lactobacillus species and sociodemographic factors on vaginal colonization with the probiotic Lactobacillus crispatus CTV-05 (LACTIN-V®). 1.6.2 Specific objectives 1) To determine the proportion of women colonized with Lactobacillus crispatus CTV05 after treatment for BV with topical metronidazole and subsequent treatment with LACTIN-V®. 2) To identify and quantify BV-associated bacteria present in the vagina before and after treatment and assess the effect these bacteria have on Lactobacillus crispatus CTV-05 vaginal colonization. 3) To identify and quantify other Lactobacillus species present in the vagina both before and after treatment and assess the effect these bacteria have on Lactobacillus crispatus CTV-05 vaginal colonization. 4) To describe the socio-demographic and sexual factors affecting Lactobacillus crispatus CTV-05 vaginal colonization in this population. 7 CHAPTER TWO 2.0 LITERATURE REVIEW 2.1 Bacterial Vaginosis 2.1.1 Definition of bacterial vaginosis Bacterial vaginosis (BV) is a type of vaginitis usually characterized by an abnormal vaginal discharge with an unpleasant fishy smell. BV is characterized microbiologically by a shift from the normal Lactobacillus-dominant vaginal flora – which helps to maintain a low pH in the vagina by generation of lactic acid, hydrogen peroxide (H2O2) and other sialidases - to their gradual or total replacement with other bacteria that are primarily anaerobic (Demba et al., 2005). The cause of this shift is not fully understood. The vaginal ecology is dynamic, where a Lactobacillus-dominant flora maintains an optimum acidic pH, which suppresses BV-associated bacteria. A change in normal bacterial flora including the reduction of lactobacilli, which may be due to the use of antibiotics or pH imbalance, allows more resistant bacteria to gain a foothold and multiply (Hawes et al., 1996). In turn these bacteria produce toxins which affect the body's natural defences and make re-colonization of healthy bacteria more difficult (Kelly et al., 2003). This elevated vaginal pH which occurs as a result of Lactobacillus reduction allows growth of both BVassociated microflora and other sexually transmitted organisms (Taha et al., 1998). 8 2.1.2 Epidemiology of bacterial vaginosis Bacterial Vaginosis (BV) is one of the most common vaginal conditions in women of reproductive age and is a risk for STIs including HIV (Taha et al., 1998). It is one of the most frequently diagnosed disorders in women attending genitourinary medicine (GUM) (Mitchell, 2004), STI and other reproductive health clinics throughout the world (Demba et al., 2005). BV accounts for 40% to 50% of all cases of vaginitis and is thought to be more common than either vulvovaginal candidiasis or trichomoniasis (Nyirjesy et al., 2007). Since approximately half of all cases of bacterial vaginosis are asymptomatic, the true prevalence of this disorder is unknown (Mitchell, 2004). BV is particularly common in sub-Saharan Africa where a high prevalence has been reported in several studies. This ranges from 44% among women presenting to STI clinics in Nairobi (Bukusi et al., 2006) with vaginal discharge to 63% among HSV-2 seropositive women in the Lake Victoria region of Tanzania (Baisley et al., 2009). A prevalence of 48% has been reported in women attending STI clinics with vaginal discharge syndrome in The Gambia (Demba et al., 2005). Taha et al. (2007) reported a BV prevalence of 46% among HIV seronegative women and 60% among HIV seropositive women in Malawi. In contrast, BV prevalence reported from industrialised countries is varied. For instance, a 13% prevalence has been reported in GUM clinic attendees in the United Kingdom (UK) (Mitchell, 2004), 11% in gynaecology clinic attendees in London and between 15% to 30% in studies of non-pregnant women in USA (Demba et al., 2005). In contrast, Brabin et al. (2005) reported BV in 33% of adolescents attending GUM clinics in the UK. Bailey 9 et al. (2004) also reported a 31% BV prevalence in a cross-sectional survey of 708 new patients attending 2 sexual health clinics for lesbians and bisexual women in the UK. Although BV appears to be associated with sexual activity, there is no clear evidence of sexual transmission (Bump and Buesching, 1988; Yen et al., 2003). BV is associated with several sexual risk factors including early sexual debut (Bukusi et al., 2006; Nilsson et al.,1997), a new sexual partner, multiple sexual partners (Smart et al., 2004), a high number of lifetime sexual partners (Nilsson et al., 1997), frequent vaginal sex (Schwebke and Desmond, 2005), and a female sexual partner (Smart et al., 2004). Other factors which have been associated with BV are low social economic status, poor hygiene, marital status, HIV infection, STIs most commonly trichomoniasis, black race, pregnancy and smoking (Bukusi et al., 2006; Cherpes et al., 2008; Morris et al., 2001). BV has also been associated with receptive anal sex before vaginal intercourse, sex with an uncircumcised male partner, lack of vaginal H2O2-producing lactobacilli, and HSV-2 seropositivity. Use of hormonal contraception and condoms has been found to have an inverse association with BV (Smart, et al., 2004). A reduced rate of bacterial vaginosis is seen among women in monogamous sexual relationships. Though BV is rare in menopausal women and adolescents prior to sexual onset, it can occur in female virgins (Burton and Reid, 2002; Papanikolaou et al., 2002). Subclinical iron deficiency (anaemia) has also been shown to be a strong predictor of bacterial vaginosis in pregnant women (Verstraelen et al., 2005). A link between psychosocial stress and bacterial vaginosis has also been shown independent of other risk factors (Nansel et al., 2006). However, though there are many 10 factors which have been independently associated with BV none of them seems either specific or to have a dose-dependent relationship with this condition. 2.1.3 Aetiology of bacterial vaginosis The microorganisms involved in BV are very diverse, but the most frequently cultivated bacteria are Gram-negative or Gram-variable coccobacilli (Jones et al., 1985), curved Gram-variable anaerobic bacteria (Jones et al., 1985), other anaerobic organisms (Prevotella spp., Peptostreptococcus spp., Fusobacterium spp.) and genital Mycoplasmas (Mycoplasma hominis and Ureaplasma urealyticum) (Bradshaw et al., 2005) [Table 2.1]. Table 2.1: Classification of commonly isolated and some recently described BVassociated bacteria species Classification BV-associated bacteria Actinobacteria (phylum) o Gardnerella vaginalis o Mobiluncus curtsii and M. mulleris o Atopobium vaginae o Eggerthela spp. Firmicutes (phylum) i) Clostridia (class) o Megasphaera spp. o Peptostreptococcus spp. o BV-Associated Bacteria 1 (BVAB1) o BVAB2 o BVAB3 Bacteroidetes (phylum) o Prevotella spp. o Porphyromonas spp. o Bacteroides spp. ii) Mollicutes (class) o Mycoplasma hominis o Mycoplasma genitalium o Ureaplasma urealyticum Fusobacteria (phylum) o Leptotrichia spp. o Sneathia spp. o Fusobacterium spp. [Source: Oakley et al., 2008 and Fredricks et al., 2007] 11 Recently, several fastidious vaginal bacteria have been identified using sequence-based detection methods which add to the list of potential bacterial pathogens in BV. These bacteria include Atopobium vaginae, Megasphaera species, Leptotrichia/Sneathia species and Bacterial Vaginosis Associated Bacteria (BVAB1, BVAB2 and BVAB3). However, despite this advancement in bacteria identification, Koch’s postulates for establishing disease causation have not been fulfilled for any bacterium or group of bacteria associated with bacterial vaginosis (Fredricks et al., 2005a). Though several bacteria have been implicated in bacterial vaginosis such as Gardnerella vaginalis and Mobiluncus curtisii, these species are also found in patients who do not meet the diagnostic criteria for BV. Thus, these bacteria are just non-specific markers of this condition. It is not known whether the primary event initiating BV is the loss of key lactobacilli or acquisition of the complex bacterial communities found in this syndrome; these may be simultaneous processes. At least 2 models exist to explain the pathogenesis of BV (Figure 2.1). The Lactobacillus depletion model suggests that there is a decrease in hydrogen peroxide producing lactobacilli as the primary event that allows for the overgrowth of facultative anaerobes resulting in BV. The primary pathogen model suggests that the entry of facultative anaerobes causes the displacement of lactobacilli thereby resulting in BV (Srinivasan and Fredricks, 2008). 12 LACTOBACILLUS DEPLETION MODEL ? Insult PRIMARY PATHOGEN MODEL Decrease in H2O2-producing lactobacilli Overgrowth of facultative & anaerobic bacterial opportunists Entry of facultative & anaerobic bacterial pathogens Displacement of lactobacilli Bacterial Vaginosis Figure 2.1: Competing models for the pathogenesis of bacterial vaginosis [Source: Srinivasan and Fredricks, 2008]. 2.1.4 Clinical presentation and diagnosis of bacterial vaginosis Malodorous homogenous greyish white vaginal discharge may be the only symptom of bacterial vaginosis, and many affected women are asymptomatic (Fredricks et al., 2005a). Some women may also experience pruritus, lower abdominal pain and pain during coitus (Rao et al., 2004). BV is commonly diagnosed either using Amsel criteria (Amsel et al., 1983) or using the Nugent scoring method (Nugent et al., 1991) depending on availability of materials, cost and experience of clinicians. The Amsel criteria is positive if any three of the following four criteria are present: microscopic examination of vaginal discharge identifies “clue cells” i.e. over 20% of vaginal epithelial cells covered in Gram negative bacteria on a wet-prep microscopic examination, anterior fornix vaginal pH >4.5, release of a fishy odour on addition of 10% KOH to the vaginal discharge (positive “whiff” test), and presence of an increased thin homogeneous white to grey adherent vaginal discharge 13 (Anukam and Reid, 2007). An alternative diagnostic approach is to use Gram’s staining of vaginal fluid to distinguish normal vaginal flora (i.e. Gram-positive rods and lactobacilli) from bacterial vaginosis flora (Gram-negative and Gram-variable morphotypes) according to the Nugent score which is based on a scoring system ranging from normal (predominantly lactobacilli rods) to BV (absence of lactobacilli and domination by Gram negative rods and Gram variable coccobacilli) (Anukam and Reid, 2007; Fredricks et al., 2005a) [Plate 2.1]. Scores between 0 and 3 represent 'normal vaginal flora', between 4 and 6 'intermediate vaginal flora', and scores between 7 and 10 are considered diagnostic for 'BV' (Demba et al., 2005). Only a few of the bacteria species present in most microbial ecosystems are amenable to propagation in the laboratory (culture) and bacterial cultivation of vaginal fluid too has not proved useful for the diagnosis of BV (Fredricks et al., 2005a). Recent advances in the use of PCR-amplified ribosomal RNA genes (16S rDNA) have improved identification of fastidious and non-cultivable organisms. The combination of PCR and denaturing gradient gel electrophoresis (DGGE) allows identification of a high number of bacterial species in the complex ecosystems of the human body (Anukam and Reid, 2007; Devillard et al., 2005; Srinivasan and Fredricks 2008; Vitali et al., 2007). Several bacteria have therefore been identified using these methods including metronidazole-resistant anaerobe Atopobium vaginae (Ferris et al., 2007). 14 2.1.5 Adverse outcomes and sequelae of bacterial vaginosis BV has been associated with many disorders of the female reproductive and urinary tracts, including urinary tract infections (Schwebke, 2003), yeast vaginitis (Bradshaw et al., 2005), gonorrhoea (Ness et al., 2005), trichomoniasis (Demirezen et al., 2005), adverse pregnancy outcomes such as preterm labour (Karaer et al., 2005) and low-birth-weight infants (Svare et al., 2006), postpartum endometritis (Jacobsson et al., 2002), increased risk of infection after gynaecologic surgery and pelvic inflammatory disease (Nyirjesy et al., 2007). The association between BV and HIV has been demonstrated by a number of cohort studies in Africa. In a prospective cohort study of commercial sex workers done in Mombasa, Kenya, an increased risk of HIV-1 infection in those women who had abnormal vaginal flora was reported (Reid, 1999). In a study done in Malawi, BV was significantly associated with antenatal and postnatal HIV seroconversion with an approximate attributable risk of 23% (Taha et al., 1998). There was a significantly increased risk of HIV seroconversion with increasing severity of vaginal disturbance among both antenatal and postnatal women. (Schwebke and Desmond, 2007a), showed that treatment and prophylaxis of asymptomatic BV with vaginal metronidazole gel is associated with decreased incidence rates of STI’s including Chlamydia infection (Schwebke and Desmond, 2007a). Thus, theoretically, treatment of bacterial vaginosis and/or replacement of the vaginal lactobacilli could reduce a woman’s risk of acquiring HIV and other sexually transmitted infections (STI) (Gardiner et al., 2003). 15 A Lactobacilli Normal Epithelial Cell B Clue Cell Plate 2.1: Gram stains of vaginal fluid from a woman without BV (A) and from a woman with BV (B). Women without BV have large numbers of Gram-positive rods (lactobacilli) while women with BV have large number of Gram-positive and Gram-variable coccobacilli (which sometimes aggregate around and adhere to epithelial cells giving rise to clue cells) with few or no visible lactobacilli. [Source: archived photomicrographs, Centre for Microbiology Research, Kenya Medical Research Institute]. 16 The current incidence of HIV infection in developing countries creates an urgent need for testing novel female-controlled approaches in clinical trials, particularly the ones that have previous evidence of safety (Bolton et al., 2008). 2.2 Treatment of Bacterial Vaginosis 2.2.1 Anti-microbial treatment Treatment of BV has been traditionally aimed at the eradication of anaerobes using antimicrobials (Austin et al., 2005). For non-pregnant women this treatment often includes oral and intravaginal therapy with clindamycin or metronidazole. Several intravaginal antimicrobial therapies containing these antimicrobials have been approved for use in various patient populations for the treatment of BV, including 2% clindamycin single-dose vaginal cream and 0.75% metronidazole vaginal gel (Nyirjesy et al., 2007). However, cure rates, although comparable for these 2 agents, are unacceptably low (50%–80%) (Schwebke and Desmond, 2007b). Mycoplasmas and Mobiluncus curtisii, which may play a role in the aetiology of BV, have in vitro resistance to metronidazole but are susceptible to macrolides. Failure to adequately treat these organisms could conceivably contribute to treatment failure. 17 2.2.2 Recurrence of BV after anti-microbial treatment Recurrence of bacterial vaginosis after antimicrobial treatment is common (Bradshaw et al., 2006a). The current recommended treatment has not been shown to prevent the recurrence of BV or abnormal vaginal flora in the majority of women (Table 2.2). Table 2.2: Bacterial vaginosis recurrence in clinical trials which used antibiotics and probiotics for the treatment of BV Treatment/ Intervention Author Bradshaw et al., 2006a N Treatment (days) 139 Oral Metronidazole (7) BV recurrence Follow up Treatment Placebo 1 Year 58% -* 409 HIV + Metronidazole gel (7 x 3 months) 3 Months 47% 46% 381 HIV - Metronidazole gel (7 x 3 months) 3 Months 33% 38% Taha et al., 2007 Clindamycin gel + L. gasseri and 6 Months 65% 46% L. rhamnosus [EcoVag®] (10 x 3) *There was no placebo group in the Bradshaw et al. (2006) study [Source: This table was constructed using published results from various studies whose references are listed]. Larsson et al.,2008 76 Among 139 women treated with oral metronidazole for 7 days and then followed up over a year for BV recurrence, recurrent BV was found in 68 (Martinez et al., 2000) women (NS 7-10) and abnormal vaginal flora in 84 (Stroman et al.,2003) of the women (NS 4-10) (Bradshaw et al., 2006a). They found that a past history of BV, a regular sex partner throughout the study, and female sex partners were significantly associated with recurrence of BV and abnormal vaginal flora. The use of hormonal contraception was found to have an inverse association with BV recurrence. Personal hygiene practices, such as vaginal douching, that disrupt the normal flora have also been show to increase BV 18 recurrence (Bradshaw et al., 2005). Incidence of persistent/recurrent BV has been associated with baseline detection of A. vaginae, BVAB1, BVAB2, BVAB3, Peptoniphilus lacrimalis and Megasphaera phylotype 2 (Bradshaw et al., 2006b; Marrazzo et al., 2008). 2.2.3 Vaginal lactobacilli The main Lactobacillus species found in healthy women include L. crispatus, L. gasseri, L. jensenii and L. iners. L. crispatus has been cultured from a majority of healthy women in a number of studies. (Yan et al., 2009) cultured L. crispatus from 94% of healthy women attending a gynaecological clinic in Beijing and 83% of women with BV, with the former having a colony count average value of 106 and the latter of 103. L. gasseri, L. iners, and L. jensenii were cultured from 85%, 68% and 43% of healthy women; and 28%, 89% and 44% of BV women, respectively. Song et al. (1999b) reported that L. crispatus and L. gasseri are the predominant vaginal isolates colonizing Japanese women. Similarly, Vasquez et al. (2002) reported that L. crispatus is the predominant vaginal species in healthy Swedish women. L. crispatus has also been found to be the predominant Lactobacillus species in women without BV in the US (Antonio et al., 2005a). Some studies have also been done to assess Lactobacillus colonization in pregnant women. Kiss et al. (2007) studied vaginal colonization in 200 healthy women in the late first trimester of pregnancy and reported that the most frequently occurring species were L. crispatus and L. gasseri, followed by L. jensenii and L. rhamnosus. This could be 19 because L. crispatus especially strain CTV-05 has been found to be highly adherent to vaginal epithelial cells of premenopausal women (Kwok et al., 2006). Lack of Lactobacillus species in the vagina has been associated with colonization with pathogenic bacteria including BV-associated species and an increase in the risk of STIs, UTIs and other genito-urinary infections (Reid et al., 2003b). Marrazzo et al. (2006) observed a significant association between cervicitis and the absence of H2O2 -producing Lactobacillus species. They also noted that of the women who were colonized with vaginal H2O2-producing Lactobacillus species, those who had cervicitis had significantly lower quantities of these lactobacilli. Vaginal washing, typically with water alone or with soap and water and recent antibiotic use (excluding metronidazole and treatments for vaginal candidiasis) have been associated with decreased likelihood of Lactobacillus isolation, including isolation of H2O2-producing strains (Baeten et al., 2009). H2O2producing lactobacilli are less common among women with Trichomonas vaginalis infection and those who are seropositive for herpes simplex virus type 2. 2.2.4 Interaction between vaginal lactobacilli and BV-associated bacteria Some in-vitro studies have shown that specific strains of Lactobacillus are able to coaggregate with G. vaginalis and block the adherence and/or displace previously adherent strains of G. vaginalis from vaginal epithelial cells. Boris et al. (1998) showed that L. acidophilus, L. gasseri and L. jensenii, isolated from the vaginal samples of healthy 20 pre-menopausal women, coaggregated in-vitro with G. vaginalis. L. acidophilus was also found to decrease the adherence and to displace previously adherent strains of G. vaginalis from vaginal epithelial cells. Mastromarino et al. (2002) found that human isolates of L. salivarius FV2 and L. gasseri 335 coaggregated in-vitro with G. vaginalis, and that vaginal tablets containing a combination of these organisms with L. brevis CD2 (which is strongly adherent to epithelial cells) reduced the adhesion by 57.7% and displaced 60.8% of G. vaginalis previously attached to these epithelial cells. Some Lactobacillus strains have also been found to have an inhibitory effect on the invitro growth of pathogens that cause BV, which may be caused, in part, by the production of H2O2. Matu et al. (2009) obtained 158 strains of Lactobacillus and assayed their supernatants for antibacterial activity on G. vaginalis, P. bivia, B. fragilis and Mobiluncus spp. They reported that none of the Lactobacillus spp. tested was able to inhibit growth of B. fragilis, but they inhibited growth of 37 of 158 (23%) of P. bivia, 45 of 158 (28%) of G. vaginalis, and 46 of 158 (29%) of Mobiluncus spp. Mastromarino et al. (2002) found that L. salivarius FV2 and L. gasseri 335, isolated from the human vagina, produced large amounts of H2O2 and inhibited the growth of G. vaginalis. McLean and Rosenstein (2000) showed that L. acidophilus 48101, isolated from the vagina of healthy women, produced large amounts of H2O2 and inhibited the growth of Bacteroides spp., Prevotella bivia and G. vaginalis isolated from vaginal swabs of women with BV. 21 Production of lactic acid by lactobacilli, which is mainly responsible for the low vaginal pH, contributes, probably even more than production of H2O2, to the inhibition of growth of G. vaginalis. An in-vitro study by McLean and McGroarty (1996) showed that the bacteriostatic effect of L. acidophilus on G. vaginalis NCTC 11292 was reduced by 60% after increasing the culture pH by the addition of NaOH, and by 30% after denaturing H2O2 with catalase. Thus, production of lactic acid, which was mainly responsible for the low pH, and, to a lesser degree, production of H2O2 by L. acidophilus, affected the growth inhibition of G. vaginalis significantly. Klebanoff et al. (1991) reported that H2O2producing lactobacilli at high concentrations inhibited the growth of G. vaginalis and Bacteroides bivius. Catalase inhibited the toxicity of H2O2-producing lactobacilli, but not that of non-H2O2-producing lactobacilli. Lower concentrations of H2O2-producing lactobacilli were toxic to G. vaginalis, but only when combined with myeloperoxidase and chloride, which have both been found in cervical mucus. The highest toxicity of this combination was obtained at pH 5–6. A pH of 4.5 had, by itself, an inhibitory effect on the growth of G. vaginalis, which was increased when the above combination was added. Production of bacteriocins by some Lactobacillus strains has also been found to play a role in the inhibition of growth of G. vaginalis, at least in-vitro. Aroutcheva et al. (2001) tested 22 Lactobacillus strains and found that 80% produced a bacteriocin that inhibited the growth of G. vaginalis. Simoes et al. (2001) showed that the growth of 28 of 36 clinical isolates of G. vaginalis was inhibited by a bacteriocin-producing L. acidophilus strain, and characterised these G. vaginalis isolates as bacteriocin-susceptible. 22 A number of studies have also shown that BV-associated bacteria have well developed virulence properties which help them overcome the above inhibitory factors expressed by lactobacilli and possibly also to resist attempts by lactobacilli to recolonize the vagina. Kelly et al. (2003) demonstrated inhibition of vaginal lactobacilli by a bacteriocin-like inhibitor produced by Enterococcus faecium. BV-associated bacteria also have the ability to form protective biofilms mainly by tenaciously adhering to vaginal epithelial cells (Patterson et al., 2010). Swidsinski et al. (2005) also showed that G. vaginalis is able to form an adherent biofilm on the vaginal epithelium of women with BV. Vaginal biopsies examined using florescent in-situ hybridization (FISH) with rRNA-targeted probes and a DNA-binding florescent stain (DAPI) have revealed G. Vaginalis biofilms in women with BV (Plate 2.2). The biofilm incorporates other bacterial groups into its layers e.g. Atopobium vaginae (Swidsinski et al., 2008), suggesting that it may enable other anaerobes to colonize the vagina. G. vaginalis also produces the toxin vaginolysin, which is a member of the cholesteroldependent family of pore-forming toxins that lyses human red blood cells and vaginal epithelial cells (Gelber et al., 2008; Randis et al., 2009). It is possible that the strong adherence by these bio-films to the vaginal epithelial cells prevents exogenous probiotic Lactobacillus species from adhering to the epithelial cells. 23 (a) (b) Plate 2.2: Vaginal biopsy from a subject with BV. A Gardnerella vaginalis biofilm (yellow) is detected at the edge of the vaginal epithelium (a) by fluorescence in situ hybridization (FISH). The yellow color is the result of using a combination of probes targeting G. vaginalis (Red), all bacteria (green), and DNA (blue). Note human vaginal epithelial cell nuclei in blue. The image on the right (b) shows a vaginal epithelial cell with a cluster of G. vaginalis breaking off the epithelium and likely forming a clue cell. [Source: Srinivasan and Fredricks, 2008] 2.2.5 Use of Lactobacillus probiotics in BV treatment Several species of Lactobacillus most of which are present in the healthy vagina have been tried as probiotics to prevent or treat urogenital infections or maintain a healthy vaginal environment (Table 2.3). Several clinical trials have been done to investigate whether specific strains of lactobacilli, administered either orally or intra-vaginally, are able to colonize the vaginas of women with symptomatic or asymptomatic BV, to reduce the colonization of pathogens, and to improve symptoms and/or signs of BV when they are present (Table 2.4). 