FFIIMODA-Fire Behavior
advertisement

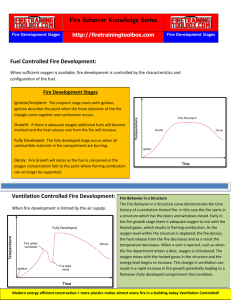
FVCC Fire Rescue FIRE BEHAVIOR OBJECTIVES 2-2.1 2-2.1.1 2-2.1.2 2-2.1.3 2-2.1.4 2-2.1.5 2-2.1.6 Identify the following terms: (3-3.10) Fire/combustion Heat Ignition temperature Flammable limits/flammable range Vapor density Solubility OBJECTIVES 2-2.2 Identify the components of the fire triangle and fire tetrahedron (3-3.10) 2-2.3 Identify the relationship of the concentration of oxygen to combustibility and life safety (3-3.1) 2-2.4 Identify four products of combustion commonly found in structural fires that create a life hazard. (3-3.1) 2-2.5 Identify the three methods of heat transfer (3-3.9, 3-3.11) OBJECTIVES 2-2.6 Identify the Law of Heat Flow 2-2.7 Identify the three physical states of matter in which fuels are commonly found. (3-3.9) 2-2.8 Identify the following conditions and explain their associated hazards and appropriate actions: (3-3.10) OBJECTIVES 2-2.8.1 2-2.8.2 2-2.8.3 2-2.8.4 2-2.8.5 2-2.8.6 2-2.8.7 Ignition/Incipient Growth/Freeburning Rollover/Flameover Flashover Fully developed Decay/Hot smoldering Backdraft OBJECTIVES 2-2.9 Identify the process of thermal layering that occurs in structural fires. (3-3.11) 2-2.10 Identify how to avoid disturbing the normal layering of heat. o IFSTA, Essentials, 4th ed, Chapter 2 o Delmar, Firefighter’s Handbook, 2000, Chapter 4 FIRE TERMS Fire/combustion Fire: Self-sustaining process of rapid oxidation of a fuel which produces heat and light Combustion: Self-sustaining chemical reaction yielding energy or products that cause further reactions These terms are often used interchangeably; most often used in fire FIRE TERMS Heat The form of energy that raises temperature The energy transferred from one body to another when the temperature of the bodies are different Can be measured by the amount of work it does Ignition temperature The minimum temperature to which a fuel, in air, must be heated to start self-sustained combustion without a separate ignition source. FIRE TERMS Flammable Limits/flammable range Highest and lowest percentage of a flammable gas or vapor, in air, that will explode or ignite Concentrations below the Lower Explosive (flammable) Limit are too “lean” to burn Concentrations above the Upper Explosive (flammable) Limit are too “rich” to burn Flammable (explosive) Range: The range between the Lower Explosive (flammable) Limit and the Upper Explosive (flammable) Limit The word explosive and flammable are often used interchangeably FIRE TERMS Vapor density: Weight of a given volume of pure vapor or gas compared to the weight of an equal volume of dry air at the same temperature and pressure Vapor density less than one indicates a vapor lighter than air Vapor density greater than one indicates a vapor heavier than air FIRE TERMS Solubility Degree to which a solid, liquid, or gas dissolves in a solvent (usually water) FIRE TRIANGLE & FIRE TETRAHEDRON Fire triangle – heat, fuel, oxygen Representative of surface combustion (smoldering fire) Fire tetrahedron Chemical chain reaction is added to the fire triangle Representative of the flaming mode of combustion RELATIONSHIP OF OXYGEN TO LIFE SAFETY Concentrations below twenty-one percent has some effect on life safety At nine percent, an individual becomes unconscious At six percent, death will occur within a few minutes In concentrations below eighteen percent, fire begins to decrease Generally concentrations below fifteen percent will not support combustion PRODUCTS OF COMBUSTION Heat Responsible for the spread of fire Cause of burns and other injuries Smoke Mixture of carbon particles and fire gases Makeup varies from fuel to fuel, all smoke is considered toxic The material burning has a direct influence on the amount and color of smoke PRODUCTS OF COMBUSTION Fire gases Carbon monoxide (CO) Carbon Dioxide (CO2) Hydrogen Cyanide Sulfur Dioxide Nitrogen Other gases depending on fuel being burned PRODUCTS OF COMBUSTION Flame (light) More complete the combustion, less luminous the flame Flame absent in smoldering fire HEAT TRANSFER Conduction Heat conducted from one body to another either by direct contact or by an intervening heat conducting medium Depends on type of conductor: metal (good), drywall (poor) Example: Metal plumbing components or electrical conduit HEAT TRANSFER