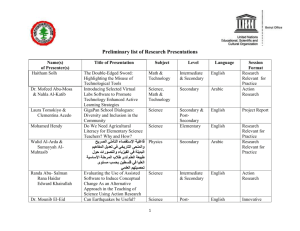
Slide 1 - USD Biology
advertisement

The water-absorption region of ventral skin of several semiterrestrial and aquatic amphibians identified by aquaporins Yuji Ogushi, Azumi Tsuzuki, Megumi Sato, Hiroshi Mochida, Reiko Okada, Masakazu Suzuki, Stanley D. Hillyard and Shigeyasu Tanaka Introduction Semi-terrestrial water balance strategy • Used by many tree frog and toad species • Use ventral pelvic patch to absorb water cutaneously – Capillaries contact basement membrane beneath epithelium • Store dilute urine in bladder for re-absorption while foraging far from water • Aquaporins (AQPs): plasma membrane proteins forming water channels into cells (present in almost all organisms) – Control water permeability across membranes – Stimulated by arginine vasotocin (AVT): causes fusion of vesicles containing AQPs with apical membrane of epithelial water absorption/reabsorption tissues Introduction • Researchers used Real Time Polymerase Chain Reaction (RT-PCR) to identify 2 forms of AQP in epithelial tissues • AQP-h2 (isoform) – Termed “urinary bladder-type” AQP – Found in urinary bladder of all study species – Found in pelvic skin region of toad and tree frog • AQP-h3 (isoform) – Termed “ventral skin-type” AQP – Found in skin but not bladder of tree frogs, toads and Rana species Study Species Hyla japonica (tree frog) Rana japonica (semi-aquatic) Bufo marinus (terrestrial toad) Xenopus laevis (aquatic) Rana nigromaculata (semi-aquatic) Rana catesbaiana aka bullfrog (semi-aquatic) Table 1. Phylogenetics of aquaporins in ventral pelvic skins of anuran species living in different habitats Habitat Species Arboreal Hyla japonica Terrestrial Bufo japonica Semi-aquatic Rana catesbeiana Semi-aquatic Rana nigromaculata Semi-aquatic Rana japonica Aquatic Xenopus laevis Pelvic Skin Bladder AQP-h2-like AQP-h3-Like Protein cDNA ( Bladder- (Ventral PelvicType) Type) AQP-h2-Like Protein (Bladder-Type) + + + + + + - + + + + - + + (but not expressed) + + Introduction • AQP-x3 mRNA homologous to AQP-h3 expressed in pelvic skin of aquatic species, Xenopus laevis – but not translated to protein • Hydrins: intermediate peptides derived from a provasotocin-neurophysin precursor – Stimulate osmotic water movement across skin and bladder – Only present in anurans – Have stimulatory effects on water permeability across pelvic skin in Hyla japonica Objectives • Examine relationship between AQP distribution in apical membranes and ATV stimulation of water permeability in hindlimb, pelvic and pectoral zones of ventral skin • Examine expression of AQP-x3 mRNA in skin of hindlimb, pelvic, pectoral, dorsal regions – Different patterns of regional specialization present in terrestrial, arboreal, and semiaquatic species • Extend observations and compare them with response of Ranid and toad species to AVT Materials and Methods: Immunohistochemistry • 4-mm sections of ventral skin mounted on slides • Reacted with fluorescent labeled anti-bodies – Nuclei stained with DAPI (appear blue) – Pelvic skin type AQP proteins (AQP-h3) stained using Alexa Fluor 488 (appears green) – Urinary bladder-type AQP proteins (AQP-h2) stained using Cy3 (appears red) • Specimens examined with microscope equipped with fluorescence attachment Materials and Methods: Western Blot Analysis III II I • Skin from hind-limb (I), pelvic (II) and pectoral (III) regions removed and homogenized • Proteins separated via gel electrophoresis, transferred to membrane, and probed (detected) using antibodies kDa Protein Molecular Weight Values I II III Materials and Methods: RT-PCR of Xenopus Ventral Skin AQP-x3 • RNA extracted from ventral skin and reverse transcribed • Gel electrophoresis • DNA Sequenced Materials and Methods: Water Permeability • Skin from pectoral, pelvic, and hindlimb regions mounted between two chambers connected by a small opening • Chamber on serosal (inner) side of skin filled with Ringer (salt) solution • Mucosal (outer side) chamber filled with water • Water movement from