24 Table 2.3: Classification of selected lactobacilli for probiotic use based on metabolic production of lactic acid from sugars Lactobacilli Classification Obligately homofermentative Lactobacillus delbreuckii group Facultative heterofermentative Lactobacillus casei group Obligately heterofermentative Lactobacillus reuteri group o Lactobacillus acidophilus o Lactobacillus casei o Lactobacillus reuteri o Lactobacillus crispatus o Lactobacillus paracasei o Lactobacillus fermentum o Lactobacillus gasseri o Lactobacillus rhamnosus o Lactobacillus johnsonii Lactobacillus salivarius group o Lactobacillus plantarum group o Lactobacillus plantarum Lactobacillus salivarius [Source: Holzapfel et al., 2001]. Reid et al. (2003) reported that daily oral intake of probiotics strains of Lactobacillus rhamnosus GR-1 and Lactobacillus fermentum RC-14 by 19 women with asymptomatic bacterial vaginosis resulted in 81% of the patients reverting to normal lactobacilli dominated microflora compared to 50% of those who took a placebo. Later, in a randomized placebo-controlled trial of 64 women given daily oral intake of the same probiotic capsule for 60 days, they reported restoration from asymptomatic bacterial vaginosis microflora to a normal lactobacilli colonized microflora in 37% women during lactobacilli treatment compared to 13% on placebo (Reid et al., 2003a). In another trial, subjects used oral metronidazole (500 mg) twice daily for seven days and were then randomized to receive the oral probiotic capsules (GR-1 and RC-14) twice daily for 30 days or a placebo. In this trial, 88% were cured in the antibiotic/probiotic group compared to 40% in the antibiotic/placebo group (Anukam et al., 2006a). In a second study using the 25 Table 2.4: Summary of Randomized Clinical Trials (RCT) using probiotics in women with bacterial vaginosis Treatment/ Intervention N Premedication (number of days) 28 none 64 none 46 none Parent et al. (1996) 32 none Vaginal tablet containing L. acidophilus + 0.03mg oestradiol (6) Placebo (6) 4 weeks Fewer cases of BV and greater number colonized with lactobacilli (p=0.05) Eriksson et al. (2005) 225 Vaginal Clindamycin ovules (3) Tampons containing 3 lactobacilli species Placebo (6) 2 menstrual cycles No statistical difference in BV cure rates after 2nd menstrual period Anukam et al. (2006a) 125 Oral Metronidazole (7) Placebo (60) 3 months 88% 40% 40 none Metronidazole gel (5) 30 days 90% 55% 100 Clindamycin gel (7) Twice daily oral capsules of L. rhamnosus GR-1 and L. fermentum RC-14 (60) Oral L. rhamnosus GR-1 and L. fermentum RC-14 (5) L. gasseri and L. rhamnosus (EcoVag®) (10x3) Placebo 30 days 77% 78% 87 Normal Women L. Crispatus CTV-05 [108 cfu] (3) L. Crispatus CTV-05 [106] 28 days Author Hallen et al. (1992) Neri et al. (1993) Shalev et al. (1996) Anukam et al. (2006b) Larsson et al. (2008) Antonio et al. (2009) Probiotic Treatment (number of days) Intravaginal capsules containing L. acidophilus (6) Intravaginal yoghurt containing L. acidophilus Dairy oral yoghurt containing L. acidophilus Outcome (Normal flora or cure) Control (number of days) Length of follow-up Treatment Control Placebo (6) 10 days 57% 0% 1) Acetic acid tampons 2) No treatment 1 month and 2 months 1 month or 2 months No treatment [Source: This table was constructed using published results from various studies whose references are listed]. 26 Decreased BV compared to acetic acid (p=0.05) and no treatment (p=0.0005) Decreased episodes of BV (p=0.001) No differences in colonization between doses BUT 60 % colonization in the two groups. same probiotic (GR-1 and RC-14) capsules were taken each night for 5 days, or 0.75% metronidazole gel, applied vaginally twice a day. Follow-up at day 6, 15 and 30 showed cure of BV in significantly more probiotic treated subjects (90% cure) compared to those treated with metronidazole gel (Anukam et al., 2006b). Some RCTs have suggested that intra-vaginal administration of L. acidophilus for 6–7 days cures women with BV significantly more frequently than administration of a placebo, administration of acetic acid, or no treatment. Hallen et al. (1992) found that significantly more women with BV were cured 7–10 days after the start of treatment with L. acidophilus when compared with those treated with a placebo. Restoration of a normal vaginal microbiota was established in significantly more (57%, 16⁄ 28) women with BV who were treated with L. acidophilus than in those receiving a placebo (none of 29). Another RCT involving pregnant women with BV showed that significantly more women treated with L. acidophilus intra-vaginally were cured of BV at both 1 and 2 months after the end of treatment when compared with women treated with acetic acid or given no treatment (Neri et al., 1993). Parent et al. (1996) found that cure was more common, and the number of vaginal lactobacilli was significantly higher, in women with BV at both 2 and 4 weeks after the start of a 6-day treatment with L. acidophilus and oestriol, when compared with women with BV who received a placebo (Table 2.4). Probiotic trials which used Lactobacillus crispatus CTV-05 (LACTIN-V®) are described elsewhere in section 2.3.2. 27 2.3 Probiotic Lactobacillus Crispatus CTV-05 (LACTIN-V®) 2.3.1 Suitability of Lactobacillus crispatus CTV-05 as a vaginal probiotic Lactobacillus crispatus CTV-05 (LACTIN-V®) is a naturally occurring human vaginal strain of L. crispatus, a gram-positive rod isolated from the vagina of a healthy woman (Hemmerling et al., 2010). Lactobacillus crispatus is found in the vaginas of healthy women and is also commonly found as a component of the natural human intestinal flora. It is a facultative anaerobe, fermentor of lactic acid, fastidious in its growth, and capable of H2O2 production. It also produces biosurfactants that inhibit pathogen adherence and it is able to prime macrophages, leukocytes, cytokines, and other host defenses (Waigankar and Patel 2011). Unlike most commercially available strains of Lactobacillus, CTV-05 adheres well to vaginal epithelial cells and is capable of colonizing the vaginal epithelium (Toba et al., 1995). Vaginal Gram stains from healthy women without BV typically show lactobacilli. Although geographic and racial variations regarding the predominant Lactobacillus have been recorded, studies in China (Yan et al., 2009), Japan (Song et al., 1999a), Europe (Kiss, et al., 2007; Vasquez et al., 2002) and USA (Antonio, et al., 2005b) have reported the predominance of L. crispatus in normal women including pregnant women. The naturally occurring vaginal Lactobacillus crispatus CTV-05 strain has thus been selected as a probiotic due to its capability to produce antibacterial materials, such as 28 H2O2, lactic acid and biosurfactants; its potentially strong adherence to vaginal epithelium; and its ability to prime macrophages, leukocytes, cytokines, and other host defenses. 2.3.2 Randomised Controlled Trials with LACTIN-V® LACTIN-V® is a lactobacilli containing probiotic which is a product of Osel Inc., Santa Clara, California, USA. It contains Lactobacillus crispatus CTV-05 which is the predominant lactic acid and H2O2 producing Lactobacillus found in the vagina of healthy women. It has been shown to be safe and acceptable in women treated for BV (Hemmerling et al., 2010). The first formulation of LACTIN-V® had been tested in several Phase І and ІІ studies at 5 x 106 CFU/capsule (Phase І) and 5 x 108 CFU/capsule (Phase І and ІІ). In the first Phase І studies LACTIN-V® was found to colonize 20% and 40% of those treated with the 5 x 106 CFU/capsule and those treated with 5 x 108 CFU/capsule respectively (Hemmerling et al., 2009). In a subsequent phase ІІ study conducted in 2005/2006 in 149 women with a history of recurrent BV who had recently been treated with metronidazole or clindamycin, LACTIN-V® colonization of 41.7% Vs. 6.2% was achieved in treatment and the placebo group respectively in one cohort and 59.3% Vs. 12.1% in the treatment and the placebo group respectively among the perfect user cohort (Osel Inc., unpublished data, 2006). Antonio et al. (2009) evaluated two potencies of gelatin capsules containing Lactobacillus crispatus CTV-05 (either 106- or 108-cfu) for vaginal colonization in 90 premenopausal 29 women without BV. Ninety percent of the women they enrolled in that study were colonized by lactobacilli (76% by H2O2 producing strains) before using the probiotic L. crispatus CTV-05. They found no statistically significant difference between the 2 potency groups with respect to vaginal colonization by L. crispatus CTV-05 at each individual visit or overall colonization, suggesting that the 2 doses were similarly efficacious at establishing colonization. Overall, they recovered CTV-05 at 1 follow-up visit or more for 60 of the 87 participants (69%). They achieved CTV-05 colonization at the 28-day visit in 51 (59%) of the 86 participants who completed all visits (Table 2.4). Thus, the colonization rates achieved by the foregoing studies were not adequate to answer questions on efficacy of LACTIN-V® to prevent BV recurrence. Higher LACTINV® colonization rates are desirable to induce cure and to prevent BV recurrence. It has thus become important to determine the factors that may influence vaginal colonization with L. crispatus CTV-05. These factors may also influence vaginal colonization with other probiotic lactobacilli being studied by other research groups worldwide for the treatment of BV. 2.3.3 Factors that may affect vaginal colonization with probiotic Lactobacillus crispatus CTV-05 Factors that may affect sustained colonization of the vagina by probiotic L. crispatus CTV-05 may be grouped into microbiologic, sociodemographic and sexual behaviour factors. The microbiologic factors can be placed into two categories: those intrinsic to the 30 Lactobacillus and those associated with other vaginal organisms including BV-associated bacteria (Vallor et al., 2001). 2.3.3.1 Possible effects of BV-associated bacteria on vaginal colonization with L. crispatus CTV-05 The prevalence of BV-associated bacteria before, throughout and after treatment and the effect they may have on colonization of the vagina with probiotic L. crispatus CTV-05 has not been previously evaluated. Several studies have demonstrated inhibition of lactobacilli by bacteriocins produced by BV-associated bacteria. Kelly et al. (2003) demonstrated inhibition of vaginal lactobacilli by a bacteriocin-like inhibitor produced by Enterococcus faecium. This species is associated with recurrent BV in women who have been treated with antibiotics. Yeoman et al. (2010) showed the genetic potential of G. vaginalis strains to produce substances that were antagonistic to bacterial isolates common to the vaginal biome. It is possible that more of the BV-associated bacteria produce bacteriocins or other substances which inhibit the growth of lactobacilli. Thus, bacteriocin production may prevent establishment of the normal vaginal microflora and could be a novel mechanism contributing to inhibition of colonization with L. crispatus CTV-05 and BV recurrence in this study. One of the most important properties of BV-associated bacteria is their ability to form protective biofilms mainly by tenaciously adhering to vaginal epithelial cells. A biofilm is 31 an aggregate of microorganisms in which cells adhere to each other and/or to a surface embedded within a self-produced matrix of extracellular polymeric substance (Braxton et al., 2005; Hall-Stoodley et al., 2004). Patterson et al. (2007) developed an in vitro model for G vaginalis bio-film and demonstrated that it tolerated 5-fold and 4-8 fold higher concentrations of H2O2 and lactic acid (respectively) than planktonic cultures. Proteolytic dissolution of biofilms reduced sensitivity to H2O2 and lactic acid. Patterson et al., (2010) went further and designed a viable assay to analyse bacterial adherence to vaginal epithelial cells, to compare biofilm-producing capacities and to assess cytotoxic activity. They reported that, of the BV-associated anaerobes they tested; only G. vaginalis demonstrated all three virulence properties combined. Their study suggests that G. vaginalis is more virulent than other BV-associated anaerobes, and that many of the bacterial species frequently isolated from BV may be relatively avirulent opportunists that colonize the vagina after G. vaginalis has initiated an infection. It is possible that G. vaginalis forms the initial biofilms which then enable other BV-associated bacteria to adhere and colonize the vagina. Swidsinski et al., (2008) followed-up 18 patients with bacterial vaginosis after a 7-day treatment with oral metronidazole and reported consistently observing the resurgence of a dense and active bacterial bio-film on the vaginal mucosa, primarily consisting of Gardnerella vaginalis and Atopobium vaginae with treatment cessation even though all patients recovered. It is possible that the strong adherence by bio-films to the vaginal 32 epithelial cells may prevent the exogenous L. crispatus CTV-05 strain used in this study from adhering to the epithelial cells and effectively colonizing the vagina. 2.3.3.2 Possible effects of other endogenous lactobacilli on vaginal colonization with L. crispatus CTV-05 There are several possible mechanisms through which endogenous lactobacilli already present in the vagina before probiotic treatment can affect colonization with L. crispatus CTV-05. First, endogenous lactobacilli could produce bacteriocins and bacteriophages harmful to exogenous L. crispatus CTV-05. It has been previously shown that some Lactobacillus strains from yogurts and other products used by women can release phages and bacteriocins that inhibit vaginal lactobacilli (Tao et al., 1997). Pavlova et al., (1997) isolated phages from vaginal lactobacilli of women with vaginal infections and demonstrated in vitro that these phages attacked and caused lyses of vaginal lactobacilli isolated from healthy women. It is possible that our study participants may harbour endogenous lactobacilli with capability to produce bacteriocins and phages capable of inhibiting or causing lyses of the probiotic L. crispatus CTV-05 used in this study. Secondly, vaginal concentrations of certain Lactobacillus species have been shown to be inversely associated with the concentration of other species. De et al., (2007) found a clear negative association between L. iners and L. gasseri and between A. vaginae and L. gasseri. Antonio et al. (2009) demonstrated that the presence of lactobacilli of the same species at enrollment could preclude colonization with L. crispatus CTV-05. It is possible 33 that some Lactobacillus species already present in the vagina could form biofilms and physically inhibit vaginal colonization with the probiotic L. crispatus CTV-05 used in this study. 2.3.3.3 Possible effects of sociodemographic and sexual behaviour factors on vaginal colonization with L. crispatus CTV-05 Several sociodemographic and sexual behaviour factors including frequency of sexual intercourse, antibiotic use, douching habits, marital status, employment, cigarette use, numbers of sex partners in the past year and a history of bacterial vaginosis have been reported to be associated with loss of colonization with H2O2-producing lactobacilli (Beigi et al., 2005; Mijac et al., 2006; Vallor et al., 2001). These factors may also affect the colonization of the vagina with exogenous Lactobacillus probiotics including L. crispatus CTV-05 used in this study. Antonio et al. (2009) showed that exposure to semen and vaginal intercourse during treatment predicts failure to become colonized by probiotic L. crispatus CTV-05. The high pH of seminal fluid may affect the adherence or survivability of CTV-05. Alternatively, a component of seminal fluid may block the adherence of CTV-05 to vaginal epithelial cells. It is also possible that CTV-05 is adherent to and is transferred by sperm (Toth et al., 1982). Sexual intercourse with the use of condoms also affects colonization, although less significantly than unprotected sex (Antonio et al., 2009). 34 CHAPTER THREE 3.0 MATERIALS AND METHODS 3.1 Study Materials 3.1.1 MetroGel® MetroGel® is an antibiotic gel consisting of 0.75% topical metronidazole and is used as a standard treatment for BV. Participants with BV as diagnosed with Amsel and Nugent criteria during the screening visit were given MetroGel® and instructed to apply vaginally once daily in the evening for five consecutive days. 3.1.2 LACTIN-V® applicator A conventional-shaped polyethylene vaginal applicator with a closed tip which contains petals that close together was used to hold the LACTIN-V® powder formulation and prevent leakage. When depressed, the plunger pushes the petals apart and pushes the formulation out into the vaginal canal. The barrel and tip measure 4.7 inches long by 0.5 inch in diameter. The applicator was earlier tested in a phase I trial (LV-005) assessing the safety, tolerability and acceptability of applicators pre-filled with LACTIN-V® or placebo control substance in 12 healthy women. The LV-005 study reported that LACTIN-V® and the applicator were acceptable to the women enrolled in the trial (Hemmerling et al., 2009). 35 3.1.3 LACTIN-V® Lactobacillus crispatus CTV-05 (LACTIN-V®) is a strain of L. crispatus, a Gram-positive rod isolated from the vagina of a healthy woman. Lactobacillus crispatus is found naturally in the vaginas of healthy women and is commonly found as a component of the natural human intestinal flora. It is a facultative anaerobe, fermentor of lactic acid, fastidious in its growth, and capable of H2O2 production. Unlike most commercially available strains of Lactobacillus, CTV-05 adheres well to vaginal epithelial cells and is capable of colonizing the vaginal epithelium. Each LACTIN-V® applicator contained 600mg of LACTIN-V® powder at a potency of 2 x 109 cfu per dose of L. crispatus CTV05 in a preservation matrix coated onto maltodextrin. The preservation matrix contained gelatin, skim milk, trehalose, xylitol, and sodium ascorbate. Both the study product and placebo control substance were manufactured at Osel’s facility in Louisville, Colorado, USA. 3.2 Study Methods 3.2.1 Study design This study assessed the treatment arm of a phase 2A blinded randomized placebocontrolled trial of 5 daily doses followed by one additional dose in week 2 and 3 of the LACTIN-V® powder (or placebo) administered vaginally via pre-filled applicators in women with BV immediately after the completion of a course of metronidazole vaginal gel (MetroGel®) applied for 5 days. 36 3.2.2 Study site The study was done at the University of California (UCSF), San Francisco General Hospital (SFGH), USA (Plate 3.1). SFGH is a licensed general acute care hospital within the Community Health Network, which is owned and operated by the City and County of San Francisco, Department of Public Health. SFGH runs several primary care and subspecialty clinics including an STI clinic where the study subjects were recruited. The bacteria identification and quantification through qPCR assays was done at the Program in Infectious Diseases, Fred Hutchinson Cancer Research Centre laboratories in Seattle, Washington, USA. 3.2.3 Study population and participant recruitment 3.2.3.1 Sample size (power calculation) This study’s sample population was part of a randomised-controlled trial (RCT) and included all the 18 women enrolled in the intervention arm of the RCT. The sample size of the reformulated/improved LACTIN-V® was chosen based on safety and clinical considerations rather than formal testing of treatment efficacy. The chosen sample size of 18 participants in the LACTIN-V® group and 6 participants in the placebo group was expected to fulfil the regulatory requirement of obtaining safety data on commonly occurring adverse events in a small number of subjects in a phase IIA study before moving into larger studies. Previous studies had shown successful LACTIN-V® colonization 37 City of San Francisco UCSF SFGH Plate 3.1: The Study Site: San Francisco General Hospital (SFGH) and University of California, San Francisco (UCSF), USA. State of California 38 The Bay Area between 40% and 60% of women treated. This being a new formulation of LACTIN-V® it was expected to achieve at least 70% colonization. In the power calculations several assumptions were made. The first assumption was that L. crispatus CTV-05 colonization would be achieved in 80% of the women without specific BV-associated bacteria species and in 20% of the women who had those bacteria. Secondly, a prevalence of between 20% and 50% of women for each of the 8 BV-associated bacteria species being studied was assumed. In the best case scenario, given an alpha of 0.05 and assuming a BV-associated bacteria prevalence of 50%, a sample size of 18 participants would give a power of 83% to detect a statistically significance risk ratio of 4 (95% CI 1.4 – 11) for those who were infected with a specific BV-associated bacteria to not get colonized with Lactobacillus crispatus CTV-05. Assuming a lower prevalence of specific BV-associated bacteria of 20% this would give a 48% power to detect a statistically significant risk ratio of 10 (1.34 – 74) (calculations done using Intercooled STATA® Version 10). 3.2.3.2 Participant recruitment Pre-menopausal women attending San Francisco State and UCSF clinics, Planned Parenthood, as well as students in university campus groups around San Francisco were targeted for recruitment. Willing participants were encouraged to visit the study clinic at the CTSI Clinical Research Centre (CCRC) at UCSF San Francisco General Hospital to attend screening for BV. Those who were clinically diagnosed and laboratory confirmed to have untreated BV during the screening visit (using the Nugent scoring system while the participant was still on site) and met the inclusion criteria were recruited into the 39 study. Information on the study was passed to potential participants using the local print and electronic media, San Francisco State and UCSF clinics as well as through outreach to university campus groups actively promoting gender and public health issues. Other big clinical providers such as Planned Parenthood Golden Gate were contacted to coordinate referrals of eligible women to the study. All advertisements in local newspapers, electronic media and radio were approved by UCSF’s Institutional Review Board (IRB). The study followed Health Insurance Portability and Accountability Act (HIPAA) guidelines where applicable. Potential participants interested in taking part in this study following media advertisement or community outreaches contacted the study personnel either by phone on a toll free 1800 number or via email and were provided with detailed information regarding the study and eligibility criteria whilst those approached by medical providers in referral clinics were provided with a description of the study by these providers who had undergone training regarding the purposes of the study. If the initial personal, telephone or email contacts established ongoing interest a preliminary appointment was made for screening at the UCSF CCRC. Participants presenting to the CCRC were counselled about study participation and written informed consent was sought (Appendix 5). Willing women were screened for study participation, and those who met the study eligibility criteria were enrolled as described in study procedures. 40 Participants were eligible for enrolment if they had regular menstrual cycles, were willing to insert pre-filled vaginal applicators, had a normal pap smear, agreed to abstain from the use of any other intravaginal product, were on an adequate method of birth control and gave consent. Participants were excluded if they were pregnant or within 2 months after pregnancy, currently breastfeeding, clinically detectable genital epithelial disruption at enrolment; urinary tract infection (UTI); HIV antibodies; history of recurrent genital herpes; syphilis; vaginal candidiasis; Trichomonas vaginalis; Neisseria gonorrhoeae, or Chlamydia trachomatis at screening visit; abnormal cervical cytology (Pap smear); initiation of a new long-acting treatment (e.g. depo-medroxyprogesterone acetate) within the past 2 months; history of recurrent vaginal infections (≥2 in past 6 months); active uncontrolled medical condition; use of a new investigational drug within 30 days or an immunosuppressive drug within 60 days prior to using the study product (Appendix 1). 