Convection Transfer of heat energy by the movement of air or liquid Heated gases rise: mushrooming Examples: Fire traveling through elevator shafts, stairways, balloon frame walls Direct flame contact is actually a form of convection heat transfer HEAT TRANSFER Radiation Transfer by heat waves Travels through space until it reaches an opaque object Light colors reflect radiant heat; dark colors absorb radiant heat Major source of fire spread to exposures (important to protect exposures from radiant heat) LAW OF HEAT FLOW Heat flows from a hot substance to a cold substance A colder substance will absorb heat until temperatures are equal 3 PHYSICAL STATES OF MATTER Solid fuels Have a definite shape and size Surface to mass ratio The more surface areas exposed – the less energy is required for ignition Pyrolysis The chemical decomposition of a substance through the action of heat The position of the fuel affects the way it burns A solid fuel in a vertical position will allow fire spread more rapidly than the same fuel in a horizontal position 3 PHYSICAL STATES OF MATTER Liquid fuels Fuel gases are generated by a process called vaporization Vaporization: The transformation of a liquid to its vapor or gaseous state Energy input usually in the form of heat Requires less energy than said fuels With liquids, the surface to volume ratio is important 3 PHYSICAL STATES OF MATTER Gaseous fuels Can be the most dangerous of all fuel types because they are already in the natural state required for ignition Must be mixed with air in the proper proportion to burn; i.e. flammable range PHASES OF FIRE Incipient/ignition 2-2.8.1 Occurs when the four elements of the fire tetrahedron come together and combustion begins Can be caused by a spark or flame Can occur when a material reaches its ignition temperature through selfheating Limited to original materials ignited Small quantity of fire gases being generated Flame temperature above 1000 degrees F. yet room temperature is only slightly increased Easiest to extinguish PHASES OF FIRE Growth/freeburning 2-2.8.2 Fire plume begins to form above the burning fuel Begins to draw air from the surrounding space into the plume Hot gases rise, hit the ceiling and spread until they reach the walls As fire grows, the overall temperature increases PHASES OF FIRE Rollover/flameover 2-2.8.3 The ignition of combustible gases which have spread throughout the fire area Differs from flashover in that only combustible gases are burning One reason why firefighters stay low when entering a burning building Controlled by extinguishing main body of fire Flame spread movement of flame away from source of ignition PHASES OF FIRE Flashover Transition between the growth stage and fully developed stage of a compartment fire Occurs when flames flash over the entire surface of a room Occurs as a result of all the materials in the room reaching their ignition temperatures Involves all exposed combustible surfaces in the compartment Temperatures range from 900 degrees F-1200 degrees F. Survivability unlikely if caught in a flashover PHASES OF FIRE Fully developed All combustible materials in the compartment are involved Releasing the maximum amount of heat and producing large amounts of fire gases Hot unburned gases are flowing from the compartment and igniting when they enter a space with abundant air PHASES OF FIRE Decay/Hot smoldering fire 2-2.8.6 As fuel is consumed, the rate of heat decreases Amount of fire diminishes and temperature begins to decline Glowing embers can maintain moderately high temperatures PHASES OF FIRE Backdraft (smoke explosion) Explosion or rapid burning of heated gases Occurs when oxygen is introduced into a smoldering fire Often caused by improper ventilation Warning signs: Pressurized smoke exiting small openings Dense gray-yellow smoke Confinement and excessive heat Little or no visible flame Smoke leaving building in “puffs” (smoke puffing out and then sucking back in) Smoke stained windows Muffled sounds PHASES OF FIRE Backdraft cont. Warning signs Sudden rapid inward movement of air when an opening is made Situation can be made less dangerous by proper ventilation. Open at highest point involved. Heated smoke and gases will be released, reducing the possibility of an explosion THERMAL LAYERING The tendency of gases to form into layers