mucosal to serosal side recorded over 30 min with Ringer solution in mucosal chamber • Followed by 30 min of Ringer solution with AVT • Skins examined by immuno-fluorescence microscopy to evaluate incorporation of AQPs into apical membrane of First Reacting Cell (FRC) layer • FRC layer: continuous barrier between outside and inside of body Materials and Methods: Water Permeability • Effect of AVT on hindlimb skin permeability compared with hydrins 1 and 2 • Skins pretreated with AVT to increase number of AQPs inserted in apical plasma membrane • Skins treated with HgCl2 • Water movement with continued AVT treatment measured for additional 30 min • Results from 5 or 6 individuals expressed as means • Statistical Analysis: data compared by SteelDwass’s test using software Results: Aquaporins in 3 skin regions Rana japonica and Rana nigromaculata: • AQP-h3 (skin-type) in hindlimb region only • Rana japonica: in basolateral, apical, and cytoplasm of FRC • Rana nigromaculata: basolateral plasma membrane Rana japonica Rana nigromaculata Results: Aquaporins in 3 skin regions Rana catesbeiana: • Greatest AQP-h3 in hindlimb • Present in small number pelvic skin cells • In hindlimb and pelvic skin, localized in basolateral plasma membrane in FRC layer • In pectoral region, dot spot only in cytoplasm of few cells in FRC layer • Intensity of labeling decreased from hindlimb to pectoral skin Hindlimb Pelvic Pectoral Results: Aquaporins in 3 skin regions B. marinus: • AQP-h3 and AQP-h2 in all regions •Predominantly in cytoplasm just beneath apical membrane •Number of cells varied among toads (less in pectoral skin of some) •Western Blot: Intensity of bands decreased from hindlimb to pectoral skin Pectoral Hindlimb Pelvis Skin-type AQP-h3 Bladder-type AQP-h2 Results: aquaporins in 3 skin regions Xenopus laevis: • Detected AQP-x3 mRNA expression in skin from pectoral, pelvic, and hindlimb regions but not dorsal skin • X. laevis skin not stimulated by AVT Results: Water permeability and movement of AQPs after stimulation with AVT Rana japonica and Rana nigromaculata: • Stimulation at hindlimb • AQP-h3 in apical plasma membrane in FRC layer Rana japonica Rana nigromaculata Results: Water permeability and movement of AQPs after stimulation with AVT Bullfrog: • Stimulation increased in order of pectoral, pelvic, hindlimb regions • Translocation of AQP-h3 protein to apical plasma membrane of FRC layer greater in hindlimb region and decreased in pelvic and pectoral region hindlimb pelvic pectoral Results: Water permeability and movement of AQPs after stimulation with AVT B. marinus: • Stimulation variable depending on individuals and regions of skin but above controls • ½ of toads: response greatest in hindlimb, declined in pelvic and pectoral skin • Other ½: response greatest in pelvic skin • Translocation of AQP-h3 and AQP-h2 to apical plasma membrane of cells in FRC layer of hindlimb, pelvic, and pectoral regions Results: Water permeability and movement of AQPs after stimulation with AVT For Bufo marinus Hindlimb Skin-type AQP-h3 Bladdertype AQP-h2 Pelvic Pectoral Results: Water permeability and dynamic movement of AQPs after stimulation of AVT and hydrins • AVT and hydrin 1 and 2 increased water permeability of hindlimb skin in R. japonica > R. nigromaculata > R. catesbeina > B. marinus • No differences among hormone response within species • Increased water flux rates (relative to controls): – 30–38 X in Rana japonica – 15 X in Rana nigromaculata – 8–12 X in Rana catesbeina – 3 or 4 X in Bufo marinus • When hindlimb skin from each species stimulated with AVT following HgCl2 treatment, ratio of water flux decreased (compared with AVT stimulation groups) Discussion: Importance of AQP-rich hindlimbs for water absorption • Area-specific rate of AVT-stimulated water flow across hindlimb skin similar for moist and dry-adapted species • Toad: AVT-stimulated water flow correlated with presence of AQPh2-like water channel in all skin regions • Rana Catesbeiana: AQP-h3-like AQP observed in