3.2.4 Study procedures 3.2.4.1 Screening Visit (Visit 0 – Day 5) The purpose of the screening visit was to identify subjects that satisfied all eligibility criteria, including the diagnosis of symptomatic or asymptomatic bacterial vaginosis. Each woman who agreed to be screened was asked to sign the informed consent form written in English. The screening visit was also timed depending on the participant’s availability to return for the enrolment visit (Day 1) within 24-72 hours after the termination of the 5-day metronidazole (MetroGel®) treatment. The metronidazole treatment for women with BV 41 was initiated as soon as possible (ideally within five days of diagnosis at the screening visit). Women who gave informed written consent at the screening visit had the following specimens taken among other procedures and assessments: [1] Vaginal swab for Gram stain and culture. [2] Vaginal swabs for Lactobacillus culture and CTV-05 strain identification with rep-PCR. [3] Vaginal swab to identify selected BV-associated bacteria by q-PCR. [4] Vaginal swabs for L. crispatus by q-PCR. Screening visit (Visit 0) Enrolment visit (Visit 1) ~ Day -5 MetroGel® Day 1 Follow-up visit (Visit 2) Final visit (Visit 3) ~ Day 28 ~ Day 10 LACTIN-V® Figure 3.1: Chart of the clinical visits and study product administration 3.2.4.2 Enrolment visit (Visit 1 – Day 1) Those women who were diagnosed with BV at the screening visit, were treated with a 5day course of 0.75% topical metronidazole gel (MetroGel®) and were eligible and consented for the study were scheduled to return for the enrolment visit which took place 24-72 hours after using the last dose of topical metronidazole. LACTIN-V® treatment was commenced in this visit once a day for 5 consecutive days and thereafter two additional doses weekly (Figure 3.1). The following tests were done in this visit: 42 [1] Vaginal swab to identify selected BV-associated bacteria by q-PCR. [2] Vaginal swabs for L. crispatus by q-PCR. 3.2.4.3 Follow-up visit (visit 2 – Day 10) In this follow up visit participants were asked about symptoms and any medical problems since the last visit which was recorded on the appropriate Symptoms Clinical Review Form (CRF) and Adverse Event CRF (Appendix 4). Vaginal swabs were also taken for Gram-stain, culture, and rep-PCR but no qPCR tests were done in this visit. 3.2.4.4 Final visit (Visit 3 – Day 28) The participants were asked about symptoms and any medical problems since the last visit which were recorded on the appropriate Symptoms CRF and Adverse Event CRF (Appendix 4). The following tests were done in this visit: [1] Vaginal swab for Gram stain. [2] Vaginal swabs for L. Crispatus CTV-05 strain identification with rep-PCR. [3] Vaginal swab to identify selected BV-associated bacteria by q-PCR. [4] Vaginal swabs for L. crispatus by q-PCR. Women who had symptomatic BV during the final visit were re-treated with topical metronidazole (MetroGel®) as a case of BV four weeks after antibiotic treatment was considered as treatment failure. 43 3.2.5 Study measurements 3.2.5.1 Variables measured (1) Main Outcome Variable: Lactobacillus crispatus CTV-05 colonization of the vagina 28 days after the start of LACTIN-V treatment. (2) Main Predictor Variables: Presence, type and quantities of BV-associated bacteria at screening and enrolment as determined by PCR-amplified ribosomal RNA genes (16S rRNA). This was measured at the screening visit (Visit 0), enrolment visit (Visit 1) and at the final visit (Visit 3). (3) Other Predictor Variables: These were participants’ sociodemographic and sexual behavioural characteristics. 3.2.5.2 Diagnosis of bacterial vaginosis Women were screened for BV using the wet mount clinical Amsel criteria evaluated by the attending clinicians. Amsel criteria were defined as presence of characteristic white, homogeneous discharge; vaginal pH greater than 4.5; positive whiff test; and greater than 20% clue cells on wet mount (Amsel et al., 1983). A score of ≥3 was considered positive for BV. Additional analysis of BV using the laboratory-based Nugent scoring system was done at Magee Women’s Research Institute and Foundation in Pittsburgh, Pennsylvania. The Nugent score was calculated by assessing for the presence on Gram stain of large Gram-positive rods (Lactobacillus morphotypes; decrease in Lactobacillus scored 0 to 4), increase of small Gram-variable rods (G. vaginalis morphotypes; scored 0 to 4) and an 44 increase of curved Gram-variable rods (Mobiluncus spp. morphotypes; scored as 0 to 2) (Nugent et al., 1991). A score of ≥7 was considered positive for BV. Only women who scored ≥3 on the Amsel criteria and ≥7 on the Nugent’s criteria were enrolled into the study. 3.2.5.3 Other laboratory procedures for exclusion/inclusion Pregnancy was tested by rapid urine β-hCG assays (QuickVue+ Pregnancy Test, Quidel Corporation, San Diego, California). Urine was evaluated by urine dipstick for evidence of urinary tract infection (Chemstrips, Roche Diagnostics Limited, West Sussex, UK). HIV antibody testing was conducted using an ELISA (Genetic Systems HIV-1/HIV-2 Plus O EIA, Bio-Rad Laboratories, Hercules, California) with confirmatory Western Blot (Genetic Systems Reflex Western Blot by Bio-Rad) for ELISA positives. Syphilis was assessed by rapid plasma reagin (Becton-Dickinson, Franklin Lakes, New Jersey) for screening with a confirmatory agglutination test (Fujirebio Serodia T. pallidum Passive Particle Agglutination, Fujirebio Diagnostics, Malvern, Pennsylvania). Cervico-vaginal swabs were evaluated for T. vaginalis, N. gonorrhoeae, and C. trachomatis using nucleotide amplification tests (Gen-Probe Aptima Combo 2 assay by Gen-Probe Inc., San Diego, California). Pap smears were conducted on cervical specimens (ThinPrep Pap test, Cytyc Corporation, Marlborough, Massachusetts). Returned used applicators were assessed for exposure to vaginal mucus using 1% trypan blue staining technique (Martinez et al., 2009). Vaginal swabs were analyzed for Lactobacillus culture (at Cedars-Sinai Medical Centre, Los Angeles, California) and L. crispatus CTV-05 by DNA fingerprint 45 identity (determined by rep-PCR) at Consolidated Laboratory Services in Van Nuys, California. 3.2.5.4 Lactobacillus crispatus CTV-05 Identification using rep-PCR Assays Single baseline samples of vaginal fluid collected with a sterile swab from the 18 intervention subjects enrolled in the trial were placed in a Port-a-Cul anaerobic transport system and analyzed semi-quantitatively for presence of lactobacilli by culture (at CedarsSinai Medical Center, Los Angeles, California). To differentiate L. crispatus CTV-05 from other lactobacilli and from other L. crispatus strains, genomic DNA was extracted from Lactobacillus-positive cultures and subjected to repetitive-sequence polymerase chain reaction (rep-PCR) DNA fingerprinting at Consolidated Laboratory Services, Van Nuys, California (Mijac et al., 2006). Two repetitive extragenic palindromic (REP) consensus primers were used in the PCR reaction to amplify a characteristic pattern of DNA fragments, with intense bands observed at 1,500, 1,650, and 2,400 bp, and minor bands at 1,950 bp and a doublet at 2,100 bp (Antonio et al., 2003). Positive identification of L. crispatus CTV-05 (strain) requires the presence of all the bands (Plate 3.2). 3.2.5.5 Sample collection, DNA extraction and qPCR Assays 3.2.5.5.1 Sample collection and storage To obtain specimens for the performance of qPCR assays, a polyurethane foam swab (Catch-All; Epicentre) was brushed against the lateral vaginal wall to collect vaginal fluid, 46 re-sheathed, and frozen until the DNA extraction step. These swabs were stored dry (no media) at - 80°C. Plate 3.2: rep-PCR DNA fingerprints generated from different production lots of Lactobacillus crispatus CTV-05 gelatin capsules produced from 1995 to 2001 (lane 3 to 7). The original strain CTV-05, isolated in 1993, was run in lane 2, and a 1-kb DNA size ladder was run in lane 1. [Source: Antonio et al. (2003)] 3.2.5.5.2 DNA Extraction At the time of bacterial qPCR assays swabs were thawed and washed with 2 ml of saline in a vortex mixer for 1 minute to dislodge cells. A 1.0-ml aliquot of saline was centrifuged, at 14,000 rpm for 10 minutes, and the pellet was re-suspended in 100μl 47 supernatant. DNA was extracted from the pellet using the Ultra Clean Soil DNA Kit (MoBio, Carlsbad, California) according to the manufacturer’s instructions. DNA was eluted in 150 µl of buffer, and 2 µl of template DNA was used for each qPCR assay. Extracted DNA was frozen until assayed by qPCR. Swabs without human contact (sham digest controls) were subjected to DNA extraction in parallel with vaginal swabs, with one extraction control for every 12 swabs to control for contamination that may arise from kit reagents or collection swabs. 3.2.5.5.3 Bacterium Specific qPCR Assays The qPCR assays employed primers and a dual-labeled fluorogenic probe hydrolyzed during PCR (TaqMan format) for detecting each bacterium’s specific 16S rRNA gene in a highly sensitive and specific fashion. All probes had a carboxyfluorescein (FAM) reporter at the 5’ end and a tetramethyl-6-carboxyrhodamine (TAMRA) quencher or Black Hole quencher at the 3’ end. Probes were used at a concentration of 150 nM in each PCR. Core reagent kits from Applied Biosystems were used to assemble master mixes containing buffer A, deoxynucleotide triphosphates (0.8 mM), magnesium (final concentration, 3 mM), uracil-N-glycosylase, and TaqGold polymerase. Forward and reverse primers were added to master mix at a concentration of 0.8 µM. All qPCRs underwent 45 cycles of amplification. Primers and probes were designed using multiple-sequence alignments of vaginal bacterial 16S rRNA gene sequences (Fredricks et al., 2005a) (Table 3.1). qPCR assays targeted several vaginal bacteria that are significantly associated with BV or vaginal health (Fredricks et al., 2007), including a Megasphaera-like bacterium, 48 Table 3.1: Primer and probe sequences used in qPCR assaysα PCR specificity Primer or probe Primer/Probe sequence PCR conditions Amplicon size (bp) Sensitivity (no. of gene copies/reaction)β 5'-ACCTGGGCTTGACATGTGCCT-3' Gvag 983F 67°Cannealing/extension for 1 min 76 2.5 5'-CATGCACCACCTGTGAACCTG-3' Gvag 1059R 5'-FAM-GCAGAGATGTGGTTTCCYTTCG-TAMRA-3' Gvag 1008-1032 5'-AATTATTGGGCTTAAAGGGCATC-3' Lepto/Sneath 559F 5'-GTACTCTAGTTCAACAGTTTTGTAG-3' Lepto 659R Leptotrichia /Sneathia 65°C annealing/extension for 1 min 100-101 2.5 5'-AGTACTCTAGTTATACAGTTTTGTAG-3' Sneath 660R Lepto/Sneath 593-618 5'-FAM-ACAAGTTGAAGGTGAAAACCTRTGGC-TAMRA-3' 5'-GATGCCAACAGTATCCGTCCG-3' Mega1 456F 55°C annealing for 39 s, 5'-CCTCTCCGACACTCAAGTTCGA-3' Megasphaera type I Mega1 667R 211 2.5 72°C extension for 30 s 5'-VIC-CCGTGGCTTTCTCTTACGGTAC-TAMRA-3' Mega 485-506 5'-TAGGCGGTYTGTTAGGTCAGGA-3' Atop 582F 67°C annealing/extension for 1 min 81 5'-CCTACCAGACTCAAGCCTGC-3' Atopobium vaginae Atop 665R 2.5 5'-FAM-CTCAACCCCTATCCGCTCCTGATTAMRA-3' Atop 610-641 5'-GGAGTGTAGGCGGCACTA-3' BVAB1 576F 57°C annealing for 39 s, 5'-CTCTCCGATACTCCAGCTCTA-3' BVAB1 BVAB1 666R 90 5-2.5 72°C extension for 30 s 5'-FAM-ACCTAAGGCTTAACCATAGGATTGCATT-3' BVAB1 610-637 5'-TTAACCTTGGGGTTCATTACAA-3' BVAB2 619F 59°C annealing for 39 s, 5'-GAATACTTATTGTGTTAACTGCGC-3' BVAB2 BVAB2 879R 260 5-2.5 65°C extension for 30 s 5'-FAM-TCTCCAGCACTCAAGCTAAACAGBHQ1–3' BVAB2 643-665 5'-CATTTAGTTGGGCACTCAGGC-3' BVAB3 1132F 5'-ACATTTGGGGATTTGCTTCGCC-3' BVAB3 BVAB3 1292R 65°C annealing/extension for 1 min 160 5-2.5 5'-FAM-TACTACAATGGCTACAACAGAGAGC-TAMRA-3' BVAB3 1232-1256 5'-ACAGGGGTAGTAACTGACCTTTG-3' Liners 453F Lactobacillus iners 65°C annealing/extension for 1 min 85 5-2.5 5’-FAM-ATCTAATCTCTTAGAACTGGCTATGTAMRA-3’ Liners 1022R 5'-TCTTGACATCTAGTGCCATTTGT-3' Lcrisp 989F 63°C annealing for 39 s, 66 5'-TGCACCACCTGTCTTAGC-3' Lactobacillus crispatus Lcrisp 1055R 5 72°C extension for 30 s Lcrisp 1010-1033 5'-FAM-CCGAAGGAACTTTGTATCTCTACTAMRA-3' αPrimer and probe sequences used in qPCR assays are displayed, along with PCR conditions, amplicon lengths, assay detection thresholds per reaction (1 – 1.5 units of TaqGold polymerase were used in each PCR). All PCRs started with a 50°C incubation step for 2 min to inactivate previous amplicons using uracil-N-glycosylase, followed by 10-min incubation at 95°C to activate the TaqGold. All PCRs used a 95°C melting step for 15s prior to the annealing step. βSensitivity data are reported as numbers of bacterial 16S rRNA gene copies per reaction. Gardnerella vaginalis 49 Atopobium vaginae, the closely related Leptotrichia and Sneathia species (Genc et al., 2004), Gardnerella vaginalis, Lactobacillus crispatus, Lactobacillus inus and three Clostridium-like bacteria which have previously been designated BVAB1, BVAB2, and BVAB3. Real time quantitative PCR (qPCR) does not have a gel procedure or a photograph, rather fluorescence was monitored after each PCR cycle in a thermal cycler. An oligonucleotide probe constructed with a reporter fluorescent dye on the 5´ end and a quencher dye on the 3´ end was used. While the probe is intact, the proximity of the quencher dye greatly reduces the fluorescence emitted by the reporter dye by fluorescence resonance energy transfer (FRET) through space. If the target sequence is present, the probe anneals downstream from one of the primer sites and is cleaved by the 5´ nuclease activity of Taq DNA polymerase as this primer is extended. This cleavage of the probe separates the reporter dye from the quencher dye, increasing the reporter dye signal and also removes the probe from the target strand, allowing primer extension to continue to the end of the template strand. Thus, inclusion of the probe does not inhibit the overall PCR process. Additional reporter dye molecules are cleaved from their respective probes with each cycle, resulting in an increase in fluorescence intensity proportional to the amount of amplicon produced. qPCR was performed in tandem (target sample and control). Two outputs were generated at the end of each PCR cycle: 1) control and target amplification curves and 2) the actual concentration of target DNA of each individual target sample in the form of an excel datasheet. The DNA concentration of the target sample was computed 50 by interpolating the difference in area under the control amplification curve and the area under the target amplification curve for each participant at a specific time point. Bacterial levels were expressed as 16S rRNA gene copies per swab. Plasmids containing 16S rRNA genes from 45 vaginal bacterial species were employed to assess detection thresholds for each bacterium-specific qPCR assay, to generate standard curves for quantitation, and to assess cross-reactivity with non-target species. To meet criteria for assay implementation, each bacterium-specific qPCR was required to detect ≤10 16S rRNA gene copies per reaction of the target species in duplicate reactions and to show no detection when 1,000,000 16S rRNA genes copies from each of 44 non-target vaginal bacterial species were employed as template DNA. Four no-template PCR controls were run with each qPCR assay. No-template controls consisted of all necessary PCR reagents for amplification except template DNA and were used to monitor for bacterial contamination of the PCR reagents. 3.2.5.5.4 Extraction and amplification controls Beta-globin gene PCR was performed on all extracted DNA samples to ensure that the swabs contacted a tissue surface during sampling and to document the presence of amplifiable DNA (Antonio et al., 2003). In addition, amplification control qPCRs using exogenous DNA were performed on all samples to detect PCR inhibitors in a more sensitive and quantitative fashion. The amplification control qPCR assay targeted a segment of the jellyfish aequorin gene using specific primers and a TaqMan probe as 51 previously described (Khot et al., 2008). A synthesized single strand of DNA coding for a portion of the jellyfish aequorin gene was added to PCR master mix at a concentration of 105 copies per reaction. Inhibition in the amplification control PCR was defined as a >2cycle increase in the threshold cycle for the extracted vaginal DNA sample compared to the no-template controls. 3.2.5.6 Sociodemographic and sexual behaviour characteristics The following sociodemographic and sexual behaviour characteristics were measured using a structured questionnaire: age, marital status; employment status, cigarette smoking, vaginal douching habits, lifetime number of sex partners, number of sex partners in the 30 days preceding this study, protected and unprotected sexual intercourse during the study, frequency of sexual intercourse during study, history of bacterial vaginosis, recent antibiotic use, adherence to the metronidazole vaginal gel and the probiotic treatment schedules (LACTIN-V®). 3.3 Data management and analysis Data was entered and managed using the Datafax Clinical Database Management System (version 3.7). The data management unit at DF/Net Research Inc. implemented quality control procedures beginning with the data entry system. All data collected for the study was thoroughly checked for accuracy, completeness, and adherence to the protocol and the study data management standard operating procedures (SOP). 52 Data was then exported to STATA™ version 10 (College Station, Texas, USA) for analysis. The analysis for colonization efficiency for the 18 participants in the LACTINV® treatment arm of the study was performed within the intent-to-treat (ITT) cohort of all participants. Frequency tables and summary statistics were made for the sociodemographic characteristics and frequency of colonization with seven BV-associated bacterial species, L. iners and L. crispatus species. Means, medians and interquartile ranges were calculated based on CTV-05 colonization status at the day 28 visit. The concentrations of bacterial rRNA per swab at screening/enrolment within subjects who colonized with CTV-05 were compared with day 28 levels using the Wilcoxon signedrank test. This test was repeated within subjects who did not colonize. Changes in the concentration of bacterial rRNA gene per swab between screening/enrolment (pretreatment) and day 28 within subjects who later colonized with CTV-05 were compared to those within subjects who did not colonize using the Wilcoxon rank-sum (Mann-Whitney) test. To assess if different species of BV-associated bacteria, L. iners, L. crispatus and sociodemographic and sexual history characteristics predicted subsequent colonization with L. crispatus CTV- 05 at day 28, univariate exact logistic regression was also performed. 3.4 Ethical Considerations All women who participated in this study gave informed written consent as approved by the institutional review boards. All CRF data (questionnaire, laboratory data and clinical examination data) was entered into a database at the study site with only the participant 53 identification number (subject ID). Informed consent forms, contact information, and other data collection instruments containing personal identifiers were stored separately in locked file cabinets in locked offices located at the project site. Only limited study research staff or sponsor representatives had access to these files. This study was part of the Phase 2A LACTIN-V® trial in women diagnosed with BV. It was conducted from April 2008 through January 2009 at the Clinical Translational Science Institute (CTSI) Clinical Research Centre at the University of California San Francisco (UCSF), USA and approved by the UCSF Committee on Human Research at UCSF (#H43476-32139-02A). This study’s protocol was also approved by the Ethical Review Committee of the Kenya Medical Research Institute (KEMRI), Nairobi, Kenya and the Jomo Kenyatta University of Agriculture and Technology (JKUAT). Safety oversight was provided by a Safety Monitor. This trial is registered at www.clinicaltrials.gov (NCT00635622). ...... ....... ....... ........ ........ ........... ................ .......................... ........ 54 CHAPTER FOUR 4.0 RESULTS 4.1 Participants’ socio-demographic and sexual behaviour characteristics The median age of the participants was 30.5 years with a range of 18-44 years (Table 4.1). The number of white participants was equal to that of black or African American participants (39%). Majority of the participants (72%) did not have post-secondary education and only six (33%) had either full-time or part-time employment. Of the 18 women in this study, majority 13 (72%) had a steady sexual partner, but only three (17%) were married. Six participants (33%) were current smokers. Half of the women reported having had their sexual debut at 15 years or below while only four (22%) had their debut at or above 18 years (Table 4.2). The women reported a median of 10 (range 1 – 99) lifetime sexual partners and a median of 1 (range 1 – 3) sexual partner within the last six months. Of the 16 participants reporting having had male sexual partners in the six months preceding this study, four (25%) reported having had two or more partners. Four participants (22%) reported having had female sexual partners in the six months preceding this study. Of the 13 women who reported having had sex in the 30 days preceding this study, four (31%) participants reported having had protected sex (used condoms), six (33%) unprotected sex and three (23%) women had both protected and unprotected sex. 55 Table 4.1: Socio-demographic characteristics of the study participants. Variable Age (years) 18 – 29 30 – 39 > 40 Race Black or African American White Other Highest level of education High school education or below Some college education Graduate degree Marital Status Married Divorced or Separated Single (never married) Steady partner, cohabiting Steady partner, not cohabiting Employment Status Student Employed full-time Employed part-time Unemployed, looking for work Unemployed, not looking for work Smoking Yes No Frequency Percentage 8 8 2 44.4 44.4 11.1 7 7 4 38.9 38.9 22.2 13 3 2 72.2 16.7 11.1 3 1 4 5 5 16.7 5.7 22.2 27.8 27.8 3 5 1 7 2 16.7 27.8 5.6 38.9 11.1 6 12 33.3 66.7 Of the 14 women who had ever been pregnant, 10 (71%) had had at least one live birth, six (Smit et al. 2001) a spontaneous abortion, 11 (79%) an elective abortion and two participants either an ectopic pregnancy or a still birth. Three women (20%) reported having had at least one BV episode when they were 18 years or less. Six women (33%) had experienced more than an ectopic pregnancy or a still birth. Three women (20%) 56 reported having had at least one BV episode when they were 18 years or less. Six women (33%) had experienced more than five BV episodes in their lifetime and 15 women had at least one BV episode in the preceding 12 months before this trial. Fourteen participants (78%) reported having ever douched or used vaginal preparations or drying agents. During the trial, 11 participants (61%) had sexual intercourse. Of those 11 participants who had sexual intercourse during trial, six (33%) had unprotected sex (no condom use). Table 4.2: Baseline sexual risk factors among the study participants Variable Sexual debut ≤15 years 16 – 17 years ≥18 years Lifetime number of sexual partners 1 2 – 10 >10 Male sexual partners in the last 6 months 0 1 ≥2 Female sexual partners in the last 6 months 0 1 ≥2 Sexual intercourse in the last 30 days Had sex Always protected sex Not always protected sex Ever pregnant Yes No Frequency Percentage 57 9 5 4 50.0 27.8 22.2 1 10 7 5.6 55.6 38.9 2 12 4 11.1 66.7 22.2 14 2 2 77.8 11.1 11.1 13 4 9 72.2 69.2 46.2 14 4 77.8 22.2 Pregnancy outcomes Live births Still births Spontaneous abortion Elective abortion Ectopic pregnancy Ever douched Yes No Age at first BV episode ≤18 years >18 years Lifetime (previous) BV episodes 1–5 6 – 10 >10 Previous BV episodes in the last 12 months 0 1 ≥2 Sexual intercourse during trial No Yes, ALWAYS protected (male condom) Yes, NOT ALWAYS protected (no male condom) Douched during trial Yes (fingers x2, tampon x1) No Menses during trial Yes No 10 1 6 11 1 55.5 5.5 33.3 61.1 5.5 14 4 66.7 33.3 3 12 20 80 11 3 3 64.7 17.6 17.6 1 9 6 6.3 56.3 37.5 7 5 6 38.9 27.8 33.3 3 15 16.7 83.3 13 5 72.2 17.8 4.2 L. crispatus CTV-05 vaginal colonization DNA fingerprints of were prepared for every sample that was found to be positive for L. crispatus at screening, enrolment and day 28 for strain identification. L. crispatus CTV-05 showed intense bands at approximately 1,500, 1,650, and 2,400 bp and minor bands appeared at approximately 1,950 bp and at 2,100 bp (Plate 4.1). 