according to temperatures Sometimes referred to as heat stratification or heat balance Hottest gases tend to be at the ceiling and cooler gases towards the floor Thermal layering is critical to firefighting operations HOW TO AVOID DISTURBING THE THERMAL LAYERING Thermal layering can be disrupted if water is applied directly into the layer without proper ventilation Results in higher temperatures at the floor level and decreased visibility. Firefighters may suffer steam burns if thermal layering is disrupted Homework Match types and states of energy to their definitions. Write the correct letters on the blanks. _____ 1. Energy developed when electrons flow through a conductor _____ 2. Energy possessed by a moving object _____ 3. Energy released when atoms are split _____ 4. Visible radiation produced at the atomic level _____ 5. Energy an object possesses that can be released in the future A. Mechanical energy D. Light B. Fusion C. Potential energy E. Electrical energy Homework Match heat and temperature terms to their definitions. Write the correct letters on the blanks. _____ 6. The amount of heat required to raise the temperature of 1 pound of water 1 degree Fahrenheit _____ 7. The amount of work done by a force of 1 newton through a distance of 1 meter _____ 8. The relationship between the calorie and the joule _____ 9. The amount of heat required to raise the temperature of 1 gram of water 1 degree Celsius _____ 10. The unit of temperature measurement in the customary system A. Degrees Fahrenheit B. Mechanical equivalent of heat C. Joule D. British thermal unit E. Calorie Homework Distinguish among the three methods of heat transfer. Mark “A” for examples of conduction, “B” for examples of convection, and “C” for examples of radiation. A = Conduction B = Convection C = Radiation _____ 11. A basement fire heats pipes enough to ignite the wood inside walls several rooms away. _____ 12. Fire spreads from the first to the third floor up a stairwell. _____ 13. The heat from a burning building ignites a second building 20 feet away. _____ 14. Hot water heats a radiator. _____ 15. A firefighter testing hose on the ramp receives a sunburn. _____ 16. The ventilation team opens the roof of a burning building and smoke and flames issue from the opening. Homework Match properties of matter to their definitions. Write the correct letters on the blanks. _____ 17. Mass, size, or volume _____ 18. How tightly the molecules of a solid substance are packed together _____ 19. Solid, liquid, or gas _____ 20. The density of a gas in relation to air _____ 21. The ratio of the mass of a given volume of liquid to the mass of an equal volume of water a. Density b. Specific gravity d. Physical state c. Physical property e. Vapor density Identify chemical reactions. Match the name of the described chemical reaction the letter provided. ___ 22. The formation of a chemical bond between oxygen and another element ___ 23. Alters the chemical makeup of a substance ___ 24 . Does not alter the chemical makeup of a substance ___ 25. Gives off energy as it occurs ___ 26. Absorbs energy as it occurs A. Physical change B. Chemical change C. Exothermic reaction D. Endothermic reaction E. Oxidation Homework Match the name of the described types of oxidation the letter provided ___ 27. Rapid oxidation ___ 28. Slow oxidation ___ 29. Instantaneous oxidation a) Explosion b) Fire c) Rust HOMEWORK A.Fuel B.Oxygen C.Chemical Reaction D.Heat 30. ____ 32. ____ 31. ____ 33. ____ Homework Select facts about oxidizing agents. Write the appropriate letters on the blanks. ___ 34. Which of the following is not a likely characteristic of an oxygen-enriched atmosphere? a. Materials burn more rapidly b. Ignition requires higher temperatures c. Some petroleum-based materials will autoignite d. Some materials will burn vigorously even though they do not burn at normal oxygen levels ___ 35. Which of the following fuels could burn in an oxygen-free atmosphere? a. Methane b. Hydrocarbon liquid c. Alcohol d. Sodium nitrate ___ 36. Oxygen concentrations as low as ___ percent can support combustion at room temperature. a. 21 b. 19 c. 18 d. 14 ___ 37. What percent oxygen does atmospheric air normally contain? a. 17 b. 21 c. 28 d. 12 Homework Identify a each false statement and mark with letter “B” and each true statement with