Experiment 3 PowerPoint
advertisement

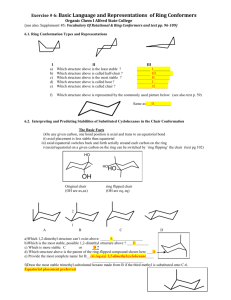
Experiment 3: STEREOCHEMISTRY AND MOLECULAR MODELING OF CYCLOALKANES THE LAB REPORT… This experiment is NOT in the current edition of the lab manual. Each student should print a copy of the FINAL LAB REPORT sheet accessible on the Blackboard Learn course website and bring it to lab to complete. Molecular model kits will be provided for assistance, or students can bring their own to lab. There is no other PRE lab or POST lab assignment for this experiment, just the completion of the FINAL LAB REPORT sheet. OBJECTIVES To practice drawing cyclohexane rings in chair conformations, and learn how to recognize axial and equatorial positions. To practice drawing monosubstituted and disubstituted cyclohexane rings in both chair conformations. To learn how to compare the stability of chair conformations, and identify which conformer is favored at equilibrium. THE LAB REPORT… Fill in the blanks using a single term from the list at the top. Use textbook for assistance if necessary. How to Draw Cyclohexane 1. Draw a wide V. 2. Draw a line going down at a 60o angle, ending just before the center of the V. 4. Draw a line parallel to the line from step 2, going down exactly as low as that line. 3. Draw a line parallel to the left side of the V ending just before the left side of the V. 5. Connect the dots. THE LAB REPORT… Follow instructions given for all problems. How to Draw the Axial and Equatorial Hydrogens 1. Draw all axial positions as parallel lines, alternating in directions. SUMMARY: All substituents are drawn like this: a a e a e e e e e 2. Draw all equatorial positions as pairs of parallel lines. parallel with this side! a a a THE LAB REPORT… Follow instructions given for all problems. Conformational Mobility of Cyclohexane-Ring Flips Conformations of Monosubstituted Cyclohexanes Although ring-flip occurs rapidly, the two conformers are not EQUAL! This conformer has more diaxial interactions, therefore is higher in energy! 1,3-Diaxial Interactions Q: What causes the difference in energy between the conformers? Steric strain due to 1,3-diaxial interactions. Q: What is a 1,3-diaxial interaction? Atoms on C1 are too close to those on C3 and C5! Drawing Both Chair Conformations: Monosubstituted rings 1. Draw a chair conformation. 2. Place the substituent in an axial position. Br 1 axial 3. Draw the ring flip and the axial group becomes equatorial. Br 1 2 axial 2 equatorial 1 Br 2 THE LAB REPORT… Follow instructions given for all problems. Drawing Both Chair Conformations: Disubstituted rings 1. Using a numbering system, determine the location and configuration of each substituent. Draw the structure using solid and dashed lines to indicate relative (cis vs. trans) stereochemistry. Br 2. Place the substituents on the first chair using the info from step 1. Br H Bromine is at C-1 and is UP H 1 3 1 2 CH2CH3 2 3 CH2CH3 Ethyl is at C-3 and is DOWN 3. Draw the second chair skeleton, and place the substituents using the info from step 1. 2 H 1 3 CH2CH3 H Br THE LAB REPORT… Follow instructions given for all problems. Conformational Analysis of Disubstituted Cyclohexanes Q: What is conformational analysis? Assessing energy of cycloalkane by summing all steric interactions. Q: Why is it important? Can help predict which conformations are more favorable and more likely to exist. THE LAB REPORT… Follow instructions given for all problems. THE LAB REPORT… This table is given on the last page of the lab report… OVERVIEW Review concepts. Draw a chair conformation of cyclohexane. Draw and label axial and equatorial positions on a chair conformation of cyclohexane. Draw chair conformation of monosubstituted cyclohexane before and after ring flip. Draw chair conformations of disubstituted cyclohexanes before and after ring flip. Compare stability of chair conformations and predict major conformer. Just a suggestion… BRING YOUR TEXTBOOK WITH YOU TO LAB! (IT MAY HELP !)