58 Overall, the L. crispatus CTV-05 study strain was recovered at either the day 10 (followup) or the day 28 (final) visits in 11 participants (61%) but only eight participants (44%) had sustained colonization up to the day 28 visit. Colonization was not sustained to the day 28 visit in three participants (17%) and seven participants (39%) did not colonize with CTV-05 at any of the two follow-up visits. A L B C D E F G H I J K 5090 4072 3054 2400 2100 1950 2036 1650 1500 1636 1018 517 Plate 4.1: Polaroid picture of one the repPCR DNA fingerprints generated from Lactobacillus crispatus CTV-05 in this study. Lane B and K were markers; Lane A, C and D were negative samples of subject 1499; Lane E, F and G are positive samples of subject 003; Lane H and I are negative samples of Subject 0466; Lane J is a positive control. Tables 4.3 shows the number and proportion of participants colonized with any Lactobacillus species at enrolment (after metronidazole treatment but prior to LACTIN59 V® treatment) and those subsequently colonization with CTV-05 at day 28. Of the six participants who were colonized with any Lactobacillus species at enrolment three (50%) subsequently colonized with CTV-05 compared to five (33%) of the 11 participants not colonized with any Lactobacillus species at enrolment. Table 4.3: Vaginal colonization with any Lactobacillus species at enrolment and subsequent Lactobacillus crispatus CTV-05 colonization at Day 28 L. crispatus CTV-05 colonization at Day 28‡ Any Lactobacillus species at enrolment‡ Positive (%) Negative (%) Total (%) Positive Negative Total 3 (50) 5 (42) 8 (44) 3 (50) 7 (58) 10 (56) 6 (100) 12 (100) 18 (100) Fisher's exact test (p) = 0.563 ‡Colonization was identified using culture and repPCR method Tables 4.4 shows the number and proportion of participants colonized with any Lactobacillus species at the day 28 visit (after both metronidazole and LACTIN-V® treatment) and those colonized with CTV-05 at the same visit. Of the 13 participants who were found to have Lactobacillus species at day 28, eight were found to have colonized with CTV-05. Table 4.4: Vaginal colonization with any Lactobacillus species and Lactobacillus crispatus CTV-05 at Day 28. L. crispatus CTV-05 colonization at Day 28‡ Any Lactobacillus species at day 28‡ Positive (%) Negative (%) Total (%) Positive Negative Total 8 (62) 0 (0) 8 (44) 5 (38) 5 (100) 10 (56) 13 (100) 5 (100) 18 (100) Fisher's exact test (p) = 0.029 ‡Colonization was identified using culture and repPCR method 60 Lactobacillus crispatus CTV-05 was not found in any of the six participants colonized with Lactobacillus species at enrolment. However, 10 days after commencing treatment with LACTIN-V nine participants (50%) where found to have colonized with CTV-05 and eight participants (44%) had CTV-05 colonization at the final day 28 visit (Figure 4.1). No Lactobacillus Lactobacillus species Proportion colonized by Lactobacillus species and CTV-05 N=18 N=18 100 90 L. crispatus CTV-05 N=18 89% 80 72% 70 60 50 40 50% 44% 33% 30 20 10 0 Enrolment Day 10 Day 28 Follow-up visits Figure 4.1: Proportion of participants colonized by Lactobacillus crispatus CTV-05 study strain amongst those colonized by any Lactobacillus species on follow-up (n=18) 61 4.3 BV-associated Bacteria qPCR Amplification Curves Standard (control) and target DNA amplification curves were generated for each participant’s sample and each target BV-associated bacteria at the three follow-up time points. The 16S rRNA concentration of each target BV-associated bacteria per swab was generated by interpolating the difference in area under the control amplification curve and the area under the target amplification curve for each participant at each specific time point. Figure 2.2 shows the G. Vaginalis standard (control) curves and target amplification curves at screening, enrolment and day 28 for subject 1606. The target curves show a higher concentration of G. vaginalis at screening compared to enrolment and day 28. Subject 1606 did not subsequently colonize with L. crispatus CTV-05 at day 28. Figure 2.3 shows standard curves and target amplification curves in the three follow-up time points for L. crispatus for subject 0106 who subsequently colonized with L. crispatus CTV-05 at day 28. The L. crispatus species 16S rRNA concentration was below threshold (undetectable) at screening and enrolment sampling times but there was high vaginal concentration at day 28. 62 (a) (b) Figure 4.2: Gardnerella vaginalis standard curves (a) and target amplification curves at screening, enrolment and day 28 for subject 1606 (b). Curves that appear very early during PCR amplification cycling e.g. the green (screening) curve in (b) indicate high concentration of 16S rRNA of the target organism whilst curves appearing almost at the end of PCR amplification cycling e.g. the orange (day 28) curve in (b) indicate very low 16S rRNA concentration or negative sample depending on the qPCR threshold. 63 (c) (d) Figure 4.3: Lactobacillus crispatus standard curves (c) and target amplification curves at screening, enrolment and day 28 for subject 0106 (d). 64 4.4 Baseline concentrations of BV-Associated Bacteria The baseline median concentrations of BV-associated bacteria and two Lactobacillus species (L. iners and L. crispatus species) grouped according to their L. crispatus CTV-05 colonization status at day 28 are shown in Table 4.5 (at screening) and Table 4.6 (at enrolment). Women who colonized with CTV-05 at day 28 had a lower median concentration of G. vaginalis, Leptotrichia/Sneathia species, A. vaginae, BVAB2, L. iners and L. crispatus species and a higher median concentration of Megasphaera species at screening compared to those who did not colonize. However, the median concentration of BVAB1 and BVAB3 was below detection limits (BDL) at screening in both women who subsequently colonized with CTV-05 and those who did not (Table 4.5). Table 4.5: Pretreatment vaginal concentration of various bacteria† Post-treatment L. Crispatus CTV-05 colonization status (Day 28) Colonizing species at screening Gardnerella vaginalis Leptotrichia/Sneathia spp. Megasphaera spp. Atopobium vaginae BVAB1 BVAB2 BVAB3 Lactobacillus iners Lactobacillus crispatus Positive (n=8) median IQR 7.88 1.11 6.17 4.60 6.73 3.48 7.09 1.34 BLD 2.74 5.62 4.04 BLD 2.40 6.82 3.99 BLD 0.46 Negative (n=10) median IQR 8.10 0.82 7.01 3.53 5.50 5.48 7.45 1.60 BLD 0.30 6.64 3.29 BLD 3.94 6.84 0.41 2.81 1.29 BLD - Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 † Concentration of bacteria in vaginal fluid expressed as log10 16S rRNA gene copies per swab 65 The median concentration of G. Vaginalis at enrolment was higher in those who did not colonize with CTV-05 at day 28 compared to those who did. The median concentrations of A. vaginae, Leptotrichia/Sneathia spp. and L. iners were higher in those who subsequently colonized with CTV-05 compared to those who did not. The concentrations of Megasphaera spp., BVAB1, BVAB2, BVAB3 and L. crispatus species were all below limits of detection at enrolment in both the women who later colonized with CTV-05 and those who did not (Table 4.6). Table 4.6: Pretreatment vaginal concentration of various bacteria just before LACTIN-V® treatment (enrolment visit) † Post-treatment L. Crispatus CTV-05 colonization status (Day 28) Colonizing species at enrolment Gardnerella vaginalis Leptotrichia/Sneathia spp. Megasphaera spp. Atopobium vaginae BVAB1 BVAB2 BVAB3 Lactobacillus iners Lactobacillus crispatus Positive (n=8) median IQR 4.54 2.77 3.21 1.02 BLD 2.34 2.74 1.57 BLD 0.15 BLD 0.00 BLD 0.15 7.75 4.53 BLD 0.39 Negative (n=10) median IQR 5.67 3.55 BLD 0.94 BLD 2.88 2.73 3.20 BLD 0.30 BLD 0.60 BLD 0.30 6.71 1.23 BLD 4.55 BLD - Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 † Concentration of bacteria in vaginal fluid expressed as log10 16S rRNA gene copies per swab The median concentrations of all BV-associated bacteria and L. iners were generally lower or below limits of detection in those who were already colonized with endogenous Lactobacillus species at enrolment compared to those who were not (Table 4.7). The opposite was true for L. crispatus species whose concentration was higher in those who 66 were already colonized with endogenous Lactobacillus species at enrolment compared to those who were not. Table 4.7: Concentrations of bacteria in vaginal fluid just before LACTIN-V® treatment (enrolment visit) grouped by Lactobacillus species colonization status in the same visit Colonizing species at enrolment* G. vaginalis Leptotrichia/Sneathia Megasphaera sp. Atopobium vaginae BVAB1 BVAB2 BVAB3 L. iners L. crispatus Lactobacillus species at enrolment† Positive (6) Negative (n=12) Median IQR Median IQR BLD 2.89 4.98 2.44 BLD 0.00 3.07 0.98 BLD 0.00 BLD 2.81 BLD 0.84 2.75 1.91 BLD 0.00 BLD 0.30 BLD 0.00 BLD 0.30 3.90 0.00 5.19 0.30 6.69 1.65 6.95 2.33 2.97 3.19 BLD 1.62 p-value‡ 0.11 0.22 0.39 0.44 0.33 0.78 0.18 0.43 0.39 *Colonization determined using quantitative PCR (qPCR) and is expressed as log10 16S rRNA gene copies per swab †Colonization determined using culture and repPCR ‡Compares changes in the bacterial rRNA gene levels at enrolment within subjects who were colonized with Lactobacillus species at enrolment to changes within subjects who were not colonized and was obtained using the Wilcoxon rank-sum (Mann-Whitney) test. BLD - Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 4.5 Effects of BV-associated bacteria on L. crispatus CTV-05 colonization After the initial metronidazole treatment, the median vaginal concentrations of all seven BV-associated bacteria declined between screening and enrolment in participants who subsequently colonized with L. crispatus CTV-05. This trend was maintained up to the day 28 visit where levels of 6 species were below limits of detection (either 375 or 750 16S rRNA gene copies per swab) with up to 7-log reductions in median values. However, participants who did not colonize with CTV-05 also experienced an initial decline of the 67 vaginal levels of all BV-associated bacteria between screening and enrolment, but the concentrations of G.vaginalis, Leptotrichia/Sneathia species, A. vaginae and BVAB2 rebounded/resurged between enrolment and day 28. L. iners levels changed little during follow-up in both those who subsequently colonized with CTV-05 and those who did not. The median concentration of L. crispatus species rRNA was below limits of detection at screening in women who later colonized with CTV-05, but rapidly increased to 108 gene copies per swab at day 28. However, participants who did not colonize with CTV-05 had a median concentration of L. crispatus species rRNA of 102.8 at screening which increased only slightly to 104.2 gene copies per swab at day 28. Figure 4.4 shows vaginal concentration of the seven BV-associated bacteria species, L. iners and L. crispatus in two representative study subjects. Subject 0106 colonized with L. crispatus CTV-05 but subject 1606 did not. Subject 0106 had a sustained reduction in vaginal concentrations of all BV associated bacteria between screening and day 28. The same subject had a marked increase in the concentration for of both L. crispatus species and L. iners especially between enrolment and day 28. In contrast, subject 1606 had sustained high vaginal of all BV-associated bacteria between screening and day 28. Her levels of L. iners decreased between enrolment and day 28 and she completely failed to colonize with L. crispatus at any of the 3 follow-up visits. 68 Leptotrichia/Snethia spp. BVAB1 Lactobacillus iners Megasphaera spp. BVAB2 Lactobacillus crispatus 9 10 8 9 7 8 7 6 6 5 5 4 4 3 3 2 1 2 Subject 0106 Subject 1606 1 0 0 Screening Enrolment Day 28 Screening Enrolment FOLLOW-UP VISIT Day 28 Figure 4.4: Variation in the vaginal concentration of various bacterial species with time expressed as log10 16S rRNA gene copies per swab in two selected subjects (Subject 0106 colonized with the probiotic Lactobacillus crispatus CTV-05 but Subject 1606 did not) Table 4.8 shows median values and interquartile ranges for bacterial rRNA gene concentrations at screening (before both metronidazole and L. crispatus CTV-05 treatment) and at day 28 post-treatment in the two outcome groups consisting of those who subsequently colonized with L. crispatus CTV-05 at day 28 and those who did not. At screening, the median values of BV-associated bacteria rRNA concentrations were generally elevated for all participants. As expected, the median concentrations of L. crispatus species rRNA (including the. 69 Log10 bacterial 16S rRNA gene copies per swab Log10 bacterial 16S rRNA gene copies per swab Gardnerella vaginalis Atopobium vaginae BVAB3 Table 4.8: Vaginal concentration of various bacteria at screening and day 28 in women grouped according to their post-treatment Lactobacillus crispatus CTV-05 colonization status. Bacterium* Gardnerella vaginalis Leptotrichia/Sneathia spp. Megasphaera spp. Atopobium spp. BVAB1 BVAB2 BVAB3 Lactobacillus iners Lactobacillus crispatus Colonized with L. crispatus CTV-05 (n=8) Screening Day 28 median IQR median IQR 7.88 6.17 6.73 7.09 BLDφ 5.62 BLDφ 6.82 BLDφ 1.11 4.60 3.48 1.34 2.74 4.04 2.40 3.99 0.46 4.33 BLDπ BLDπ BLDπ BLDφ BLDφ BLDφ 6.78 7.98 3.23 1.81 1.94 2.53 0.15 1.25 0.73 2.22 2.63 Not colonized with L. crispatus CTV-05 (n=10) Screening Day 28 P value† median 0.01 0.15 0.08 0.01 0.16 0.03 0.29 0.94 0.01 8.10 7.01 5.50 7.45 BLDφ 6.64 BLDφ 6.84 2.81 IQR median IQR P value‡ P value§ for colonization vs. non-colonization 0.82 3.53 5.48 1.60 0.30 3.29 3.94 0.41 1.29 7.31 4.48 BLDπ 6.62 BLDφ 4.17 BLDφ 6.50 4.24 1.41 4.93 3.18 3.83 0.30 4.39 3.41 1.78 3.87 0.05 0.26 0.10 0.15 0.94 0.54 0.27 0.96 0.07 0.29 0.79 0.66 0.03 0.22 0.18 0.85 0.98 0.01 *Concentration was determined using quantitative PCR (qPCR) and is expressed as log10 16S rRNA gene copies per swab IQR – Interquartile range BLD - Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 †Compares changes in the bacterial rRNA gene levels between screening and day 28 within subjects who colonized with L. crispatus CTV-05 and was obtained using the Wilcoxon signed-rank test. ‡Compares changes in the bacterial rRNA gene levels between screening and day 28 within subjects who did not colonize with L. crispatus CTV-05 and was obtained using the Wilcoxon signed-rank test. §Compares changes in the bacterial rRNA gene levels within subjects who colonized with L. crispatus CTV-05 to changes within subjects who did not colonize and was obtained using the Wilcoxon rank-sum (Mann-Whitney) test. ₣ Assay detection threshold = 375 16S rRNA gene copies per swab π Assay detection threshold = 375 16S rRNA gene copies per swab φ Assay detection threshold between 375 and 750 16S rRNA gene copies per swab 70 CTV-05 study strain) increased significantly from below limit of detection at screening to 107.8 at day 28 in participants who colonized with L. crispatus CTV-05 (p=0.01). The median concentration of L. crispatus also increased from 102.8 to 104.2 16S rRNA gene copies per swab in participants who did not colonize at day 28 (p=0.07). However, this increase in concentration of L. crispatus rRNA between screening and day 28 was more pronounced in participants who subsequently colonized with L. crispatus CTV-05 compared to those who did not (p=0.01). Additionally, A. vaginae showed a significantly greater change in concentrations between screening and day 28 (p=0.03) among participants who subsequently colonized with CTV-05 (from 107.1 to 102.5 16S rRNA gene copies per swab) compared to participants who did not (from 107.5 to 106.6 16S rRNA gene copies per swab). Table 4.9 shows median values and interquartile ranges for bacterial rRNA gene concentrations at enrolment (following the 5-day course of MetroGel® but prior to CTV-05 treatment) and at day 28 in the two outcome groups consisting of those who colonized with L. crispatus CTV -05 at day 28 and those who did not. Immediately after completing the initial MetroGel® treatment, at enrolment the median levels of BV associated bacteria rRNA and L. iners differed little between those who later colonized with CTV-05 and those who did not. The median vaginal concentration of A. vaginae decreased from 102.7 16S rRNA gene copies per swab at enrolment to below limits of detection at day 28 in those who colonized with CTV-05. However, the concentration of A. vaginae increased from 102.7 gene copies per swab at enrolment to 106.6 at day 28 in those who did not colonize with CTV-05. This difference in changes of A. vaginae concentration between the two groups was statistically significant (p=0.04). As 71 Table 4.9: Vaginal concentration of various bacteria at enrollment and day 28 in women grouped according to their post-treatment Lactobacillus crispatus CTV-05 colonization status. Colonized with L. crispatus CTV-05 (n=7) Enrollment Day 28 Bacterium* Gardnerella vaginalis Leptotrichia/Sneathia spp. Megasphaera spp. Atopobium spp. BVAB1 BVAB2 BVAB3 Lactobacillus iners Lactobacillus crispatus median IQR median 4.54 3.21 BLDπ 2.74 BLDφ BLDφ BLDφ 7.75 BLDφ 2.77 1.02 2.34 1.57 0.15 0.00 0.15 4.53 0.39 4.33 BLDπ BLDπ BLDπ BLDφ BLDφ BLDφ 6.78 7.98 IQR P value† 3.23 1.81 1.94 2.53 0.15 1.25 0.73 2.22 2.63 0.55 0.34 0.99 0.34 ... 0.91 0.09 0.93 0.02 Not colonized with L. crispatus CTV-05 (n=10) Enrollment Day 28 median IQR median IQR P value‡ P value§ for colonization vs. non-colonization 5.67 BLDπ BLDπ 2.73 BLDφ BLDφ BLDφ 6.71 BLDφ 3.55 0.94 2.88 3.20 0.30 0.60 0.30 1.23 4.55 7.31 4.48 BLDπ 6.62 BLDφ 4.17 BLDφ 6.50 4.24 1.41 4.93 3.18 3.83 0.30 4.39 3.41 1.78 3.87 0.13 0.12 0.35 0.02 0.32 0.22 0.05 0.96 0.61 0.19 0.06 0.60 0.04 0.37 0.40 0.48 0.99 0.003 *Concentration was determined using quantitative PCR (qPCR) and is expressed as log10 16S rRNA gene copies per swab IQR – Interquartile range BLD – Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 † Compares changes in the bacterial rRNA gene levels between enrollment and day 28 within subjects who colonized with L. crispatus CTV-05 and was obtained using the Wilcoxon signed-rank test. ‡ Compares changes in the bacterial rRNA gene levels between enrollment and day 28 within subjects who did not colonize with L. crispatus CTV-05 and was obtained using the Wilcoxon signed-rank test. § Compares changes in the bacterial rRNA gene levels within subjects who colonized with L. crispatus CTV-05 to changes within subjects who did not colonize and was obtained using the Wilcoxon rank-sum (Mann-Whitney) test. π Assay detection threshold = 375 16S rRNA gene copies per swab φ Assay detection threshold between 375 and 750 16S rRNA gene copies per swab 72 expected, the median concentration of L. crispatus species rRNA (all strains including the CTV-05 study strain) increased from below limits of detection at enrolment to 108 at day 28 in participants who subsequently colonized with L. crispatus CTV-05 (p=0.02). There was a less pronounced increase in the median concentration of L. crispatus from below limit of detection to 104 16S rRNA gene copies per swab in participants who did not colonize with CTV-05 at day 28 (p=0.6). When comparing the change in concentration of L. crispatus rRNA between enrolment and day 28, the increase in CTV-05 levels was significantly greater in participants who colonized with CTV-05 compared to those who did not, p=0.003)........ ............................................................................................... 4.6 Factors associated with L. crispatus CTV-05 vaginal non-colonization Univariate analysis using exact logistic regression showed an inverse association between vaginal colonization with any of the 7 species of BV-associated bacteria at screening and subsequent colonization with L. crispatus CTV- 05 at day 28 (Table 4.10). However, these associations were not statistically significant. Vaginal colonization with L. iners or L. crispatus at screening was also inversely associated with L. crispatus CTV-05 colonization, though these associations were also not statistically significant. Sexual intercourse during trial was significantly inversely associated with colonization with CTV-05 whether or not the sex was protected (OR 0.02; 95% CI 0.001 - 0.32). CTV05 colonization was not significantly influenced by having menses in the course of the clinical trial (OR 2.250; 95% CI 0.11 – 7.7). Black ethnicity for participants was inversely 73 Table 4.10: Un-adjusted risks of colonization with Lactobacillus crispatus CTV-05 at the Day 28 visit (using exact logistic regression) Risk factors for CTV-05 colonization L. crispatus CTV-05 colonization at Day 28β Positive Negative n (%) n (%) Odds Ratio (OR) 95% CI pvalue Colonizing species at screening (nα) n=8 n=10 Gardnerella vaginalis (n=17) Leptotrichia/Sneathia spp.(n=13) Megasphaera spp. (n=12) Atopobium vaginae (n=16) BVAB1 (n=4) BVAB2 (n=13) BVAB3 (n=8) Lactobacillus iners (n=15) Lactobacillus crispatus (n=7) 7 (88) 5 (63) 5 (63) 7 (88) 2 (25) 5 (63) 3 (38) 6 (75) 3 (38) 10 (100) 8 (80) 7 (70) 9 (90) 2 (20) 8 (80) 5 (50) 9 (90) 4 (40) 0.80 0.42 0.71 0.78 1.33 0.42 0.60 0.33 0.90 0.0 – 31 0.05 – 3.4 0.07 – 7.9 0.04 – 15 0.14 – 12 0.05 – 3.4 0.09 – 4.0 0.02 – 4.5 0.13 – 6.1 0.89 0.41 0.74 0.86 0.80 0.42 0.59 0.41 0.91 6 (75) 2 (25) 0 (0) 2 (25) 6 (75) 1 (10) 9 (90) 5 (50) 4 (40) 7 (70) 1.00 (referent) 0.05 0.06 0.08 1.28 ... 0.001 – 0.68 0.0 – 0.59 0.006 – 1.2 0.16 – 10 ... 0.018 0.015 0.07 0.95 4 (50) 3 (38) 1 (12) 4 (40) 4 (40) 2 (20) 1.00 (referent) 0.75 0.50 ... 0.10 – 5.8 0.03 – 8.0 … 0.78 0.62 4 (50) 4 (50) 4 (40) 6 (60) 1.00 (referent) 0.66 … 0.10 – 4.4 … 0.67 Sociodemographic factors Sexual intercourse during trial No Yes Always protected Not always protected Menses during trial Race: White Black Others Age: < 30 years ≥ 30 years nα = number of participants with concentrations of specific bacterial 16S rRNA gene copies per swab above the detection thresholds shown in Table 3.1 β Colonization determined using culture and repPCR (BV-associated bacteria species, L. iners and L. crispatus were all determined using qPCR) associated with CTV-05 colonization (OR 0.4; 95% CI 0.05 – 3.4) compared to white ethnicity. We found no statistically significant difference in CTV-05 colonization when 74 we compared colonization in women aged more than thirty (≥30) years with those less than thirty (<30) years (OR 1.1; 95% CI 0.16 – 7.5). Univariate analysis (exact logistic regression) results using presence of BV-associated bacteria at enrolment as a predictor of subsequent L. crispatus CTV-05 colonization at day 28, showed an inverse association between these BV-associated bacteria colonization with L. crispatus CTV-05. However, these associations were not statistically significant. Vaginal colonization with L. iners or L. crispatus at enrolment was also inversely associated with L. crispatus CTV-05 colonization, though these associations were also not statistically significant. Colonization with Lactobacillus species at enrolment, however, seemed to be associated with CT-05 colonization (OR 2; 95% CI 0.27–14.8) though this association was not statistically significant. 4.7 Correlation between post-treatment vaginal levels of L. crispatus species and pretreatment levels of BV-associated bacteria Table 4.11 shows the Pierson correlation coefficients for the correlation between screening (pretreatment) levels of the 7 BV-associated bacterial species, L. iners and L. crispatus species (all strains including the CVT-05 strain), and their subsequent concentrations at day 28 (post-treatment). All the BV-associated bacteria had positive correlation coefficients with each other except for BVAB1 which had negative correlations with most other BV-associated bacteria. The vaginal concentration of L. crispatus species at day 28 had a negative correlation with all the BV associated bacteria, 75 Table 4.11: Pearson correlation coefficients of bacteria vaginal concentrations at the screening (pretreatment) visit correlated with bacterial concentrations at the day 28 visit (post-treatment) Bacteria at screening (pretreatment) Gardnerella vaginalis Leptotrichia/Sneathia spp. Megasphaera spp. Atopobium vaginae BVAB1 BVAB2 BVAB3 Lactobacillus iners Lactobacillus crispatus L. crispatus -0.34 -0.41* -0.22 -0.38 -0.17 -0.32 -0.49** -0.42* 0.16 G. vaginalis 0.54** 0.72*** 0.22 0.64** 0.10 0.60** 0.38 0.35 -0.28 Bacteria at Day 28 (post-treatment) Leptotrichia/ Megasphaera Atopobium BVAB1 Sneathia spp. spp. vaginae 0.23 0.24 0.42* -0.09 0.57** 0.24 0.54** -0.05 0.09 0.35 0.06 -0.40 0.36 0.33 0.51** -0.13 0.31 0.41* 0.11 0.13 0.56 0.22 0.54** -0.10 0.73*** 0.51** 0.47** -0.19 0.28 0.18 0.24 0.05 -0.16 -0.32 -0.20 -0.13 *p value ≤ 0.10 ** p value ≤ 0.05 *** p value ≤ 0.001 76 BVAB2 BVAB3 0.20 0.53** -0.03 0.32 0.29 0.52** 0.68** 0.27 -0.14 0.11 0.44* 0.06 0.26 0.22 0.42* 0.70** 0.26 -0.35 Lactobacillus iners 0.59** 0.30 0.20 0.31 0.14 0.33 0.15 0.41* 0.10 though these correlation was not statistically significant except that with BVAB3 (p=0.04). Post-treatment levels of L. crispatus species also had a negative correlation with L. iners (p=0.08). Table 4.12 shows the Pierson correlation coefficients for the correlation between enrolment levels of L. crispatus species (all strains including the CVT-05 strain), L. iners and the 7 BV-associated bacterial species and their subsequent concentrations at day 28. The vaginal concentration of L. crispatus species at day 28 had a negative correlation with concentrations of all the BV associated bacteria at enrolment except BVAB3. All the BVassociated bacteria had positive correlation coefficients with each other except for BVAB3 which had negative correlations with all other BV-associated bacteria except Megasphaera spp. However, these associations were not statistically significant in most instances. Post-treatment levels of L. crispatus species also had a negative correlation with L. iners (p=0.08). To determine whether the correlation coefficient was a reasonable numerical summary of the association between the post-treatment levels of L. crispatus species and the levels of other BV-associated bacteria at enrolment, a scatter plot smoother (Lowess smoother plot) was constructed. The Lowess smoother plot was included in each of the scatter plots to assess linearity in the scatter plots. For example, the enrolment concentrations of both G.vaginalis (p=0.35) and L. iners (p=0.08) were shown to be inversely correlated with that of L. crispatus species at day 28. Figure 4.5 and Figure 4.6 show the representative Lowess smoother plots for these two correlations. 77 Table 4.12: Pearson correlation coefficients of bacteria vaginal concentrations at the enrolment visit correlated with bacterial concentrations at the day 28 visit (post-treatment) Bacteria at Day 28 (post-treatment) Leptotrichia/ Megasphaera Atopobium Lactobacillus Bacteria at L. crispatus G. vaginalis BVAB1 BVAB2 BVAB3 Sneathia spp. spp. vaginae iners Enrolment (pretreatment) Gardnerella vaginalis -0.27 0.62** 0.38 0.36 0.59** 0.20 0.40 0.32 0.38 Leptotrichia/Sneathia spp. -0.39 0.40 0.44* 0.64** 0.32 0.40 0.43* 0.40 0.15 Megasphaera spp. -0.32 0.41* 0.31 0.62** 0.33 0.22 0.29 0.26 0.15 Atopobium vaginae -0.34 0.42* 0.32 0.58** 0.39 0.29 0.34 0.27 0.08 BVAB1 -0.33 0.21 0.18 0.15 0.21 0.99*** 0.28 0.43* -0.04 BVAB2 -0.35 0.40 0.42* 0.55** 0.34 0.26 0.41* 0.34 0.22 BVAB3 0.09 -0.15 -0.05 0.18 -0.17 -0.40* -0.13 -0.27 -0.18 Lactobacillus iners -0.43* 0.23 0.36 0.23 0.15 0.10 0.33 0.26 0.66** Lactobacillus crispatus -0.01 -0.17 0.04 -0.01 -0.08 -0.15 0.05 -0.16 0.29 *p value ≤ 0.10 ** p value ≤ 0.05 *** p value ≤ 0.001 78 2 4 6 8 10 . 