the letter “A”. ___ 38. The shape and size of a fuel affects its ignitability. ___ 39. As a fuel's surface-to-mass ratio decreases, its ignitability increases. ___ 40. When a solid fuel is in a vertical position, fire spread is more rapid than when it is in a horizontal position. ___ 41. Liquid fuels have physical properties that increase the difficulty of extinguishment and the hazard to personnel. ___ 42. Volatility is the ease with which a liquid gives off vapor. ___ 43. Flammable limits — how rich or how lean the fuel vapor can be and still burn — are recorded in handbooks and are usually reported at ambient temperatures and atmospheric pressures. Homework Identify stages of compartment fire development. Match the name of the stage described in the blank provided. ___ 44. The fire becomes fuel controlled, the amount of fire diminishes, and temperatures within the compartment begin to decline. ___ 45. The combustible materials in the compartment and the gases given off by pyrolysis ignite. ___ 46. Rising hot gases hit the ceiling and spread outward until they hit the compartment walls; the depth of the gas layer begins to increase and causes radiant heating of combustible materials. ___ 47. The four elements of the combustion tetrahedron come together; can be piloted or non-piloted. ___ 48. All combustible materials in the compartment are involved in fire, releasing the maximum amount of heat possible and producing large volumes of fire gases. a. Ignition b. growth c. flashover d. fully developed e. decay Homework Select facts about the products of combustion. Write the appropriate letters on the blanks. ___ 49. Which of the following does not happen to a fuel when it burns during incomplete combustion? A. It undergoes a chemical change B. It releases gases and liquids C. The elements making up the fuel are destroyed D. Some of it is converted to energy ___ 50. Which of the following products of combustion would not be present in a smoldering fire? a. Fire gases b. Smoke c. Heat d. Flame ___ 51. What happens to the flame as combustion becomes more complete? a. It becomes cooler and less luminous b. It becomes cooler and more luminous c. It becomes hotter and less luminous d. It becomes hotter and more luminous ___ 52. What is the most common of the hazardous substances contained in smoke? a. Carbon monoxide b. Chlorine c. Hydrogen cyanide 4. Carbon dioxide ___ 53. What causes the most deaths in fires? a. Radiant heat b. Smoke c. Contact with heat conductive surfaces d. Direct flame contact Homework Select facts about fire extinguishment theory. (1 pt. each, 4/5) 54. What type of extinguishment theory does flooding an area with carbon dioxide illustrate? a. Fuel removal b. Chemical flame inhibition c. Oxygen dilution d. Temperature reduction 55. Which of the following illustrates extinguishment by fuel removal? a. Flooding an area with an inert gas b. Establishing a negative heat balance c. Blanketing with foam d. Allowing the fire to burn until all fuel is consumed 56. Which of the following is an example of an extinguishing agent used to interrupt the chemical chain reaction and stop flaming? a. Carbon dioxide b. Halon c. Water d. Class A foam 57. Which of the following fuels cannot be easily extinguished through chemical flame inhibition? a. Low flash point liquids and flammable gases b. Solid flaming fuels c. High flash point liquids d. Solid smoldering fuels 58. Which of the following is a polar solvent? a. Alcohol b. Gasoline c. Petroleum jelly d. Motor oil Homework Match fire classes to their descriptions. Write the correct letters on the blanks. _____ 59. Energized electrical equipment _____ 60. Ordinary combustibles _____ 61. Flammable and combustible liquids and gases _____ 62. Combustible metals A. Class A B. Class B C. Class C D. Class D Homework Match fire classes to their primary extinguishment methods. Write the correct numbers on the blanks. _____ 63. The application of non-conducting extinguishing agents such as halon, dry chemicals, or carbon dioxide _____ 64. Blanketing and smothering with fuel-specific extinguishing agents (usually dry powder agents) _____ 65. Cooling or quenching with water or “wet water” foams _____ 66. Smothering, blanketing, or oxygen exclusion with various extinguishing agents A. Class A B. Class B C. Class C D. Class D