BioUT.1.2.Biochemistry
advertisement

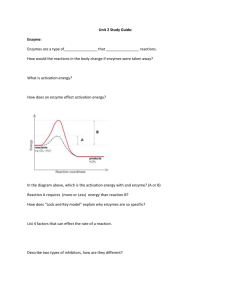
Biochemistry Biology 20 Chemistry of Life • 98% to 99% of living matter is made up of 6 atoms – Carbon, Oxygen, Hydrogen, Nitrogen, Phosphorus and Sulphur • Metabolism is the sum of all chemical reactions • that take place within the cell The four most important organic compounds are: – Proteins, Carbohydrates, Lipids and Nucleic Acids Types of Proteins • Proteins function in the growth, repair and • protection of living tissue Special Proteins in the human body include: Name Antibodies Enzymes Hormones Function Immune Response Catalyse Reaction Chemical Messengers Example Antibody A Sucrase Insulin Protein Structure • Proteins are built from 20 different types of amino acids – 8 essential amino acids (obtained from food) • AA joined by peptide bonds • Polypeptide Chains Proteins Proteins can be almost any 3D shape, therefore the often work by using a lock and key fit Chemistry of Proteins Leucine • Chemically composed of C, H, O and N • Broke down for energy after carbohydrates and lipids • (Anorexia) Deamination – breakdown of proteins into amino acids – Proteins Ammonia Urea Urine (Poisonous) (Safe) • Denaturation – a temporary change in the shape of • protein do to expose to extreme heat, acidity or radiation Coagulation – permanent change in the shape of a protein. Formation and Breaking of Chemical Bonds • Dehydration Synthesis – • • removing water in order to form a new bond Hydrolysis – using water to aid in the breaking of a bond Dehydration Synthesis and Hydrolysis are used to join or break apart proteins, carbohydrates and lipids Characteristics of a Carbohydrate • Function: The most readily available source of energy for our body. • Chemically composed of Carbon, Hydrogen and Oxygen in 1:2:1 ratio. C6h12O6 - glucose Types of Carbohydrates • Monosaccharide – Simple sugars – Glucose (fuel for the human body), fructose, ribose • Disaccharides – Combination of two monosaccharides. – Sucrose (table sugar) – glucose and fructose • Polysaccharides – Formed from long chains of monosaccharides (joined by dehydration synthesis) – Stored form of glucose in plants and animals • Starch – stored form of glucose in plants • Cellulose – component of the cell wall, cannot be digested in humans. • Glycogen – stored for of glucose in humans Characteristics of Lipids • Lipids are used for energy, storage • • • and cushioning. Lipids compose the cell membrane, help with the absorption of vitamins and help build hormones. Chemically composed of carbon, hydrogen and oxygen but in no fixed ratio. Composed of glycerol and fatty acids (joined by dehydration synthesis) Types of Lipids • Monoglycerides – Composed of 1 glycerol and 1 fatty acid • Diglycerides – Composed of 1 glycerol and 2 fatty acids • Triglycerides – Composed of 1 glycerol and 3 fatty acids Case Study – Fats and Health Pg 248 Nucleic Acids • Building blocks of the genetic code • Forms the DNA which in turn is the • molecule that makes up your chromosome (DNA – deoxyribose nucleic acid) Composed of 4 nitrogen bases – Adenine Thymine – Cytosine Guanine • Nucleotide – Sugar/Phosphate backbone and nitrogen base Test Substances • Benedict’s Solution – tests for monosaccharides – Blue Orange/Red • Iodine – tests for starches – Amber Blue/Black • Beuret’s Reagent – tests for peptide bonds (proteins) – Blue Pink/Purple • Sudan IV Solution – tests for lipids – Clear Pink/Red • Translucence test (brown bag) – tests for lipids – Bag becomes transparent The Importance of Water • • • • • • • • • • • • Absorbs and releases energy (moderates temperature) Is the medium in which metabolic reactions take place in organisms Is an excellent solvent Composes about 60% of the cells mass Cushions the body organs Lubricates body parts – joints Cleaning – tears and saliva Important for the breakdown of chemicals – hydrolysis and dehydration synthesis Dissolves particles for transportation Provides H+ and OH- for acid-base reactions It comprises 70% of the human body Covers two-thirds of out planet ( 97% salt water, 3% fresh water) Characteristics of Water • Occurs in all there states: liquid, gas, solid • High freezing point (o degree Celsius) and high boiling point • • • • • (100 degree Celsius) High heat of vaporization – amount of energy needed to turn liquid into gas (cools our body – sweat) High heat of fusion – amount of energy needed to turn a solid into a liquid. Cohesion – tendency of like molecules to cling together Adhesion – tendency of unlike molecules to cling together (capillary action) Surface tension Acids and Bases • H2O H+ + OH• Producing acids and bases – – – Neutral substances H+ = OHAcidic substances H+ > OHBasic substances H+ < OH- • The pH scale • Buffers – Absorb excess fluctuations in the levels of acids and bases. Maintain a substance at a specific pH. • Neutralizers – Maintains the pH at 7. – – – Stomach ph 2.4 Blood pH 7 Vagina ph 4.5 • Examples Enzymes • Enzymes are special • • proteins that increase the speed (catalyze) of chemical reactions Enzymes are not used up in the chemical reaction. Examples of enzymes Enzyme Substrate Location Amylase Starch Mouth ATPase ADP Mitochondria Lactase Lactose Sm. Intestine Lipase Lipids Sm. Intestine Maltase Maltose Sm. Intestine Pepsin Proteins Stomach Enzyme - Lock and Key Fit • Substrate – the molecule that • • • fits with the enzyme Enzyme-Substrate complex – when a molecule and enzyme become locked together Product – end result of the reaction Active Site – the area on the enzyme in which the substrate attaches. Rate of Enzyme Activity • Factors that affect the rate of enzyme activity: – Temperature • 37 C is optimal • Colder or warmer they slow down • Denaturation at 45 C – Acidity • pH sensitive. Designed to function of specific pH – Substrate Concentration • Decrease substrate concentration, decrease enzyme activity and visa versa – End Product Concentration • As end product concentration increases enzyme activity will • decrease Some end product are poisonous in high concentration Coenzymes and Cofactors • Alter the active site so the enzyme will fit a new substrate. • Normally vitamins Competitive Inhibitors • Block the active site of an enzyme. • • Therefore prevents contact from the substrate Most competitive inhibitors are reversible and only temporarily block the active site. Cyanide – irreversible inhibitor. Prevents to production of ATP by the mitochondria.