2 4 6 log G. vaginalis at enrolment 8 10 bandwidth = .8 Figure 4.5: A scatter plot of post-treatment Lactobacillus crispatus species concentration in vaginal fluid expressed as log10 16S rRNA gene copies per swab versus that of Gardnerella vaginalis with a Lowess smoother plot. 4.8 The effects of the concentration of BV-associated bacteria on BV persistence (Nugent score ≥7) Table 4.13 shows median values and interquartile ranges for bacterial rRNA gene concentrations at screening (before both metronidazole and L. crispatus CTV-05 treatment) and at day 28 post-treatment in two outcome groups consisting of those who had a Nugent score ≥7 (indication persistent BV) and those with a Nugent score of <7 at day 28. As expected, the reduction in median values of BV-associated bacteria rRNA 79 vaginal concentration were more pronounced in participants who had a Nugent score of <7 at day 28 compared to those whose Nugent score was ≥7. 2 4 6 8 10 . 2 4 6 log L. iners at enrolment 8 bandwidth = .8 Figure 4.6: A scatter plot of post-treatment Lactobacillus crispatus species concentration in vaginal fluid expressed as log10 16S rRNA gene copies per swab versus that of Lactobacillus iners with a Lowess smoother plot. G. vaginalis showed a significantly greater change in concentrations between screening and day 28 among participants with a Nugent score of <7 at day 28 (from 107.4 to 103 16S rRNA gene copies per swab) compared to participants Nugent score was ≥7 (from 108.2 to 107.5 16S rRNA gene copies per swab) (p=0.003). A similar trend was observed with A. 80 vaginae (p=0.026). As expected, the median concentrations of L. crispatus species rRNA (including the CTV-05 study strain) increased significantly more in participants with a Nugent score of <7 at day 28 (from 102.8 gene copies per swab at screening to 107.8 at day 28) compared to participants with a Nugent score of ≥7 (102.6 to 103.6 16S rRNA gene copies per swab) (p=0.001) (Table 4.13). 81 Table 4.13: Concentration of bacteria in vaginal fluid at screening and 28 days after LACTIN-V treatment in women with Nugent score <7 and those with Nugent score ≥7 Nugent score ≥7 (n=10) Screening Day 28 Nugent score <7 (n=8) Screening Day 28 P value‡ for colonization vs. median IQR* median IQR* P value† median IQR* median IQR* P value† non-colonization Gardnerella vaginalis 7.38 1.24 3.09 2.05 0.014 8.17 0.44 7.45 1.36 0.075 0.006 ₣ Leptotrichia/Sneathia spp. 3.27 4.27 BLD 0.00 0.049 7.10 1.75 6.09 4.82 0.610 0.369 Megasphaera spp. 6.07 4.75 BLD₣ 0.00 0.056 6.51 2.95 5.35 3.63 0.153 0.247 ₣ Atopobium vaginae 6.63 2.85 BLD 0.25 0.019 7.83 0.54 6.94 1.47 0.093 0.026 φ φ φ φ BVAB1 BLD 0.30 BLD 0.30 0.317 BLD 2.29 BLD 0.30 0.482 0.951 BVAB2 2.98 3.46 BLDφ 0.00 0.049 6.70 1.37 5.90 4.39 0.307 0.281 φ BVAB3 2.57 0.48 BLD 0.30 0.461 3.67 3.64 4.07 3.41 0.185 0.569 Lactobacillus iners 6.70 3.94 6.78 2.12 0.726 6.84 0.78 6.50 1.95 0.721 0.722 Lactobacillus crispatus 2.81 1.31 7.98 1.70 0.010 2.57 0.57 3.63 2.23 0.041 0.001 Bacterium* *Concentration was determined using quantitative PCR (qPCR) and is expressed as log10 16S rRNA gene copies per swab IQR – Interquartile range BLD – Below Limit of Detection (assay detection thresholds) for each bacterium species as shown in Table 3.1 † Compares changes in the bacterial rRNA gene levels between screening and day 28 within and was obtained using the Wilcoxon signed-rank test. ‡ Compares changes in the bacterial rRNA gene levels between screening and day 28 within subjects who had a Nugent score of <7 at day 28 to changes within subjects who had a Nugent score ≥7 at day 28 and was obtained using the Wilcoxon rank-sum (Mann-Whitney) test. § Compares changes in the bacterial rRNA gene levels within subjects from screening to day 28 time points π Assay detection threshold = 375 16S rRNA gene copies per swab φ Assay detection threshold between 375 and 750 16S rRNA gene copies per swab 82 CHAPTER FIVE 5.0 DISCUSSION, CONCLUSION AND RECOMMENDATIONS 5.1 Discussion 5.1.1 Proportion of Women Successfully Colonized with L. crispatus CTV-05 Despite BV being a rather common vaginal disorder in women in their reproductive age and being associated with adverse outcomes including HIV, the pathophysiological process underlying this disorder is still poorly understood. Probably for this reason, the treatment of BV with antibiotics has been reported to be at best unsatisfactory and BV recurrences of 40% to 60% have been reported at six months of follow up. Several trials have been done in the last decade to try and recolonize the vagina with exogenous Lactobacillus species but although the cure rates reported are satisfactory, there are still large numbers of BV-positive women who do not colonize with the exogenous lactobacilli in these studies. At the time this study was carried out there was no other published study done find out the reasons for non-colonization in these women. In this study vaginal colonization with L. crispatus CTV-05 was achieved in 61% of women at either day 10 or the day 28 follow-up visits in the intention to treat (ITT) cohort. Forty four percent of the participants were colonized by CTV-05 at day 28. It is difficult to compare the CTV-05 vaginal colonization findings from the current study with what has been reported in a majority of other studies using probiotics for the treatment of BV 83 because of differences in the species and strain of Lactobacillus used in the various studies (Table 2.4). Moreover, a different mode of delivery to the vagina (prefilled applicator as opposed to the use of gelatin vaginal capsules) was used. Although Antonio et al. (2009) used L. crispatus CTV-05 to assess safety and vaginal colonization, their study was done in 90 young women without BV using two potencies of gelatin vaginal capsules containing CTV-05. They reported that 69% of the participants were colonized with CTV05 at one follow-up visit or more and that 59% of their participants were positive for CTV-05 at day 28 which is similar to findings in this study. The current study was also different from other studies with regard to diagnostic criteria since participants had to fulfil both the Amsel and Nugent criteria to qualify for enrolment. Most studies use either one of the diagnostic criteria but rarely both. The main outcome in this study was the proportion of women colonized with L. crispatus CTV-05 as opposed to the Nugent score, clinical cure or BV resolution which have been reported as outcomes in previous lactobacilli probiotic studies. Mastromarino et al. (2009) reported treatment success (Nugent score <7) in 61% of Lactobacillus-treated patients compared to 19% of placebo-treated patients after 21 days of follow-up. Although they reported a Lactobacillus colonization rate of 74% at day 21, the probiotic they used was not named and the colonizing Lactobacillus species was not identified. Martinez et al. (2009) using 2 vaginal capsules of L. rhamnosus GR-1 and L. reuteri RC-14 every morning for 4 weeks following a single 2g dose of tinidazole reported a cure rate of 87.5% in the Lactobacillus group compared to 50% in the placebo group. However, these trials 84 did not study the possible reasons why up to 50% of the Lactobacillus treated women failed to colonize with the Lactobacillus or why their BV did not resolve. 5.1.2 Effects of BV-Associated Bacteria on Vaginal Colonization with L. crispatus CTV-05 This study sought to determine whether the vaginal presence and quantities of BVassociated bacterial species, as reflected by bacterial DNA concentrations in vaginal swabs before/after antibiotic and probiotic treatment, are associated with L. crispatus CTV-05 colonization. The study was based on the hypothesis that persistently high concentrations of BV-associated bacteria prior to treatment with the probiotic would prevent colonization with the exogenous Lactobacillus. The results revealed that women who colonized with L. crispatus CTV-05 at day 28 generally had lower median concentrations of various BV-associated bacteria at screening compared to those who did not colonize (Table 4.5). Additionally, there was a sustained reduction of vaginal levels of the seven BV-associated bacteria in women who subsequently colonized with CTV-05 which was maintained up to the day 28 visit. However, participants who did not colonize with CTV-05 also experienced an initial decline of the vaginal levels of all BV-associated bacteria between screening and enrolment (after MetroGel® treatment), but the concentrations of G. vaginalis, Leptotrichia/ Sneathia species, A. vaginae and BVAB2 rebounded between enrolment and day 28 (Table 4.8, Table 4.9 and Figure 4.4). 85 The qPCR data on the vaginal concentrations of fastidious BV-associated bacteria prior to LACTIN-V® treatment suggest that there is an association between these levels and subsequent colonization of the vagina with exogenous L. crispatus CTV-05. Although there is an association between the presence of specific BV-associated bacteria and the likelihood of vaginal colonization with CTV-05 the data from this study suggests that the vaginal concentration of these pathogenic bacteria may be a more important influence on CTV-05 colonization than their presence in the vaginal vault. This probably explains why G. vaginalis has been isolated in up to 50% of women without BV using cultivation methods and 70% of women without BV using PCR methods (Srinivasan and Fredricks, 2008). It is possible that a high vaginal concentration of G. vaginalis and other BVassociated bacteria as opposed to their mare presence in the vaginal vault is what causes BV. In this study, there was an increase in the median concentrations of some of BVassociated bacteria between enrolment and day 28 in women who did not colonize with CTV-05. In contrast, the median concentrations of these bacteria remained the same or reduce in women who colonized with CTV-05 in the same period of follow up. For example, the median concentration of G.vaginalis in those who colonized with CTV-05 was 104.5 and 104.3 16S rRNA gene copies per swab at the enrolment and day 28, respectively compared to 105.8 and 107.3 at the enrolment and day 28, respectively, in those who did not colonize. A similar trend was also observed with Leptotrichia/Sneathia species, Atopobium vaginae and BVAB2 suggesting an association between the vaginal concentration of these BV-associated species and CTV-05 colonization. 86 Tosun et al. (2007) reported that 70% and 53% of G. vaginalis isolates were resistant to metronidazole and clindamycin, respectively. G. vaginalis and A. vaginae have been shown to colonize the vaginal epithelium as bio-films, which most likely play a role in their vaginal re-colonization and relapse of BV. Patterson et al. (2007) developed an in vitro model for G. vaginalis bio-film and demonstrated that it tolerated 5-fold and 4-8 fold higher concentrations of H2O2 and lactic acid, respectively, than planktonic cultures. Proteolytic dissolution of biofilms reduced sensitivity to H2O2 and lactic acid. Patterson et al. (2010) went further and designed a viable assay to analyse bacterial adherence to vaginal epithelial cells, to compare biofilm-producing capacities and to assess cytotoxic activity. They reported that, of the BV-associated anaerobes they tested; only G. vaginalis demonstrated all three virulence properties combined. Their study suggests that G. vaginalis is more virulent than other BV-associated anaerobes, and that many of the bacterial species frequently isolated from BV may be relatively avirulent opportunists that colonize the vagina after G. vaginalis has initiated an infection. It is possible that G. vaginalis forms the initial biofilms which then enable other BV-associated bacteria to adhere and colonize the vagina. Swidsinski et al. (2008) followed-up 18 patients with bacterial vaginosis after a 7-day treatment with oral metronidazole and reported consistently observing the resurgence of a dense and active bacterial bio-film on the vaginal mucosa, primarily consisting of Gardnerella vaginalis and Atopobium vaginae with treatment cessation even though all patients recovered. It is possible that these metronidazole resistant biofilms were present 87 in those of our study participants whose concentration of specific BV-associated bacteria remained high after metronidazole treatment. Further, it is also possible that these biofilms adhered strongly to the vaginal epithelial cells thus preventing the exogenous L. crispatus CTV-05 from adhering to the epithelial cells. It is thus crucial for future probiotic studies to try and use a higher dose of oral and intravaginal antibiotics and for a longer period of time (e.g. 2 weeks) to try and break down these bio-films before probiotic treatment. Probiotic treatment should also be carried out for a longer period of time to ensure vaginal colonization with high numbers of H2O2 and lactic acid producing Lactobacilli. As expected, there was a higher concentration of BV-associated bacteria DNA and a lower concentration of L. crispatus species DNA in participants with a Nugent score ≥7 compared to those with a score of <7. However, contrary to expectations we found no difference in the concentration of L. iners by Nugent score. 5.1.3 Effects of other Vaginal Lactobacilli on Colonization with L. crispatus CTV-05 Gram stains of vaginal fluid smears from women without BV typically show Grampositive rods, with cultures revealing a predominance of Lactobacillus species. However, studies have revealed geographic and racial variations regarding the predominant Lactobacillus species colonizing the vagina in both normal and BV-positive women. Studies in China (Yan et al., 2009), Japan (Song et al., 1999a), Europe (Kiss et al., 2007; 88 Vasquez et al., 2002) and USA (Antonio et al., 2005a) have reported the predominance of L. crispatus in normal women including pregnant women. L. jensenii, L. gasseri and L. acidophilus are additional species reported to be predominant in normal women. In contrast, Anukam et al. (2006c) reported that L. iners is the most abundant vaginal lactobacillus species in healthy premenopausal Nigerian women. However, more than half of the participants in that study had either BV (14.2%) or an intermediate score (51%) on the Nugent criteria. Matu et al. (2009) reported a higher diversity of lactobacilli in normal Kenyan women compared to BV-positive women. They found L. Jensenii as the predominant species in both groups but also isolated L. iners in 15% of women without BV and 12% of women with BV. Fredricks et al. (2005a) using a combination of broadrange PCR assays of 16S rDNA and fluorescence in situ hybridization (FISH) performed directly on vaginal fluid, found L. crispatus to be the predominant species in BV-negative women and L. iners to be the predominant Lactobacillus species in BV-positive women. This study also found high levels of L. iners in women who were BV-negative at day 28 as well as those who were BV positive (Table 4.13). There were also high pretreatment levels of L. iners at screening in both the women who subsequently colonize with exogenous L. crispatus CTV-05 and those who did not (Table 4.8). The level of L. iners at enrolment was higher in women who subsequently colonize with exogenous L. crispatus CTV-05 compared to those who did not (Table 4.9). This difference was not statistically significant. These results suggest that L. iners may not be intimately associated with the pathogenesis of BV. It may also suggest that L. iners is more resistant to replacement by 89 BV-associated bacteria and may in fact, replace other lactobacilli in BV and probably also inhibit attempts by exogenous L. crispatus to re-colonize the vagina. It may also be a transient Lactobacillus which is replaced by the higher H2O2 producing species once the healing process and return to normal flora begins. A high vaginal level of endogenous L. crispatus was found to be significantly associated with a reduced chance of colonization with the exogenous probiotic L. crispatus CTV-05. The findings in this regard were similar to those of Antonio et al. (2009) who studied L. crispatus CTV-05 colonization in women without BV. Ninety percent of the women they enrolled in that study were colonized by lactobacilli (76% by H2O2 producing strains) before using the probiotic L. crispatus CTV-05. It is also likely that these women had higher pretreatment vaginal concentrations of L. crispatus than women with BV such as we enrolled in the present study. They reported increased CTV-05 non-colonization (OR 34; 95% CI 4 – 286) if one was already colonized with L. crispatus before probiotic treatment. It is important to note here that in this study, there was an increase in the concentration of L. crispatus species in both those who were culture and rep-PCR positive for L. crispatus CTV-05 and those who were not. 15 out of 18 participants (83%) had qPCR detectable L. crispatus species at day 28 and only 8 (53%) these participants had L. crispatus CTV-05 strain detectable by culture and rep-PCR. This could be interpreted as a failure to detect CTV-05 using culture and rep-PCR in some participants when it was indeed present or the colonization with other strains of L. crispatus after metronidazole 90 treatment which could have precluded successful colonization by the exogenous strain of L. crispatus. 5.1.4 Effects of Socio-demographic and Sexual Factors on Colonization with L. crispatus CTV-05 A lot of information is available regarding the effects of socio-demographic and sexual factors on BV acquisition. However, little information is available on the effect of these factors on vaginal colonization with exogenous Lactobacillus probiotics. In 250 adolescents with a history of douching, sex within the past week was associated with increased likelihood of BV in multivariate analysis (Schwebke et al., 2004). Schellenberg et al. (2008) found that longer self-reported time since last sexual intercourse was independently associated with increased counts of bacterial cell-units (BCU) per gram of vaginal fluid. A high BCU was associated with normal Hay–Ison score suggesting the presence and higher quantities of Lactobacillus in these women with longer periods of sexual abstinence. In this study, we found that vaginal intercourse anytime from the start of LACTIN-V® treatment to the day 28 final visit significantly decreased the likelihood of successful CTV-05 colonization (Table 4.10). A similar observation was previously also reported by (Antonio, et al., 2009). The high pH of seminal fluid or one of its components may affect the adherence of CTV-05 to vaginal epithelial cells and/or its survival in the vaginal vault. Toth et al. (1982) reported that spermatozoa may provide a vehicle for bacteria present in the seminal fluid prior to ejaculation and for those already present in 91 the cervix or vagina. It is thus possible that CTV-05 is adherent to and is transferred by sperm deeper into or out of the vaginal vault reducing the likelihood of colonization. Protected sexual intercourse using condoms was found to also significantly reduce the likelihood of CTV-05 colonization. The concentration of BV-associated bacteria was found to be higher in women who reported having had sex during trial compared to those who did not. It is possible that these BV-associated bacteria were re-introduced into the vaginal vault through sexual intercourse from the partners of women who had unprotected intercourse and from the skin of the external genital area for those who had protected intercourse. This could also partly be due to the reason that sexual partners of women enrolled in the study were not offered antibiotic treatment. Although participants enrolled in this study were advised against douching and indeed none of them reported doing it during the trial, sexual intercourse is associated with douching or vaginal washing and it is possible that this may contribute to the low probiotic colonization in women who have intercourse during trial. Annang et al. (2006) identified commonly reported reasons for douching as: “to cleanse after menses” and “to feel fresh”. They also reported an association between frequent douching and douching after sex. Schwebke et al. (2004) found a five-fold increase of BV in women who reported douching after menses compared with those who did not. However, due to the cross sectional design of their study they were not able to describe the independent effects of sexual activity on the vaginal flora separate from that of douching. 92 5.1.5 Limitations The study had several limitations. First, the sample size was small having been drawn from the treatment arm of a Phase IIA trial which was designed to investigate safety and efficacy of LACTIN-V® (L. crispatus CTV-05). However, the study still showed significant association between the vaginal concentration of some BV-associated bacteria DNA and the likelihood of colonization with L. crispatus CTV-05. Second, vaginal fluid culture and repPCR were used in this study to identify the CTV-05 probiotic strain. It is possible that CTV-05 was not detected in some participants using culture and rep-PCR when it was indeed present. Fifteen out of 18 participants (83%) had qPCR detectable L. crispatus species at day 28 but only 8 of them (53%) were found positive for L. crispatus CTV-05 using culture and repPCR. During early follow-up visits CTV-05 may be present in low quantities undetectable by culture and repPCR. Third, qPCR assays were performed targeting six fastidious bacteria recently associated with BV using molecular methods (Leptotrichia/Sneathia species, Megasphaera species, Atopobium vaginae, BVAB1, BVAB2 and BVAB3) and one cultivated bacteria that has been associated with BV (G. vaginalis). Quantitative PCR assays were also done for two cultivated bacteria that have been associated with vaginal health (L. crispatus and L. iners). There are many additional vaginal bacteria that could be assayed using the qPCR platform and which may play a role in the pathogenesis of BV and possibly too affect vaginal colonization with exogenous lactobacilli. Future research should seek to measure 93 how these bacterial concentrations correlate with disease status and how they influence colonization with endogenous and exogenous lactobacilli. Fourth, our detection threshold for each assay was 375 to 750 copies per swab. The use of a larger fraction of vaginal fluid DNA for each assay would reduce the detection thresholds but would also compromise one’s ability to run multiple assays. Finally, the results of this study may not be generalizable to women treated for BV with oral antibiotics or to women not initially treated with metronidazole vaginal gel before probiotic treatment. In addition, all the women enrolled in this study had recurrent BV, 6 of them with more than 2 episodes of BV in the one year preceding this trial. One of the strengths of this study is the extensive use of PCR controls to monitor for falsepositive and false-negative results, thereby increasing the reliability of the bacterial qPCR data reported. 5.2 Conclusion L. crispatus CTV-05 effectively colonized the vagina in 61% of the women enrolled in the study. Forty four percent of the women retained this colonization at the final day 28 follow-up visit. These findings are similar that reported in other previous studies using either LACTIN-V or other Lactobacillus probiotics vaginally or orally. 94 The median vaginal concentrations of bacterial DNA from 7 BV-associated bacteria species including several fastidious species at screening (before metronidazole treatment) and enrolment (before L. crispatus CTV-05 treatment) seemed to affect colonization by CTV-05 at the final day 28 visit. The decrease of BV-associated bacterial species between screening and day 28 follow-up visit was more marked in those who subsequently colonized with CTV-05 compared to those who did not. Although there was little change in the median concentration of G. vaginalis and Atopobium vaginae between enrolment and day 28 in the women who colonized with CTV-05, there was an increase in the median concentration of the two species in women who did not colonize with CTV-05, significantly so with Atopobium vaginae. The median concentration of Leptotrichia/Sneathia species between enrolment and day 28 decreased in the women who colonized with CTV-05, but increased in women who did not colonize with CTV-05. This difference was statistically significant. The null hypothesis was thus rejected for two of the BV-associated bacteria (Atopobium vaginae and Leptotrichia/Sneathia) but could not be rejected in others. The median concentrations of L. iners DNA did not significantly change with time or with CTV-05 colonization status. In contrast, the median concentrations of L. crispatus DNA significantly increased between enrolment and day 28 in both the women who colonized with CTV-05 and those who did not. However, those who colonized with CTV-05 had a significantly higher increase in the median concentration of L. crispatus species DNA 95 compared to those who did not. The null hypothesis was thus rejected for L. crispatus species effect on CTV colonization but could not be rejected for L. iners. Among the sociodemographic factors sex during trial whether protected or unprotected has a negative effect on L. crispatus CTV-05 colonization. Other factors including age, race and menstruation during trial were found to be insignificantly associated with CTV05 vaginal colonization. The null hypothesis was thus rejected for the effects of sex during trial on CTV-05 colonization but could not be rejected for other social demographic characteristics including age, race and menstruation during trial. The results of this study suggest that vaginal colonization status of probiotic L. crispatus CTV-05 correlates with vaginal concentrations of BV-associated bacteria DNA including some fastidious species and provide supporting evidence that these bacteria are important in the pathogenesis of BV and the success of treatment with both antibiotics and probiotics. The study also provides evidence that some vaginal Lactobacillus and sexual intercourse during treatment affect vaginal colonization with exogenous probiotic Lactobacillus. 