all skin regions • Rana japonica and Rana nigromaculata: AQP-h3-like AQP observed only in hindlimb • Greater response of Toad vs. Rana species in vivo could result from relative area of skin that contains AQPs rather than an area-specific response • HgCl2 inhibited water flux across hindlimb skin under AVTstimulation. – AQP proteins are mercury sensitive, so this proves waterflux was mediated by AQPs Discussion: Physiological and behavioral variables that affect water absorption • Variable area-specific water flux across toad skin could result from greater dependence on vascular perfusion relative to thinner frog skin • Behavioral water absorption response – Skin pressed to moist surface – Large increase in blood flow to absorbing area of seat patch – Insertion of AQPs into apical membranes of FRC skin layer Discussion: Phylogenetic significance of AQPs in ventral pelvic skin • Largest superfamilies of anurans are Hyloidea (includes modern tree frog and toad species) and Ranoidea (includes Ranids (typical frogs) • AQP-h2-like proteins not only in bladder, but in skin of tree frog and toad species, which also have more pelvic patches – Apomorphic (only these lineages have this character) • AQP-h3 found in toad, tree frog, and Ranid species – Pleisiomorphic (likley shared with common ancestors) – Present in all ventral skin regions of Rana Catesbeiana, while only present in hindlimbs of Rana japonica and Rana nigromaculata • “New World” Rana genus recently reclassified as Lithobates, including Rana Catesbeiana • Rana japonica and Rana nigromaculata remain in “Old World” Rana genus Discussion: Expression of 2 AVT-stimulated AQPs in skin of toad and tree frog species • AQP-h2 homolog detected in bladder of all species examined but in skin of only toad and tree frog species • mRNA encoding AQP-h3 homolog identified in skin but not bladder of all species examined • Based on genetic analyses of Xenopus tropicalis, likely that h2and h3-like AQPa2 genes were generated by local gene duplication of AQP2 in anuran lineage • For contemporary anurans h2-like AQPa2 occurs in bladder, while h3-like AQPa2 is expressed in ventral skin • In toad and tree frog species, h2-like AQPa2 gene may have undergone a change to express this gene in the ventral skin, not just the bladder – Might give terrestrial species an advantage: cutaneous water absorption / adaptiaton to drier environments Discussion: A unique AQP in aquatic Xenopus • No hydro-osmotic response to AVT • Identified mRNA for AQP-x3 in pelvic skin homologous to that for AQP-h3, but contains extra C-terminal tail preventing translation • AQP-x3 present in all 3 skin regions • Data lacking on possibility of expression during dry periods Discussion: Regulation of AQP expression by AVT and related peptides • Hydrin 1 and 2 stimulated water permeability of hindlimb skin of toad and tree frog species at level equivalent to AVT • Km values for cAMP production by tree frog V2-type AVT receptor suggests hydrin 1 and 2 share a common receptor • Both peptides generated from down-regulation in posttranslational processing • Xenopus laevis : secretes hydrin 1 and AVT but shows no hydro-osmotic response to either in skin • Xenopus laevis : AVT and hydrin 1 stimulate water reabsorption from bladder – May be involved in water balance during aestivation Perspectives and Significance • Anurans have 2 AQP isoforms stimulated by AVT to increase water absorption across ventral skin and re-absorption from bladder • All species examined express AQP-h2-like AQPs in bladder • Only semi-terrestrial toad and tree-frog species express AQPh3-like AQPs and AQP-h2-like AQPs in skin • Semi-aquatic Ranids express only AQP-h3 in skin, primarily in ventral surface of hindlimbs • Aquatic Xenopus laevis transcribes mRNA for homologs of both isoforms but a C-terminal sequence prevents translation • Future studies needed to examine species differences in expression of AQP-h2 and AQP-h3 to examine phylogenetic relationships associated with water balance adaptations