5.3 Recommendations 1. This study achieved vaginal colonization with L. crispatus CTV-05 in 61% of women with BV at both day 10 and day 28 visit. Higher colonization rates are desirable for 96 optimal effects on BV cure rates. Ways to improve the qualities of the probiotic lactobacilli to enhance their persistence in the vaginal vault need to be sought. 2. BV has a high rate of relapsing or recurrence. Efforts to understand this enigmatic condition will benefit from a greater understanding of the complex bacterial biofilms found in the human vagina and how they change over time. The detection and quantification of all important BV-associated bacteria or consortia by PCR including those not assayed in this study may be useful for monitoring of the response to antibiotic and probiotic therapy. 3. Lengthening the 7-day metronidazole and the 7-day CTV-05 dosing period evaluated in this study may increase colonization by the probiotic strain. Additional studies of the product will be needed to assess whether L. crispatus CTV-05 will be a useful adjunct to antibiotic treatment for the prevention of recurrent BV. 4. Selecting BV-positive participants who are not already colonized with endogenous lactobacilli would enhance the chance of probiotic Lactobacillus colonization. This means that vaginal instillation with Lactobacillus probiotics would be most effective in women with severe BV. 5. Using both Amsel and Nugent diagnostic criteria to define BV in probiotic studies would also lead to a more consistent case definition and increase the comparability of the resulting data. 6. Women on treatment with vaginal Lactobacillus probiotics should be strongly advised against sexual intercourse during the course of treatment with the probiotic. This would enhance they chance of colonizing with the exogenous Lactobacillus. 97 REFERENCES Abad C.L. and Safdar N. (2009). The role of lactobacillus probiotics in the treatment or prevention of urogenital infections--a systematic review. J.Chemother., 21, (3) 243-252 Amsel, R., Totten, P.A., Spiegel, C.A., Chen, K.C., Eschenbach, D. and Holmes, K.K. (1983). Nonspecific vaginitis. Diagnostic criteria and microbial and epidemiologic associations. Am.J.Med., 74, (1) 14-22. Annang, L., Grimley, D.M. and Hook, E.W., III (2006). Vaginal douche practices among black women at risk: exploring douching prevalence, reasons for douching, and sexually transmitted disease infection. Sex Transm.Dis., 33, (4) 215-219. Antonio M.A.D. and Hillier S.L. (2003). DNA Fingerprinting of Lactobacillus cripatus Strain CTV-05 by Repetitive Element Sequence-Based PCR Analysis in a Pilot Study of Vaginal Colonization. The Journal of Clinical Microbiology, 41(5), 1881-1887 Antonio, M.A., Hawes, S.E., and Hillier, S.L. (1999). The identification of vaginal Lactobacillus species and the demographic and microbiologic characteristics of women colonized by these species. J.Infect.Dis., 180, (6) 1950-1956. Antonio, M.A., Meyn, L.A., Murray, P.J., Busse, B. and Hillier, S.L. (2009). Vaginal colonization by probiotic Lactobacillus crispatus CTV-05 is decreased by sexual activity and endogenous Lactobacilli. J.Infect.Dis., 199, (10) 1506-1513. 98 Anukam, K., Osazuwa, E., Ahonkhai, I., Ngwu, M., Osemene, G., Bruce, A.W. and Reid, G. (2006a). Augmentation of antimicrobial metronidazole therapy of bacterial vaginosis with oral probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14: randomized, double-blind, placebo controlled trial. Microbes.Infect., 8, (6) 14501454. Anukam, K.C., Osazuwa, E., Osemene, G.I., Ehigiagbe, F., Bruce, A.W. and Reid, G. (2006b). Clinical study comparing probiotic Lactobacillus GR-1 and RC-14 with metronidazole vaginal gel to treat symptomatic bacterial vaginosis. Microbes.Infect., 8, (12-13) 2772-2776 Anukam, K.C., Osazuwa, E.O., Ahonkhai, I. and Reid, G. (2006c). Lactobacillus vaginal microbiota of women attending a reproductive health care service in Benin city, Nigeria. Sex Transm.Dis., 33, (1) 59-62 Anukam, K.C. and Reid, G. (2007). Organisms associated with bacterial vaginosis in Nigerian women as determined by PCR-DGGE and 16S rRNA gene sequence. Afr.Health Sci., 7, (2) 68-72 Aroutcheva, A., Gariti, D., Simon, M., Shott, S., Faro, J., Simoes, J.A., Gurguis, A. and Faro, S. (2001). Defense factors of vaginal lactobacilli. Am.J.Obstet.Gynecol., 185, (2) 375-379 99 Austin, M.N., Beigi, R.H., Meyn, L.A. and Hillier, S.L. (2005). Microbiologic response to treatment of bacterial vaginosis with topical clindamycin or metronidazole. J.Clin.Microbiol., 43, (9) 4492-4497 Baeten, J.M., Hassan, W.M., Chohan, V., Richardson, B.A., Mandaliya, K., NdinyaAchola, J.O., Jaoko, W. and McClelland, R.S. (2009). Prospective study of correlates of vaginal Lactobacillus colonisation among high-risk HIV-1 seronegative women. Sex Transm.Infect., 85, (5) 348-353 Bailey, J.V., Farquhar, C. and Owen, C. (2004). Bacterial vaginosis in lesbians and bisexual women. Sex Transm.Dis., 31, (11) 691-694 Bartleson, N.R. (1992). Bacterial vaginosis: a subtle yet serious infection. Nurse Pract.Forum., 3, (3) 130-134 Beasley, S.S., Takala, T.M., Reunanen, J., Apajalahti, J. and Saris, P.E. (2004). Characterization and electrotransformation of Lactobacillus crispatus isolated from chicken crop and intestine. Poult.Sci., 83, (1) 45-48 Beigi, R.H., Wiesenfeld, H.C., Hillier, S.L., Straw, T. and Krohn, M.A. (2005). Factors associated with absence of H2O2-producing Lactobacillus among women with bacterial vaginosis. J.Infect.Dis., 191, (6) 924-929 Bolton, M., van der, S.A. and Cohen, C.R. (2008). Probiotics: potential to prevent HIV and sexually transmitted infections in women. Sex Transm.Dis., 35, (3) 214-225 100 Boris, S., Suarez, J.E., Vazquez, F. and Barbes, C. (1998). Adherence of human vaginal lactobacilli to vaginal epithelial cells and interaction with uropathogens. Infect.Immun., 66, (5) 1985-1989 Brabin, L., Roberts, S.A., Fairbrother, E., Mandal, D., Higgins, S.P., Chandiok, S., Wood, P., Barnard, G. and Kitchener, H.C. (2005). Factors affecting vaginal pH levels among female adolescents attending genitourinary medicine clinics. Sex Transm.Infect., 81, (6) 483-487 Bradshaw C.S., Mortom A.N., Garland S.M., Horvath L.B. and Kusevska L. (2005). Evaluation of a Point-of-Care Test BVBlue and Clinical and Laboratory Criteria for Diagnosis of Bacterial Vaginosis. The Journal of Clinical Microbiology, 43(3), 1304-1308 Bradshaw, C.S., Morton, A.N., Garland, S.M., Morris, M.B., Moss, L.M. and Fairley, C.K. (2005). Higher-risk behavioral practices associated with bacterial vaginosis compared with vaginal candidiasis. Obstet.Gynecol., 106, (1) 105-114 Bradshaw, C.S., Morton, A.N., Hocking, J., Garland, S.M., Morris, M.B., Moss, L.M., Horvath, L.B., Kuzevska, I. and Fairley, C.K. (2006a). High recurrence rates of bacterial vaginosis over the course of 12 months after oral metronidazole therapy and factors associated with recurrence. J.Infect.Dis., 193, (11) 1478-1486 Bradshaw, C.S., Tabrizi, S.N., Fairley, C.K., Morton, A.N., Rudland, E. and Garland, S.M. (2006b). The association of Atopobium vaginae and Gardnerella vaginalis 101 with bacterial vaginosis and recurrence after oral metronidazole therapy. J.Infect.Dis., 194, (6) 828-836 Braxton, E.E., Jr., Ehrlich, G.D., Hall-Stoodley, L., Stoodley, P., Veeh, R., Fux, C., Hu, F.Z., Quigley, M. and Post, J.C. (2005). Role of biofilms in neurosurgical devicerelated infections. Neurosurg.Rev., 28, (4) 249-255 Bukusi, E.A., Cohen, C.R., Meier, A.S., Waiyaki, P.G., Nguti, R., Njeri, J.N. and Holmes, K.K. (2006). Bacterial vaginosis: risk factors among Kenyan women and their male partners. Sex Transm.Dis., 33, (6) 361-367 Bump, R.C. and Buesching, W.J., III (1988). Bacterial vaginosis in virginal and sexually active adolescent females: evidence against exclusive sexual transmission. Am.J.Obstet.Gynecol., 158, (4) 935-939 Burton, J.P. and Reid, G. (2002). Evaluation of the bacterial vaginal flora of 20 postmenopausal women by direct (Nugent score) and molecular (polymerase chain reaction and denaturing gradient gel electrophoresis) techniques. J.Infect.Dis., 186, (12) 1770-1780 Carey, J.C., Klebanoff, M.A., Hauth, J.C., Hillier, S.L., Thom, E.A., Ernest, J.M., Heine, R.P., Nugent, R.P., Fischer, M.L., Leveno, K.J., Wapner, R. and Varner, M. (2000). Metronidazole to prevent preterm delivery in pregnant women with asymptomatic bacterial vaginosis. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N.Engl.J.Med., 342, (8) 534-540 102 Cherpes, T.L., Hillier, S.L., Meyn, L.A., Busch, J.L. and Krohn, M.A. (2008). A delicate balance: risk factors for acquisition of bacterial vaginosis include sexual activity, absence of hydrogen peroxide-producing lactobacilli, black race, and positive herpes simplex virus type 2 serology. Sex Transm.Dis., 35, (1) 78-83 Chondros P., Gunn J., Chondros P., Grover S., Hurley S. and Garland S. (2004). The PAV Trial: Does Lactobacillus prevent post-antibiotic vulvovaginal candidiasis? Protocol of randomised controlled trial. BMC Family Practice, 5(5), 1-10 Cribby, S., Taylor, M. and Reid, G. (2008). Vaginal microbiota and the use of probiotics. Interdiscip.Perspect.Infect.Dis., 2008:256490. 2009 Mar 29., 256490 Czaja, C.A., Stapleton, A.E., Yarova-Yarovaya, Y. and Stamm, W.E. (2007). Phase I trial of a Lactobacillus crispatus vaginal suppository for prevention of recurrent urinary tract infection in women. Infect.Dis.Obstet.Gynecol., 2007:35387., 35387 De, B.E., Verhelst, R., Verstraelen, H., Alqumber, M.A., Burton, J.P., Tagg, J.R., Temmerman, M. and Vaneechoutte, M. (2007). Quantitative determination by real-time PCR of four vaginal Lactobacillus species, Gardnerella vaginalis and Atopobium vaginae indicates an inverse relationship between L. gasseri and L. iners. BMC.Microbiol., 7, 115 Demba, E., Morison, L., van der Loeff, M.S., Awasana, A.A., Gooding, E., Bailey, R., Mayaud, P. and West, B. (2005). Bacterial vaginosis, vaginal flora patterns and vaginal hygiene practices in patients presenting with vaginal discharge syndrome in The Gambia, West Africa. BMC.Infect.Dis., 5, (1) 12 103 Demirezen, S., Korkmaz, E. and Beksac, M.S. (2005). Association between trichomoniasis and bacterial vaginosis: examination of 600 cervicovaginal smears. Cent.Eur.J.Public Health, 13, (2) 96-98 Devillard, E., Burton, J.P. and Reid, G. (2005). Complexity of vaginal microflora as analyzed by PCR denaturing gradient gel electrophoresis in a patient with recurrent bacterial vaginosis. Infect.Dis.Obstet.Gynecol., 13, (1) 25-31 Eriksson, K., Carlsson, B., Forsum, U. and Larsson, P.G. (2005). A double-blind treatment study of bacterial vaginosis with normal vaginal lactobacilli after an open treatment with vaginal clindamycin ovules. Acta Derm.Venereol., 85, (1) 42-46 FAO & WHO (2001). Regulatory and clinical aspects of dairy probiotics: Food and Agriculture Organization of the United Nations and World Health Organization Expert Consultation Report. Online Report, Online Report. (2001). Online Report, WHO. Ref Type: Online Source Ferris, M.J., Norori, J., Zozaya-Hinchliffe, M. and Martin, D.H. (2007). Cultivationindependent analysis of changes in bacterial vaginosis flora following metronidazole treatment. J.Clin.Microbiol., 45, (3) 1016-1018 Fredricks, D.N., Fiedler, T.L. and Marrazzo, J.M. (2005a). Molecular identification of bacteria associated with bacterial vaginosis. N.Engl.J.Med., 353, (18) 1899-1911 104 Fredricks, D.N., Fiedler, T.L. and Marrazzo, J.M. (2005b). Molecular identification of bacteria associated with bacterial vaginosis. N.Engl.J.Med., 353, (18) 1899-1911 Fredricks, D.N., Fiedler, T.L., Thomas, K.K., Oakley, B.B. and Marrazzo, J.M. (2007). Targeted PCR for detection of vaginal bacteria associated with bacterial vaginosis. J.Clin.Microbiol., 45, (10) 3270-3276 Gardiner G.E., Heinmann C., Bruce A.W., Beurman D. and Reid G. (2003). Persistence of Lactobacillus fermentum RC-14 and Lactobacillus rhamnosus GR-1 but not L. rhamnosus GG in the Human Vagina as Demonstrated by Randomly Amplified Polymorphic DNA. Clinical and Diagnostic laboratory Immunology, 9(1), 92-96 Gelber, S.E., Aguilar, J.L., Lewis, K.L. and Ratner, A.J. (2008). Functional and phylogenetic characterization of Vaginolysin, the human-specific cytolysin from Gardnerella vaginalis. J.Bacteriol., 190, (11) 3896-3903 Genc, M.R., Vardhana, S., Delaney, M.L., Onderdonk, A., Tuomala, R., Norwitz, E. and Witkin, S.S. (2004). Relationship between a toll-like receptor-4 gene polymorphism, bacterial vaginosis-related flora and vaginal cytokine responses in pregnant women. Eur.J.Obstet.Gynecol.Reprod.Biol., 116, (2) 152-156 Hall-Stoodley, L., Costerton, J.W. and Stoodley, P. (2004). Bacterial biofilms: from the natural environment to infectious diseases. Nat.Rev.Microbiol., 2, (2) 95-108 105 Hallen, A., Jarstrand, C. and Pahlson, C. (1992). Treatment of bacterial vaginosis with lactobacilli. Sex Transm.Dis., 19, (3) 146-148 Hawes, S.E., Hillier, S.L., Benedetti, J., Stevens, C.E., Koutsky, L.A., WolnerHanssen, P. and Holmes, K.K. (1996). Hydrogen peroxide-producing lactobacilli and acquisition of vaginal infections. J.Infect.Dis., 174, (5) 1058-1063 Hemmerling, A., Harrison, W., Schroeder, A., Park, J., Korn, A., Shiboski, S. and Cohen, C.R. (2009). Phase 1 dose-ranging safety trial of Lactobacillus crispatus CTV-05 for the prevention of bacterial vaginosis. Sex Transm.Dis., 36, (9) 564-569 Hemmerling, A., Harrison, W., Schroeder, A., Park, J., Korn, A., Shiboski, S., Foster-Rosales, A. and Cohen, C.R. (2010). Phase 2a study assessing colonization efficiency, safety, and acceptability of Lactobacillus crispatus CTV-05 in women with bacterial vaginosis. Sex Transm.Dis., 37, (12) 745-750 Jacobsson, B., Pernevi, P., Chidekel, L. and Jorgen Platz-Christensen, J. (2002). Bacterial vaginosis in early pregnancy may predispose for preterm birth and postpartum endometritis. Acta Obstet.Gynecol.Scand., 81, (11) 1006-1010 Jones, B.M., Geary, I., Alawattegama, A.B., Kinghorn, G.R. and Duerden, B.I. (1985). In-vitro and in-vivo activity of metronidazole against Gardnerella vaginalis, Bacteroides spp. and Mobiluncus spp. in bacterial vaginosis. J.Antimicrob.Chemother., 16, (2) 189-197 106 Karaer, A., Boylu, M. and Avsar, A.F. (2005). Vaginitis in Turkish women: symptoms, epidemiologic - microbiologic association. Eur.J.Obstet.Gynecol.Reprod.Biol., 121, (2) 211-215 Kelly, M.C., Mequio, M.J. and Pybus, V. (2003). Inhibition of vaginal lactobacilli by a bacteriocin-like inhibitor produced by Enterococcus faecium 62-6: potential significance for bacterial vaginosis. Infect.Dis.Obstet.Gynecol., 11, (3) 147-156 Khot, P.D., Ko, D.L., Hackman, R.C. and Fredricks, D.N. (2008). Development and optimization of quantitative PCR for the diagnosis of invasive aspergillosis with bronchoalveolar lavage fluid. BMC.Infect.Dis., 8:73., 73 Kiss, H., Kogler, B., Petricevic, L., Sauerzapf, I., Klayraung, S., Domig, K., Viernstein, H. and Kneifel, W. (2007). Vaginal Lactobacillus microbiota of healthy women in the late first trimester of pregnancy. BJOG., 114, (11) 1402-1407 Klebanoff, S.J., Hillier, S.L., Eschenbach, D.A. and Waltersdorph, A.M. (1991). Control of the microbial flora of the vagina by H2O2-generating lactobacilli. J.Infect.Dis., 164, (1) 94-100 Kwok, L., Stapleton, A.E., Stamm, W.E., Hillier, S.L., Wobbe, C.L. and Gupta, K. (2006). Adherence of Lactobacillus crispatus to vaginal epithelial cells from women with or without a history of recurrent urinary tract infection. J.Urol., 176, (5) 2050-2054 107 Larsson, P.G., Stray-Pedersen, B., Ryttig, K.R. and Larsen, S. (2008). Human lactobacilli as supplementation of clindamycin to patients with bacterial vaginosis reduce the recurrence rate; a 6-month, double-blind, randomized, placebo-controlled study. BMC.Womens Health., 8:3., 3 Marrazzo, J.M., Thomas, K.K., Fiedler, T.L., Ringwood, K. and Fredricks, D.N. (2008). Relationship of specific vaginal bacteria and bacterial vaginosis treatment failure in women who have sex with women. Ann.Intern.Med., 149, (1) 20-28 Marrazzo, J.M., Wiesenfeld, H.C., Murray, P.J., Busse, B., Meyn, L., Krohn, M. and Hillier, S.L. (2006). Risk factors for cervicitis among women with bacterial vaginosis. J.Infect.Dis., 193, (5) 617-624 Martinez, B., Sillanpaa, J., Smit, E., Korhonen, T.K. and Pouwels, P.H. (2000). Expression of cbsA encoding the collagen-binding S-protein of Lactobacillus crispatus JCM5810 in Lactobacillus casei ATCC 393(T). J.Bacteriol., 182, (23) 6857-6861 Martinez, R.C., Franceschini, S.A., Patta, M.C., Quintana, S.M., Gomes, B.C., De Martinis, E.C. and Reid, G. (2009). Improved cure of bacterial vaginosis with single dose of tinidazole (2 g), Lactobacillus rhamnosus GR-1, and Lactobacillus reuteri RC-14: a randomized, double-blind, placebo-controlled trial. Can.J.Microbiol., 55, (2) 133-138 Mastromarino, P., Brigidi, P., Macchia, S., Maggi, L., Pirovano, F., Trinchieri, V., Conte, U. and Matteuzzi, D. (2002). Characterization and selection of vaginal 108 Lactobacillus strains for the preparation of vaginal tablets. J.Appl.Microbiol., 93, (5) 884893 Mastromarino, P., Macchia, S., Meggiorini, L., Trinchieri, V., Mosca, L., Perluigi, M. and Midulla, C. (2009). Effectiveness of Lactobacillus-containing vaginal tablets in the treatment of symptomatic bacterial vaginosis. Clin.Microbiol.Infect., 15, (1) 67-74 Matu, M.N., Orinda, G.O., Njagi, E.N., Cohen, C.R. and Bukusi, E.A. (2009). In vitro inhibitory activity of human vaginal lactobacilli against pathogenic bacteria associated with bacterial vaginosis in Kenyan women. Anaerobe., 20 (1) 1-6 McLean, N.W. and McGroarty, J.A. (1996). Growth inhibition of metronidazolesusceptible and metronidazole-resistant strains of Gardnerella vaginalis by Lactobacilli in vitro. Appl.Environ.Microbiol., 62, (3) 1089-1092 McLean, N.W. and Rosenstein, I.J. (2000). Characterisation and selection of a Lactobacillus species to re-colonise the vagina of women with recurrent bacterial vaginosis. J.Med.Microbiol., 49, (6) 543-552 Mijac, V.D., Dukic, S.V., Opavski, N.Z., Dukic, M.K. and Ranin, L.T. (2006). Hydrogen peroxide producing lactobacilli in women with vaginal infections. Eur.J.Obstet.Gynecol.Reprod.Biol., 129, (1) 69-76 available from: PM:16814920 Mitchell, H. 2004. Vaginal discharge--causes, diagnosis, and treatment. BMJ, 328, (7451) 1306-1308 109 Morris, M.C., Rogers, P.A. and Kinghorn, G.R. (2001). Is bacterial vaginosis a sexually transmitted infection? Sex Transm.Infect., 77, (1) 63-68 Nansel, T.R., Riggs, M.A., Yu, K.F., Andrews, W.W., Schwebke, J.R. and Klebanoff, M.A. (2006). The association of psychosocial stress and bacterial vaginosis in a longitudinal cohort. Am.J.Obstet.Gynecol., 194, (2) 381-386 Neri, A., Sabah, G. and Samra, Z. (1993). Bacterial vaginosis in pregnancy treated with yoghurt. Acta Obstet.Gynecol.Scand., 72, (1) 17-19 available from: PM:8382424 Ness, R.B., Kip, K.E., Soper, D.E., Hillier, S., Stamm, C.A., Sweet, R.L., Rice, P. and Richter, H.E. (2005). Bacterial vaginosis (BV) and the risk of incident gonococcal or chlamydial genital infection in a predominantly black population. Sex Transm.Dis., 32, (7) 413-417 Nilsson, U., Hellberg, D., Shoubnikova, M., Nilsson, S. and Mardh, P.A. (1997). Sexual behavior risk factors associated with bacterial vaginosis and Chlamydia trachomatis infection. Sex Transm.Dis., 24, (5) 241-246 Nugent, R.P., Krohn, M.A. and Hillier, S.L. (1991). Reliability of diagnosing bacterial vaginosis is improved by a standardized method of gram stain interpretation. J.Clin.Microbiol., 29, (2) 297-301 Nyirjesy, P., McIntosh, M.J., Steinmetz, J.I., Schumacher, R.J. and Joffrion, J.L. (2007). The effects of intravaginal clindamycin and metronidazole therapy on vaginal 110 mobiluncus morphotypes in patients with bacterial vaginosis. Sex Transm.Dis., 34, (4) 197-202 Oakley, B.B., Fiedler, T.L., Marrazzo, J.M. and Fredricks, D.N. (2008). Diversity of human vaginal bacterial communities and associations with clinically defined bacterial vaginosis. Appl.Environ.Microbiol., 74, (15) 4898-4909 Ocana, V., Silva, C. and Nader-Macias, M.E. (2006). Antibiotic susceptibility of potentially probiotic vaginal lactobacilli. Infect.Dis.Obstet.Gynecol., 14, (3) 18182 Papanikolaou, E.G., Tsanadis, G., Dalkalitsis, N. and Lolis, D. (2002). Recurrent bacterial vaginosis in a virgin adolescent: a new method of treatment. Infection, 30, (6) 403-404 Parent, D., Bossens, M., Bayot, D., Kirkpatrick, C., Graf, F., Wilkinson, F.E. and Kaiser, R.R. (1996). Therapy of bacterial vaginosis using exogenously-applied Lactobacillus acidophili and a low dose of estriol: a placebo-controlled multicentric clinical trial. Arzneimittelforschung., 46, (1) 68-73 Patterson, J.L., Girerd, P.H., Karjane, N.W. and Jefferson, K.K. (2007). Effect of biofilm phenotype on resistance of Gardnerella vaginalis to hydrogen peroxide and lactic acid. Am.J.Obstet.Gynecol., 197, (2) 170-177 Patterson, J.L., Stull-Lane, A., Girerd, P.H. and Jefferson, K.K. (2010). Analysis of adherence, biofilm formation and cytotoxicity suggests a greater virulence potential of 111 Gardnerella vaginalis relative to other bacterial-vaginosis-associated anaerobes. Microbiology., 156, (Pt 2) 392-399 Pavlova, S.I., Kilic, A.O., Mou, S.M. and Tao, L. (1997). Phage infection in vaginal lactobacilli: an in vitro study. Infect.Dis.Obstet.Gynecol., 5, (1) 36-44 Randis, T.M., Kulkarni, R., Aguilar, J.L. and Ratner, A.J. (2009). Antibody-based detection and inhibition of vaginolysin, the Gardnerella vaginalis cytolysin. PLoS.One., 4, (4) e5207 Rao, P.S., Devi, S., Shriyan, A., Rajaram, M. and Jagdishchandra, K. (2004). Diagnosis of bacterial vaginosis in a rural setup: Comparison of clinical algorithm, smear scoring and culture by semiquantitative technique. Indian J.Med.Microbiol., 22, (1) 47-50 Reid G. (1999). The Scientific Basis for Probiotic Strains of Lactobacillus. Applied and Environmental Microbiology, 65(9), 3763-3766 Reid G., Charbonneau D., Erb J., Kochanowski B. and Beurman D. (2003a). Oral use of Lactobacillus rhamnosus GR-1 and L. fermentum RC-14 significantly alters vaginal flora: randomised placebo-controlled trial in 64 healthy women. FEMS Immunology and Medical Microbiology, 35, 131-134 Reid G., Jass J., Sebulsky M.T. and McCormick J.K. (2003b). Potential Uses of Probiotics in Clinical Practice. Clinical Microbiology Reviews, 16(4), 658-672 112 Reid, G., Jass, J., Sebulsky, M.T. and McCormick, J.K. (2003). Potential uses of probiotics in clinical practice. Clin.Microbiol.Rev., 16, (4) 658-672 Schellenberg, J., Blake, B.T., Lane, M., Cheang, M. and Plummer, F. (2008). Flow cytometric quantification of bacteria in vaginal swab samples self-collected by adolescents attending a gynecology clinic. J.Microbiol.Methods., 73, (3) 216-226 Schwebke, J.R. (2003). Gynecologic consequences of bacterial vaginosis. Obstet.Gynecol.Clin.North Am., 30, (4) 685-694 Schwebke, J.R. and Desmond, R. (2005). Risk factors for bacterial vaginosis in women at high risk for sexually transmitted diseases. Sex Transm.Dis., 32, (11) 654-658 Schwebke, J.R. and Desmond, R. (2007a). A randomized trial of metronidazole in asymptomatic bacterial vaginosis to prevent the acquisition of sexually transmitted diseases. Am.J.Obstet.Gynecol., 196, (6) 517-6 Schwebke, J.R. and Desmond, R.A. (2007b). A randomized trial of the duration of therapy with metronidazole plus or minus azithromycin for treatment of symptomatic bacterial vaginosis. Clin.Infect.Dis., 44, (2) 213-219 Schwebke, J.R., Desmond, R.A. and Oh, M.K. (2004). Predictors of bacterial vaginosis in adolescent women who douche. Sex Transm.Dis., 31, (7) 433-436 Shalev, E., Battino, S., Weiner, E., Colodner, R. and Keness, Y. 1996. Ingestion of yogurt containing Lactobacillus acidophilus compared with pasteurized yogurt as 113 prophylaxis for recurrent candidal vaginitis and bacterial vaginosis. Arch.Fam.Med., 5, (10) 593-596 Simoes, J.A., Aroutcheva, A., Heimler, I., Shott, S. and Faro, S. (2001). Bacteriocin susceptibility of Gardnerella vaginalis and its relationship to biotype, genotype, and metronidazole susceptibility. Am.J.Obstet.Gynecol., 185, (5) 1186-1190 Smart, S., Singal, A. and Mindel, A. (2004). Social and sexual risk factors for bacterial vaginosis. Sex Transm.Infect., 80, (1) 58-62 Smit, E., Oling, F., Demel, R., Martinez, B. and Pouwels, P.H. (2001). The S-layer protein of Lactobacillus acidophilus ATCC 4356: identification and characterisation of domains responsible for S-protein assembly and cell wall binding. J.Mol.Biol., 305, (2) 245-257 Song, Y.L., Kato, N., Matsumiya, Y., Liu, C.X., Kato, H. and Watanabe, K. (1999a). Identification of and hydrogen peroxide production by fecal and vaginal lactobacilli isolated from Japanese women and newborn infants. J.Clin.Microbiol., 37, (9) 3062-3064 Song, Y.L., Kato, N., Matsumiya, Y., Liu, C.X., Kato, H. and Watanabe, K. (1999b). Identification of Lactobacillus species of human origin by a commercial kit, API50CHL. Rinsho Biseibutshu.Jinsoku.Shindan.Kenkyukai.Shi, 10, (2) 77-82 Soper, D.E., Livengood, C., Sheehan, K.L., Fenner, D.E., Martens, M.G., Nelson, A.L., Ismail, M., Thorp, J.M., Lappin, M., Fleury, F.J., Long, B.J., Blackwelder, T., 114 Sweet, R.L. and Sagov, S. (1998). Safety and efficacy comparison of two dosing regimens of MetroGel-Vaginal(R) in the treatment of bacterial vaginosis: a multicenter, randomized, single-blind, parallel comparison. Prim.Care Update.Ob Gyns., 5, (4) 150 Srinivasan, S. and Fredricks, D.N. (2008). The human vaginal bacterial biota and bacterial vaginosis. Interdiscip.Perspect.Infect.Dis., 2008:750479. Epub;%2009 Feb 16., 750479 Stroman, P., Muller, C.C. and Sorensen, K.I. (2003). Heat shock treatment increases the frequency of loss of an erythromycin resistance-encoding transposable element from the chromosome of Lactobacillus crispatus CHCC3692. Appl.Environ.Microbiol., 69, (12) 7173-7180 Svare, J.A., Schmidt, H., Hansen, B.B. and Lose, G. (2006). Bacterial vaginosis in a cohort of Danish pregnant women: prevalence and relationship with preterm delivery, low birthweight and perinatal infections. BJOG., 113, (12) 1419-1425 Swidsinski, A., Mendling, W., Loening-Baucke, V., Ladhoff, A., Swidsinski, S., Hale, L.P. and Lochs, H. (2005). Adherent biofilms in bacterial vaginosis. Obstet.Gynecol., 106, (5 Pt 1) 1013-1023 Swidsinski, A., Mendling, W., Loening-Baucke, V., Swidsinski, S., Dorffel, Y., Scholze, J., Lochs, H. and Verstraelen, H. (2008). An adherent Gardnerella vaginalis biofilm persists on the vaginal epithelium after standard therapy with oral metronidazole. Am.J.Obstet.Gynecol., 198, (1) 97-6 115 Taha T.E., Hoover D.R., Dallabetta G.A., Kumwenda N.I., Mtimavalye L.A., Yang L.P. and Liomba G.N. (1998). Bacterial vaginosis and disturbances of vaginal flora: association with increased acquisition of HIV. Aids, 12, 1699-1706 Taha, T.A., Osta, W., Kozhaya, L., Bielawski, J., Johnson, K.R., Gillanders, W.E., Dbaibo, G.S., Hannun, Y.A. and Obeid, L.M. (2004). Down-regulation of sphingosine kinase-1 by DNA damage: dependence on proteases and p53. J.Biol.Chem., 279, (19) 20546-20554 Taha, T.E., Kumwenda, N.I., Kafulafula, G., Makanani, B., Nkhoma, C., Chen, S., Tsui, A. and Hoover, D.R. (2007). Intermittent Intravaginal Antibiotic Treatment of Bacterial Vaginosis in HIV-Uninfected and -Infected Women: A Randomized Clinical Trial. PLoS.Clin.Trials, 2, (2) e10 Tao, L., Pavlova, S.I., Mou, S.M., Ma, W.G. and Kilic, A.O. (1997). Analysis of Lactobacillus products for phages and bacteriocins that inhibit vaginal lactobacilli. Infect.Dis.Obstet.Gynecol., 5, (3) 244-251 Toba, T., Virkola, R., Westerlund, B., Bjorkman, Y., Sillanpaa, J., Vartio, T., Kalkkinen, N. and Korhonen, T.K. (1995). A Collagen-Binding S-Layer Protein in Lactobacillus crispatus. Appl.Environ.Microbiol., 61, (7) 2467-2471 Tosun, I., Alpay, K.S., Ciftci, H., Buruk, C.K., Aydin, F., Kilic, A.O. and Erturk, M. (2007). Biotypes and antibiotic resistance patterns of Gardnerella vaginalis strains 116 isolated from healthy women and women with bacterial vaginosis. Mikrobiyol.Bul., 41, (1) 21-27 Toth, A., O'Leary, W.M., and Ledger, W. (1982). Evidence for microbial transfer by spermatozoa. Obstet.Gynecol., 59, (5) 556-559 Vallor, A.C., Antonio, M.A., Hawes, S.E. and Hillier, S.L. (2001). Factors associated with acquisition of, or persistent colonization by, vaginal lactobacilli: role of hydrogen peroxide production. J.Infect.Dis., 184, (11) 1431-1436 Vasquez, A., Jakobsson, T., Ahrne, S., Forsum, U. and Molin, G. (2002). Vaginal Lactobacillus flora of healthy Swedish women. J.Clin.Microbiol., 40, (8) 2746-2749 Verstraelen, H., Delanghe, J., Roelens, K., Blot, S., Claeys, G. and Temmerman, M. (2005). Subclinical iron deficiency is a strong predictor of bacterial vaginosis in early pregnancy. BMC.Infect.Dis., 5, 55 Vitali, B., Pugliese, C., Biagi, E., Candela, M., Turroni, S., Bellen, G., Donders, G.G., and Brigidi, P. (2007). Dynamics of vaginal bacterial communities in women developing bacterial vaginosis, candidiasis or no infection analysed by PCR-denaturing gradient gel electrophoresis and real-time PCR. Appl.Environ.Microbiol. Waigankar, S.S. and Patel, V. (2011). Role of probiotics in urogenital healthcare. J.Midlife.Health, 2, (1) 5-10 117 Wilks, M., Wiggins, R., Whiley, A., Hennessy, E., Warwick, S., Porter, H., Corfield, A. and Millar, M. (2004). Identification and H(2)O(2) production of vaginal lactobacilli from pregnant women at high risk of preterm birth and relation with outcome. J.Clin.Microbiol., 42, (2) 713-717 Yan, D.H., Lu, Z., and Su, J.R. (2009). Comparison of main Lactobacillus species between healthy women and women with bacterial vaginosis. Chin Med.J.(Engl.)., 20;122, (22) 2748-2751 Yen, S., Shafer, M.A., Moncada, J., Campbell, C.J., Flinn, S.D., and Boyer, C.B. (2003). Bacterial vaginosis in sexually experienced and non-sexually experienced young women entering the military. Obstet.Gynecol., 102, (5 Pt 1) 927-933 Yeoman, C.J., Yildirim, S., Thomas, S.M., Durkin, A.S., Torralba, M., Sutton, G., Buhay, C.J., Ding, Y., Dugan-Rocha, S.P., Muzny, D.M., Qin, X., Gibbs, R.A., Leigh, S.R., Stumpf, R., White, B.A., Highlander, S.K., Nelson, K.E., and Wilson, B.A. (2010). Comparative genomics of Gardnerella vaginalis strains reveals substantial differences in metabolic and virulence potential. PLoS.One., 5, (8) e12411 Yuki, N., Shimazaki, T., Kushiro, A., Watanabe, K., Uchida, K., Yuyama, T. and Morotomi, M. (2000). Colonization of the stratified squamous epithelium of the nonsecreting area of horse stomach by lactobacilli. Appl.Environ.Microbiol., 66, (11) 5030-5034 118 Zarakolu, P., Sahin Hodoglugil, N.N., Aydin, F., Tosun, I., Gozalan, A. and Unal, S. (2004). Reliability of interpretation of gram-stained vaginal smears by Nugent's scoring system for diagnosis of bacterial vaginosis. Diagn.Microbiol.Infect.Dis., 48, (2) 77-80 Zingler, V.C., Cnyrim, C., Jahn, K., Weintz, E., Fernbacher, J., Frenzel, C., Brandt, T. and Strupp, M. (2007). Causative factors and epidemiology of bilateral vestibulopathy in 255 patients. Ann.Neurol., 61, (6) 524-532 119 APPENDICES APPENDIX 1: ENROLMENT CASE REVIEW FORM (CRF) Screening ID # [ ] [ ] [ ] [S] Subject Identification# [][][][]-[] Visit Date Informed consent obtained Date of Birth I. [ ] [ ] [ ] [ ] [ ] [ ] (date) [ ] [ ] [ ] [ ] [ ] [ ] (date) [ ] [ ] [ ] [ ] [ ] [ ] (date) [ ] [ ] [ ] [ ] [ ] [ ] (date) DEMOGRAPHICS [1] Do you consider yourself to be Hispanic or Latino? [ ] No [ ] Yes If “Yes” go to Q3 [2] What race do you consider yourself to be: [ ] Black or African American [ ] White [ ] Asian [ ] American Indian or Alaska Native [ ] Native Hawaiian or Other Pacific Islander [ ] Other (specify) ………………………………. [3] How many years of formal education did you complete? [ ] [ ] [4] What is the highest level of education you completed? [ ] Did not complete high school [ ] Completed high school [ ] Completed junior college [ ] Completed college (undergraduate degree) [ ] Completed graduate degree [5] What is your current employment status? (mark all that apply) [ ] Student [ ] Employed full time [ ] Employed part-time [ ] Unemployed, looking for work [ ] Unemployed, not looking for work [ ] Other (please specify) ……………………………. 120 II. MEDICAL HISTORY [6] Have you had any medical problems that resulted in you being in a hospital, seeing a doctor regularly, or taking a medication on a daily basis? □ Yes If yes, list diagnoses below: □ No Onset year ………………. ………………. ………………. ………………. ………………. 6a.…………………………… 6b.…………………………… 6c.…………………………… 6d.……………………………. 6e.……………………………. Ongoing □Yes □No □Yes □No □Yes □No □Yes □No □Yes □No [7] Do you have any other medical problems or ongoing conditions that were not controlled by medicinal, psychological, or behavioral therapy? □ Yes If yes, list below: 7a………………………………………. 7b………………………………………. 7c………………………………………. 7d………………………………………. 7e………………………………………. □ No [8] Have you had symptoms of any medical conditions within the past 30 days? □ Yes If yes, list symptoms below: □ No 8a………………………………………. 8b………………………………………. 8c………………………………………. 8d………………………………………. 8e………………………………………. [9] ASSESSED BY STAFF: Are any symptoms listed in item 3 clinically significant? □ Yes □ No [10] Are you allergic to anything? □ Yes □ No Next page If yes, mark all that apply: □ Penicillin □ Hay fever □ Foods □ Latex □ Spermicides □ Other antibiotics (specify) ……………………………. □ Any other allergy (specify) ……………………………. 121 [11] Do you have any serious systemic allergic disease (e.g., urticaria or atopic dermatitis) □ Yes If yes, specify: ……………………………… □ No [12] Did you ever experience vaginal irritation after vaginally applied agents (e.g. KY jelly)? □ Yes If yes, how many incidences …………………. □ No [13] Are you currently taking any medications (including prescription drugs, contraceptives, antibiotics, vitamins, supplements)? □ Yes If yes, specify: ………………………. □ No [14] □ Yes Have you ever taken investigational drugs? If yes, specify …………………………….. □ No FOR SITE PI USE ONLY: [15] Is the participant stable on all medications or therapies? □ Yes □ No Investigator’s signature: …………………………………………… 122 III. GYNAECOLOGY [16] HISTORY What was the first day of your last menstrual period (LMP) [ ] [ ] [ ] (Date) 16a. How many days is your average menstrual cycle? [ ] (Days) 16b. How many days does your period last? [ ] (Days) 16c. Do you use pads, tampons, both or neither during your period? (Mark all that apply [ ] Pads [ ] Tampons [ ] Cups [ ] other (specify) ……….. 16d. In the past 3 months, have you had any abnormal menstrual cycles? [ ] No [ ] Yes [ ] Cycle length less than 21days [ ] Cycle length more than 35 days [ ] Intermediate bleeding / spotting [ ] Stable on continuous OCP or long acting progestin [17] Have you ever douched or used vaginal preparations or drying agents? [ ] Yes [ ] No b) If yes to 17a. In the last 30 days, how many times have you douched or used other vaginal preparations or drying agents? [ ] (number of times) If “No” go to Q18 [18] Are you currently using any method of birth control? [ ] Yes [ ] No If “No” go to Q19 18 (b) What birth Control Methods/hormonal regulation are you using currently (mark a response for EACH item) [ ] Yes [ ] No – Condoms [ ] Yes [ ] No – Pills (for birth control) [ ] Yes [ ] No – Pills (for hormonal regulation) [ ] Yes [ ] No – Depo (injection) [ ] Yes [ ] No – Norplant (sub-dermal implants) [ ] Yes [ ] No – Abstinence [ ] Yes [ ] No – IUD [ ] Yes [ ] No – Male partner with vasectomy [ ] Yes [ ] No – Lesbian orientation [ ] Yes [ ] No - Tubal ligation (Bartleson 1992) [ ] Yes [ ] No – Skin patch [ ] Yes [ ] No – Hysterectomy [ ] Yes [ ] No – Other (Specify) [19] What birth control method(s) do you plan to use over the next 1-2 months, while you are still on this study a. [ ] Same as above b. [ ] Other (Specify) ………………. 123 [20] Have you ever had any of these infections? How often in the last 6 months (>=2 ineligible) Number of times in your life Infection UTI (urinary tract infection) Pyelonephritis (kidney infection) Vaginal yeast infection Pelvic Inflammatory Disease (PID) Genital Herpes Trichomonas Chlamydia Gonorrhoea HIV Bacterial Vaginosis Syphilis Other, specify: ___________ [ [ [ [ [ [ [ [ [ [ [ [ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ] ] ] ] ] ] ] ] ] ] ] ] [ [ [ [ [ [ [ [ [ [ [ [ Do you smoke? [ ] Yes [ ] No 124 ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ] ] ] ] ] ] ] ] ] ] ] ] Date of last diagnosis (<21 days ineligible) [ [ [ [ [ [ [ [ [ [ [ [ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ] ] ] ] ] ] ] ] ] ] ] ] [ [ [ [ [ [ [ [ [ [ [ [ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ][ ] ] ] ] ] ] ] ] ] ] ] ] Diagnosed by physician (Yes or No) [ [ [ [ [ [ [ [ [ [ [ [ ] ] ] ] ] ] ] ] ] ] ] ] [ [ [ [ [ [ [ [ [ [ [ [ ] ] ] ] ] ] ] ] ] ] ] ] APPENDIX 2: ELIGIBILITY CRITERIA Please Mark a Response for Each Item 1) Are you between 18 – 40 years of age? 2) Are you willing to be asked a series of questions, including some about your medical health and some about your sexual history? 3) Have you ever had sex (including vaginal intercourse) or been to an OB-Gyn for a pelvic exam? 4) Are you willing to have a full check by a nurse or a doctor to see if you can be in the study? This will include a physical exam, testing for sexually transmitted infections and pregnancy, a vaginal exam with a speculum, and colposcopy (to examine and photograph the inside of your vagina) 5) Will you live in this area for 2 months or more and be available for follow up visits? 6) If you join this study, can you come to the clinic at least 3 more times over the next 4 to 6 weeks? 7) Do you have a regular menstrual cycle (21-35 days) or have you been amenorrheic for at least 3 months due to use of a long acting progestin, continuous use of oral contraceptives or hysterectomy? 8) Are you willing to insert pre-filled vaginal applicators once per day? 9) Are you willing to abstain from inserting anything (including tampons, fingers, contraceptive creams, sponges, douches, lubricants, etc.) into your vagina unless prescribed by a physician, for the duration of the study? 10) Are you willing to abstain from sexual intercourse 72 hours before enrollment until after Day 10 of the study and using condoms until the end of the study? 11) Are you willing to use an adequate reliable method of birth control until after Day 10 of the study and using condoms until the end of the study? 12) Are you willing to abstain from participating in any other research studies for the duration of this trial (until after the last study visit on Day 30?) 13) Are you able to read and understand English and capable of providing informed consent? 14) Are you currently in mutually monogamous relationship or not sexually active? Screening ID [] [] [] [S] Date [][] [][] [][] Staff signature and date: ___________________________ 125 YES Eligible NO Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible APPENDIX 3: ELIGIBILITY CRITERIA (FORM 2) 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. Please Mark a Response for Each Item In the past 21 days, have you had a vaginal or cervical infection with Trichomoniasis, Gonorrhea, Chlamydiasis, Syphilis, a Urinary Tract Infection, Bacterial Vaginosis or genital Herpes? Have you been diagnosed with Gonorrhea, Chlamydiasis, Trichomoniasis or Syphilis on 2 or more occasions during the previous 6 months? Do you have a history of recurrent herpes genitalis? Are you currently pregnant or have you had a pregnancy (including an abortion, miscarriage, birth, or current pregnancy) in the past 2 months? Are you currently breastfeeding or are you planning to over the next 4 to 6 weeks?? In the past 2 weeks, have you received antibiotic or antifungal therapy (Vaginal or systemic)? In the past 25 days (or 10 half lives, whichever is longer), have you used an investigational drug? Are you in menopause? In the past 3 days, have you used a vaginal contraceptive ring (e.g. NuvaRing_) or plan to during the course of the study? Do you have any medical conditions or acute illnesses or significant disease(s) (e.g. diabetes, cancer, psoriasis, etc.)? Do you have HIV? In the past 60 days, have you used an immunosuppressive drug? Do you have any history of drug or alcohol abuse? Are you allergic to any component of LACTIN-V, the placebo control substance, Metronidazole, latex, or have you had an allergic reaction to any medication (i.e. rash, joint swelling, nephritis, anaphylactic shock)? In the past 3 months, have you started using a new long acting treatment form of hormonal birth control (i.e. Depo-Provera)? In the past 3 months, have you had an intrauterine device (IUD) inserted or removed? In the past 3 months, have you had any gynecologic surgery (hysterectomy or pelvic surgery? In the past 3 months, have you had cervical cryotherapy, LEEP, or cervical laser therapy? Do you plan to have surgery in the next 4 to 6 weeks? Have you previously participated in a LACTIN-V trial? Mark YES if: Participant has any social or medical condition (e.g., neurological, gastrointestinal, renal, hepatic, cardiovascular, pulmonary, metabolic, endocrine, hematological, genitourinary or other major disorder), or psychiatric illness (e.g., depression, schizophrenia) that, in the opinion of the Investigator, would preclude provision of informed consent, make participation in the study unsafe, complicate interpretation of study outcome data, or otherwise interfere with achieving the study objectives. Screening ID [] [] [] [S] Staff signature and date: __________________ Date [] [] [] [] [] [] 126 NO YES Eligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible Ineligible APPENDIX 4: FINAL VISIT CASE REVIEW FORM (CRF) Screening ID [ ] [ ] [ ] [S] I. [ ] [ ] [ ] [ ] [ ] [ ] (date) RECENT MEDICAL HISTORY 1. Have you had any medical problems since your last visit? [ ] No [ ] Yes (Please list the diagnosis below) 1) ……………………………………………………………… 2) …………………………………………………………….. 3) …………………………………………………………….. 4) …………………………………………………………….. [ ] (tick if still ongoing) [ ] (tick if still ongoing) [ ] (tick if still ongoing) [ ] (tick if still ongoing) 2. Have you taken an investigational drug (a drug not yet approved or still in trial) since you last visit? [ ] No [ ] Yes (Please specify) …………………………………………… 3. Have you taken any antibiotics or medications (including prescription medications, topical medications or hormonal medications) since your last visit? [ ] No [ ] Yes (Please specify) …………………………………………… II. BIRTH CONTROL 4. Has your birth control method changed since your last visit? [ ] No [ ] Yes (Please specify method) …………………………………………… 127 III. UROGENITAL HISTORY 5. Since your last visit, have you had any of the following infections? Infection No Yes UTI (urinary tract infection) Pyelonephritis (kidney infection) Vaginal yeast infection Pelvic Inflammatory Disease (PID) Genital Herpes Trichomonas Chlamydia Gonorrhoea HIV Other, specify: ___________ [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] If “Yes” indicate number of times [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] [ ][ ] IV. GYNAECOLOGICAL HISTORY 6. First day of your last menstrual period? [ ] (day) [ ] (month) [ ] (year) 7. Since your last visit, have you experienced any spotting, or bleeding, other than menstrual bleeding? [ ] Yes [ ] No 8. Since you last visit, did you use pads, or tampons, or both during your period? (Mark all that apply) Yes No [] [ ] Pads [] [ ] Tampons [] [ ] Cups [] [ ] Other (Specify) ……………………………….. 9. Since your last visit, have you douched or vaginally inserted anything other than the study applicator? [ ] Yes [ ] No 9 (a) How long ago did you last douche or vaginally inserted anything other than the study applicator? [ ] (No. of days since) 9 (b) Since your last visit, how many times have you douched or vaginally inserted anything other than the study applicator? [ ] (No. of times) (Go to the next section) 128 V. SEXUAL ACTIVITY HISTORY 10. Since your last visit, how many times did you have sex? [ ] [ ] (No. of times had sex) 11. Since your last visit, how many sex partners did you have? [ ] [ ] (No. of sex partners) 12. How many days has it been since the last time you had sexual intercourse? (Days since the last sexual intercourse) [][] 13. If you had sex wit a man, did your partner use condoms each time? [ ] Yes [ ] No [ ] N/A VI. STUDY PRODUCT ADHERENCE 14. Please write down insertion times for each day of product use. DAY 01 02 03 04 05 12 19 MM DD YY [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] [] Time of 1st Insertion (24 hour clock) [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] Time of 2nd Insertion (24 hour clock) [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] [ ]:[ ] 129 Reasons for “not on schedule” or “missed” insertion ……………………………………….. ……………………………………….. ……………………………………….. ……………………………………….. ……………………………………….. ……………………………………….. ……………………………………….. APPENDIX 5: CONSENT FORM UNIVERSITY OF CALIFORNIA, SAN FRANCISCO CONSENT TO PARTICIPATE IN A RESEARCH STUDY Study Title: Factors Affecting Vaginal Colonization with Probiotic Lactobacillus Crispatus: A Study of the Intervention Cohort in a Phase IІA Clinical Trial of Lactin-V This is a medical research study. A designated member of the research team led by your study doctors Dr. Craig Cohen, Dr. Anke Hemmerling, and Dr. Abner Korn from the UCSF Department of Obstetrics, Gynecology and Reproductive Sciences will explain this study to you. Medical research studies include only people who choose to take part. Take your time to make your decision about participating. You may discuss your decision with your family and friends and with your health care team. If you have any questions, you may ask our team or your study doctor. You are being asked to take part in this study because you are a woman with bacterial vaginosis. Why is this study being done? Over 300 women have already used Lactin-V safely as a gelatin capsule. However, the purpose of this study is to test the safety and acceptability of Lactin-V as a powder in a pre-filled applicator. Lactin-V is not approved by the United States Food and Drug Administration (Abad and Safdar 2009). This is a Phase IIa study, which means that the goal is to see if the study drug is safe, acceptable and effective at colonizing the vagina with Lactobacillus and to find out what factors may hinder this colonization. The investigators hope to learn about the side effects and how the body handles the drug. In four previous research studies testing Lactin-V in a gelatin capsule, no serious safety problems with Lactin-V have been reported. Lactin-V is a powder and will be administered with an applicator into the vagina. The powder contains Lactobacillus crispatus. Lactobacillus is a type of bacteria that is naturally found in the vagina of healthy women. Too low amounts of Lactobacillus may allow “harmful” bacteria and viruses to survive in the vagina. This may cause genital infections like bacterial vaginosis. It is thought that Lactin-V may help to decrease the number of bacterial vaginosis infections by increasing the amount of normal Lactobacillus crispatus in the vagina. The study is being conducted by UCSF and is sponsored by Osel, Inc., the manufacturer of the investigational drug Lactin-V being used in this study. Osel, Inc. pays for the conduct of this study, including part of Dr. Cohen’s, Dr. Hemmerling’s, and Dr. Korn’s 130 salary, and is providing the study drug at no cost to the researcher or the study participant. What will happen if I take part in this research study? At the initial screening visit, you will be tested to determine if you currently have a bacterial vaginosis infection and meet all other criteria necessary to participate in this study. If you are diagnosed with bacterial vaginosis at this visit, you will be prescribed a 5 day course of 0.75% topical Metronidazole (Soper, Livengood, Sheehan, Fenner, Martens, Nelson, Ismail, Thorp, Lappin, Fleury, Long, Blackwelder, Sweet, & Sagov 1998), the standard antibiotic therapy recommended for bacterial vaginosis. After the screening visit, the study will include 3 more visits over approximately 28 days. You will be asked to not use vaginal products such as vaginal douches or tampons from 72 hours before enrollment until after your third, and last, visit (Day 28). In addition, you will be asked to not have intercourse from 72 hours before enrollment until your second visit (Day 10). You will also be asked to refrain from sex for 48 hours after study product administration on Day 12 and Day 19, and 72 hours before your third, and last, study visit (Day 28). In the times you are sexually active, we ask that you use an adequate method of birth control throughout the study. If eligible and agreeing to participate in the study, you will return for the enrollment visit 24-72 hours after you finish your antibiotic treatment for your bacterial vaginosis infection, and then start taking the study product Lactin-V for 5 consecutive days. One week after your fifth dose, you will be asked to take the product once per week for two weeks. You will return for a follow-up visit 10 days after the enrollment visit. You will return for a final visit about 28 days after the enrollment visit. You will be asked to complete a diary form for each product administration, noting side effects and time/date of the application. At all study visits a thorough interview about your experiences with the product, a gynecological exam, including colposcopy (to examine the inside of your vagina with a speculum and through a microscope for an illuminated and magnified view, and to take digital photographs of your genital area) and the collection of vaginal samples, will be performed. During the last visit you will be invited to participate in a focus group discussion about your experiences with the product. Before you begin the main part of the study... First you will need to have a “screening” exam. Some people may not be able to join the research study because of information found during the screening tests. Based on the results of the screening, we will either ask you to: 1. Start antibiotic treatment for your bacterial vaginosis infection and enroll in the study, or 2. First be treated for an infection other than bacterial vaginosis, be retested and then possibly enroll, or 3. Not enroll in the study (not be eligible). 131 Only women with bacterial vaginosis are eligible for participation in this study. We will determine today whether you are eligible to participate in this study. The screening visit will take about 120 minutes. You will need to have the following “screening” exams, tests or procedures to find out if you can be in the main part of the study: Medical and Reproductive History, contact information: The study staff will ask you questions about your health and sexual practices. You will receive counseling about HIV and other infections passed during sex. These infections are called syphilis, gonorrhea, chlamydia, trichomoniasis, and genital herpes. In addition, we will ask you questions about where you live, and how you can be contacted (e.g. phone number, e-mail). The study staff will use this information to remind you of scheduled visits. If you miss a visit, the study staff will try to contact you by phone and e-mail. Then they will try to reach you through the contact people that you list. If they talk to these people, they will not tell them why they are trying to reach you. Acceptability questionnaire: You will be asked to fill out an acceptability questionnaire about your experiences with bacterial vaginosis, your expectations for a vaginal product like LACTIN-V and your experience with vaginal products in the past. Urine sample: You will be asked to give a urine sample for laboratory tests. Pregnancy testing: Because the effect of this drug is unknown in fetuses,, pregnant women may not participate in this study. A urine test will be done at every visit to make sure you are not pregnant. If you are pregnant, you will not be eligible for the study. The study staff will refer you to an appropriate clinic or other services that you may need. If the study is still ongoing after your pregnancy, you can come back here to find out if you are eligible then. Physical Examination: You will have a physical examination, similar to those done for regular medical care. Blood drawing (venipuncture): You will be asked to give a blood sample for laboratory tests. Approximately 4 teaspoons of blood will be drawn by inserting a needle into a vein in your arm for these tests. They will test your blood for HIV and syphilis. Sometimes test results are not clearly positive but also not negative. In that case, we will do more tests until we know the result for sure. HIV testing: Some of the blood taken for laboratory tests will be used to test for HIV (the AIDS test). You will talk about what it may mean to know the results of these tests, and whether you are prepared to receive the test results. You must agree to receive your test results to be in the research study. You will receive the test results in person and will be counseled about the meaning of these results before and after the test. Gynecological Examination: You will also have an exam of your genital area and inside your vagina to help the study staff determine if you have any sores or signs of an infection. The study nurse will collect fluid from your vagina with a swab to test for infections. These infections are called gonorrhea, chlamydia, and trichomoniasis. The vaginal swabs will also be used to test for other vaginal 132 conditions known as candidiasis, and bacterial vaginosis. Lastly, swabs will be collected to measure the amount of naturally occurring Lactobacillus you have, and to measure other bacteria commonly associated with bacterial vaginosis. The study clinician will also use a swab to collect a sample from your cervix to test for abnormalities that could lead to cervical cancer. This test is called a “Pap test”. If you are diagnosed with bacterial vaginosis at this visit, we will confirm this with laboratory tests and supply you with a topical antibiotic ointment, (0.75% Metronidazole, brand name MetroGel) that you will apply to your vagina for five days. If you do not have a current bacterial vaginosis infection, you are not eligible to participate in the study at this time. Most of these other tests will take 3-5 days to be processed. The results of these tests will affect whether you will be eligible for the study. If you are not eligible for the study, the study staff will tell you about other studies you may be eligible for, if any. If you have syphilis, gonorrhea, chlamydia, or trichomoniasis, we will give you medicine to treat these infections. After successful treatment, you may be invited for rescreening. If you have HIV or an abnormal Pap test, we will refer you to available sources of medical care, counseling and other services you may need. You will be asked to read and sign this informed consent document if you choose to participate in the screening for the study. You will need to sign this document before any of the study-related tests and procedures are performed. This screening visit will take about 2 hours. During the main part of the study... If the screening exams, tests or procedures show that you can continue to be in the study, and you choose to take part, then you will have the following tests and procedures done. You will be asked to come back to the clinic for a minimum of three more visits: the enrollment visit (First Visit – Day 1), then about 10 days later (Second Visit--Day 7-12), and then 28 days later (Third Visit--Day 24-32). Enrolment Visit (Day 1): The enrollment visit will take place 24-72 hours after you have completed your antibiotic treatment for bacterial vaginosis, and will last about 120 minutes. During this visit, the following procedures will take place: 133 Update on Medical and Reproductive History, Contact information: You will update information on where you live and how to keep in contact with you. The study staff will use this information to remind you of scheduled visits. If you miss a visit, the study staff will try to contact you by phone and email. They will try to reach you through the contact people that you list. If they talk to these people, they will not tell them why they are trying to reach you. You will answer interview questions about your sexual practices. Gynecological Examination: You will have an exam of your genital area and inside your vagina. An instrument called a speculum will be used to examine your vagina and cervix. During this exam the study clinician will look through a lens called a “colposcope.” The lens works like a magnifying glass to help the study clinician see abnormalities. The lens is attached to a camera that will be used to take a picture of your vagina and cervix. During the exam, the study clinician will collect several samples of vaginal fluids with cotton swabs. Urine sample: You will be asked to give a urine sample for laboratory tests. Pregnancy testing: Because the effects of this drug are unknown to a fetus, pregnant women may not participate in this study. If you become pregnant during the study despite the request of abstinence from vaginal intercourse, your participation in the study will have to be terminated. We will refer you to an appropriate clinic or other services that you may need. If you have a new infection, other than bacterial vaginosis, you will not be eligible to continue participating. You will be given medicine, if needed, and asked to come back here after taking all of the medicine. At that time, you may be eligible for the research study. If you have no signs of infection, other than bacterial vaginosis, during the screening and the enrollment exam, have a negative pregnancy test and choose to participate in the study: You will be "randomized" into one of the study groups described below. Randomization means that you are put into a group by chance. A computer program will place you in one of the groups. Neither you nor the study personnel can choose the group you will be in and neither you nor the study personnel will know if you got LACTIN-V or the placebo (an inactive substance). You will be placed in 1 of 2 study groups. The first group will consist of women who all received LACTIN-V powder. The second group will consist of women who all received a placebo powder. You have a three in four chance of being placed in the LACTIN-V group and one in four chance of being placed in the placebo group. Both groups are very important to this study. Participants in both groups will have the same study visits. All participants must refrain from sexual 134 intercourse until after the second study visit on Day 10 after enrollment, then 48 hours after administering the study product on Day 12 and Day 19. Finally, we ask that you refrain from sex 72 hours before your final visit on Day 28. You will be given the study product, and told how you use it over the next 5 days, then the following two weeks. You will be asked to administer the applicator for the first time while you are in the clinic and study staff will ask if you have any questions. Please feel free to ask staff any type of questions during this explanation or any time during the study. You will be told how to complete the “Study Diary”. The “Study Diary” is a list that you keep during the study to mark when you use the applicators and whether you are experiencing any problems when you use them. You will be told how to tell the difference between normal vaginal discharge and vaginal discharge that is discolored or has an abnormal smell. Study staff will explain that if at any point you feel discomfort, notice discolored or abnormal discharge, or are in pain, you should call the study clinician or come here to the clinic. If the staff finds that a study product is causing you problems, they may ask you to stop using the product, either for a short time or permanently. Even if you stop using the product, you will be asked to stay in the study and have your visits as originally planned. You will be asked to bring your used ‘applicators’, and Study Diary with you to each study visit. Visit 2 (Day 10): This visit will take about 90 minutes. At this visit, Update on Medical and Reproductive History, Contact information: The study staff will make sure we have the correct information about where you live and other questions about you, your health, and your sexual practices. Review of the “Study Diary”: The study staff will talk to you about your use of the product. They will collect and count the used ‘applicators’ that you have brought from home. Study staff will also go over the “Study Diary” to see how you marked using the product and if you have any questions. You will be asked questions about how your body feels since starting to use the product and if it feels any different. . Gynecological Examination: You will have an exam of your genital area and inside your vagina. An instrument called a speculum will be used to examine your vagina and cervix. During this exam the study staff will look through the “colposcope” and take pictures of your vagina and cervix. The clinician will collect several samples of vaginal fluids with cotton swabs. Urine sample: You will be asked to give a urine sample for laboratory tests. 135 Pregnancy testing: Because the drugs in this study can affect a fetus, pregnant women may not participate in this study. If you become pregnant, during the study despite the request of abstinence from vaginal intercourse, your participation in the study will have to be terminated. We will refer you to an appropriate clinic or other services that you may need. If you have any evidence of an infection, or if you reported any symptoms of an infection of the genital tract, the study doctor will collect fluid from your vagina with a swab to test for infection. If you have evidence of an infection, you will be given medicine, if needed, and asked to come back here after taking all of the medicine. 136 Visit 3 (Day 28): This visit will take about 150 minutes, and will be like Visit 2 (at Day 10). The only difference will be that there are 3 additional procedures: Review of the “Study Diary”: The study staff will talk to you about your use of the product since the last visit. They will collect and count the used ‘applicators’ that you have brought from home. Study staff will also go over the “Study Diary” to see how you marked using the product and if you have any questions. You will be asked questions about how your body feels since stopping the use of the product and if it feels any different. Acceptability Questionnaire: You will be asked to fill out an acceptability questionnaire now that you’ve used the study product, and share your suggestions for improvement. Acceptability Focus Group: You will be invited to participate in a focus group to discuss your experiences with the study product with several other participants (approximately 3-4 participants). The focus group will take place either on the same day as your visit 3, before or after your other procedures planned for this visit to minimize the waiting time, or at another day. It will take approximately 45 minutes. You will only use your first name during the discussion. This focus group will be audio taped to make sure that the researchers afterwards can analyze all discussion points without forgetting important information. You can choose to complete the Visit 3 without participating in the focus group discussion. The tapes will be destroyed when the study is complete. How long will I be in the study? Participation in this study, including the visits, the time for administration of the product and documentation in the Study Diary Card at home will take a total of about 15 hours over a period of 28 days. Can I stop being in the study? Yes. You can decide to stop at any time. Tell the study team if you are thinking about stopping or decide to stop. They will tell you how to stop your participation safely. It is important to tell the study team if you are thinking about stopping so that they can evaluate any risks from the LACTIN-V, and discuss what alternative follow-up care and testing could be most helpful for you. The study doctor may stop you from taking part in this study at any time if he/she believes it is in your best interest, if you do not follow the study rules, or if the study is stopped. What side effects or risks can I expect from being in the study? You may have side effects while on the study. Everyone taking part in the study will be watched carefully for any side effects. However, doctors don't know all the side effects that may happen. Side effects may be mild or very serious. Your health care team may give you medicines to help lessen side effects. Many side effects go away soon after you stop taking LACTIN-V. In some cases, side effects can be serious, long lasting, or may never go away. You should talk to the study team about any side effects you 137 experience while taking part in the study. Major side effects are not likely since the LACTIN-V powder contains bacteria that normally live in a healthy vagina. Risks and side effects related to the study product LACTIN-V and the study procedures include: Risks from taking the study product LACTIN-V: Likely Vaginal, genital or menstrual symptoms: vaginal discharge Less Likely o Gastrointestinal symptoms: abdominal (Yuki et al. 2000) pain, constipation, diarrhoea, nausea, vomiting o Urinary symptoms: needing to urinate urgently, needing to urinate at night and pain with urination o Vaginal, genital or menstrual symptoms: genital itching, bleeding between menstrual periods, delayed menstrual periods, vaginal odor, vaginal burning sensation, vaginal irritation, vaginal bleeding and vaginal dryness, genital swelling, rash, vaginal candidiasis (yeast infection) o Other symptoms: lower back pain Rare but serious Allergy: A possible risk of using the LACTIN-V product or the placebo product is an allergic reaction. It is possible to have an allergic reaction to one of the ingredients (Lactobacillus crispatus and the preservative powder). In rare cases, if not properly treated, an allergic reaction could be life-threatening. Risks from study procedures: Likely Randomization risks: You will be assigned to LACTIN-V or the placebo by chance, and if you receive the LACTIN-V treatment you may experience more side effects than with the placebo. Blood drawing (venipuncture) risks: Drawing blood may cause temporary discomfort from the needle stick, bruising, and rarely, infection. Medical History Review: You may become embarrassed, worried, or anxious when discussing your sexual practices, ways to protect against HIV and other infections passed during sex, and your test results. You may become worried or anxious while waiting for your test results. Genital Exam: You may experience pain or discomfort when the clinician checks inside your vagina; there may be a small amount of bleeding during or after the vaginal exam. Reproductive risks: You should not become pregnant while on this study because it is unknown whether the drugs in this study may affect an unborn baby. You should not breastfeed a baby while on this study. It is important to understand that you must use birth control while on this study. Check with your study doctor about what kind of birth control methods to use and how long to use them. Some methods might not be approved for use in this study. If you become pregnant during the study, the study staff will refer you to available sources of medical care and other services you may need. You will stop using the study product, but keep 138 coming here for study visits as originally planned. We will ask you to have a study visit after the pregnancy is completed so that we can find out about your pregnancy. HIV testing risks: Being tested for HIV may cause anxiety regardless of the test results. A positive test indicates that you have been infected with the HIV virus, but no one knows for certain when, if ever, you will become sick with AIDS or a related condition. Receiving positive results may make you very upset. If other people learn about your positive test results, you may have trouble obtaining insurance or employment. If your test is negative, there is still the possibility that you could be infected with the HIV virus and test positive at some time in the future. Also, it is always possible that the test results could be wrong. Risks regarding your privacy of information: During the focus group acceptability survey at Visit 3, the researchers will ask you sensitive information about your satisfaction with the product in a group setting. You will only use your first name and all participants are asked not to tell anyone outside the group what any particular person said in the group. However, the researchers cannot guarantee that everyone will keep the discussions private. This focus group will be audio taped. The audiotapes will be kept in a locked file cabinet and will be destroyed after the completion of the study. Unknown Risks: The experimental treatments may have side effects that no one knows about yet. The researchers will let you know if they learn anything that might make you change your mind about participating in the study. For more information about risks and side effects, ask the study team or your study doctor. Are there benefits to taking part in the study? There may be no direct benefit to you from participating in this study. However, this study will help doctors learn more about LACTIN-V, and it is hoped that this information will help in the treatment of future patients with bacterial vaginosis. What other choices do I have if I do not take part in this study? You can choose to receive your annual women’s health exam with your health care provider. Please talk to your doctor about your choices before deciding if you will take part in this study. Will my medical information be kept private? We will do our best to make sure that the personal information in your medical record is kept private. However, we cannot guarantee total privacy. Your personal information may be given out if required by law. If information from this study is published or presented at scientific meetings, your name and other personal information will not be used. 139 During the focus group acceptability survey at Visit 3 about your satisfaction with the product, the researchers will ask you and the other people in the group to use only first names during the group session. They will also ask you not to tell anyone outside the group what any particular person said in the group. However, the researchers cannot guarantee that everyone will keep the discussions private. This focus group will be audio taped to make sure that the researchers afterwards can analyze all discussion points without forgetting important information. Like all your files, the audiotapes will be kept in a locked file cabinet during the study. The audiotapes will be destroyed after the completion of the study. Organizations that may look at and/or copy your medical records for research, quality assurance, and data analysis include: The United States Food and Drug Administration (Abad & Safdar 2009) Study monitors Study staff Study auditors, including representatives of Osel Inc., the company that is developing LACTIN-V The Committee on Human Research at UCSF San Francisco Department of Public Health (California state law requires that we report positive test results for syphilis, gonorrhea, chlamydia, and HIV) Participation in research may involve a loss of privacy, but information about you will be handled as confidentially as possible. A medical record will be created because of your participation in this study. Your consent form and some of your research test results will be included in this record. You will be identified by a code, and personal information from your records will not be released without your written permission. Hospital regulations require that all health care providers treat information in medical records confidentially. What are the costs of taking part in this study? You will not be charged for any of the study activities. Will I be paid for taking part in this study? In return for your time, effort and travel expenses, you will be paid a maximum of $320 for taking part in this study: You will be reimbursed up to a maximum of $80 for your time, your travel costs and parking expenses for coming to each visit. You will not have to show your receipts for parking or travel expenses. Should an additional interim visit be required, you will also receive up to a maximum of $80 for your time, your travel costs and parking expenses at this visit. You will receive cash payment after each visit. 140 What happens if I am injured because I took part in this study? It is important that you tell your study team or your study doctors Dr. Cohen, Dr. Hemmerling or Dr. Korn, if you feel that you have been injured from taking part in this study. You can tell the doctor in person or call him/her at 415-597-4963. Treatment and Compensation for Injury: If you are injured as a result of being in this study, treatment will be available. The costs of the treatment may be covered by the University of California or the study sponsor Osel, Inc., depending on a number of factors. The University and the study sponsor do not normally provide any other form of compensation for injury. For further information about this, you may call the office of the Committee on Human Research at 415- 476-1814. What are my rights if I take part in this study? Taking part in this study is your choice. You may choose either to take part or not to take part in the study. If you decide to take part in this study, you may leave the study at any time. No matter what decision you make, there will be no penalty to you and you will not lose any of your regular benefits. Leaving the study will not affect your medical care. You can still get your medical care from our institution. We will tell you about new information or changes in the study that may affect your health or your willingness to continue in the study. In the case of injury resulting from this study, you do not lose any of your legal rights to seek payment by signing this form. 141 Who can answer my questions about the study? You can talk to your study team about any questions or concerns you have about this study. Contact your study doctors at any time. Researcher Institution Contact Dr. Craig Cohen University of California, San Francisco 1-415-597-9192 Dr. Anke Hemmerling University of California, San Francisco 1-415-597-4963 Dr. Abner Korn University of California, San Francisco 1-415–885–7788 Additionally, you can contact the dedicated 24-hour LACTIN-V hotline 1-866-LACTINV (1-866-522-8468), or send an e-mail to the Study Coordinator, William Harrison, at lactinv@globalhealth.ucsf.edu. For questions about your rights while taking part in this study, call the office of the Committee on Human Research, UCSF's Institutional Review Board (a group of people who review the research to protect your rights) at 415-476-1814. ************************************************************ CONSENT You have been given copies of this consent form and the Experimental Subject's Bill of Rights to keep. PARTICIPATION IN RESEARCH IS VOLUNTARY. You have the right to decline to participate or to withdraw at any point in this study without penalty or loss of benefits to which you are otherwise entitled. If you wish to participate in this study, you should sign below. Date I also agree to participate in the focus group at the final visit. Participant's Printed Name for Consent ________________________________________________ Participant’s Signature for Consent Date Person Obtaining Consent 142 APPENDIX 6: KEMRI SCIENTIFIC STEERING COMMITTEE APPROVAL 143 APPENDIX 7: KEMRI ETHICAL REVIEW COMMITTEE APPROVAL 144 APPENDIX 8: UCSF COMMITTEE ON HUMAN RESEARCH APPROVAL 145