citric acid cycle
advertisement

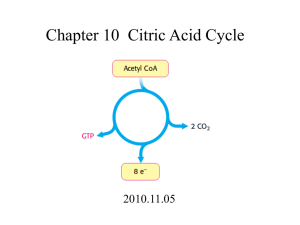
KAPITOLA 10 Energetický metabolismus III • katabolismus a anabolismus mastných kyselin • glyoxalátový cyklus • močovinový cyklus • lokalizace a fyziologické aspekty KAPITOLA 10 Energetický metabolismus III • Fatty acids catabolism and anabolism • Glyoxylate cycle • Urea cycle • Localization and physiological aspects !!! The glyoxylate cycle and its relationship to the citric acid cycle. The orange reaction arrows represent the glyoxylate cycle, and the blue arrows, the citric acid cycle. Notice that the glyoxylate cycle bypasses the two decarboxylation steps of the citric acid cycle, and that two molecules of acetyl-CoA enter the glyoxylate cycle during each turn, but only one enters the citric acid cycle. The glyoxylate cycle was elucidated by Hans Kornberg and Neil Madsen in the laboratory of Hans Krebs. The reactions of the glyoxylate cycle (in glyoxysomes) proceed simultaneously with, and mesh with, those of the citric acid cycle (in mitochondria), as intermediates pass through the cytosol between these compartments. Regulation of isocitrate dehydrogenase activity determines the partitioning of isocitrate between the glyoxylate cycle and the citric acid cycle. When isocitrate dehydrogenase is inactivated by phosphorylation (by a specific protein kinase), isocitrate is directed into biosynthetic reactions via the glyoxylate cycle; when the enzyme is activated by dephosphorylation (by a specific phosphatase), isocitrate enters the citric acid cycle, and ATP production results. Fatty acid entry into mitochondria via the acyl-carnitine/carnitine transporter. After its formation at the outer surface of the inner mitochondrial membrane, fatty acylcarnitine moves into the matrix by facilitated diffusion through the transporter. In the matrix, the acyl group is transferred back to CoA, freeing carnitine to return to the intermembrane space via the same transporter. The acyltransferase I and II enzymes are bound to the outer and inner surfaces, respectively, of the mitochondrial inner membrane. This entry process is the rate-limiting step for oxidation of fatty acids in mitochondria. Stages of fatty acid oxidation. Stage 1: A long-chain fatty acid is oxidized to yield acetyl residues in the form of acetyl-CoA. Stage 2: The acetyl residues are oxidized to CO2 via the citric acid cycle. Stage 3: Electrons derived from the oxidations of stages 1 and 2 are passed to O2 via the mitochondrial respiratory chain, providing the energy for ATP synthesis by oxidative phosphorylation. Regulation of metabolite flow from pyruvate through the citric acid cycle. The pyruvate dehydrogenase complex is allosterically inhibited at high [ATP]/[ADP], [NADH]/[NAD+], and [acetyl-CoA]/[CoA] rations, all of which indicate the energy-sufficent metabolic state. When these rations decrease, allosteric activation of pyruvate oxidation results. The rate of flow through the citric acid cycle can be limited by the availability of the substrates oxaloacetate and acetyl-CoA or by the depletion of NAD+ by its conversion to NADH, which slows the three oxidation steps for which NAD+ is the cofactor. Feedback inhibition by succinyl-CoA, citrate, and ATP also slows the cycle by inhibiting early steps. In muscle tissue, Ca2+ signals contraction and stimulates energy-yielding metabolism to replace the ATP consumed by contraction. The fatty acid oxidation (-oxidation) pathway. (a) In each pass through this sequence, one acetyl residue (shaded red) is removed in the form of acetyl-CoA from the carboxyl end of palmitate (C16), which enters as palmitoyl-CoA. (b) Six more passes through the pathway yield seven more molecules of acetyl-CoA, the seventh arising from the last two carbon atoms of the 16-carbon chain. Eight molecules of acetyl-CoA are formed in all. Oxidation of unsaturated FA Comparison of oxidation of fatty acids as it occurs in animal mitochondria and in animal and plant peroxisomes. The peroxisomal system differs in two respects: 1) in the first oxidative step electrons pass directly to O2, generating H2O2, and 2) the NADH formed in oxidation cannot be reoxidized, and the peroxisome must export reducing equivalents to the cytosol. (These eventually are passed on to mitochondria.) Fatty acid oxidation in glyoxysomes occurs by the peroxisomal pathway. In mitochondria, acetyl-CoA is further oxidized via the citric acid cycle. Acetyl-CoA produced by peroxisomes and glyoxysomes is exported; the acetate from glyoxysomes serves as a biosynthetic precursor. Electrons removed from fatty acids during oxidation pass into the mitochondrial respiratory chain and eventually to O2. The structures I through IV are enzyme complexes that catalyze portions of the electron transfer to oxygen. Fatty acyl-CoA dehydrogenase feeds electrons into an electrontransferring flavoprotein (ETFP) containing an iron-sulfur center, which in turn reduces a lipidsoluble electron carrier, ubiquinone (UQ, or coenzyme Q). -Hydroxyacyl-CoA dehydrogenase transfers electrons to NAD+, and the resulting NADH is reoxidized by NADH dehydrogenase (Complex I of the respiratory chain). Propionate produced from odd-chain fatty acids is converted to succinate. Succinate dehydrogenase, which acts in the citric acid cycle, feeds electrons into the respiratory chain at Complex II. Cytochrome c (cyt c) is a soluble electron carrier that transfers electrons between Complexes III and IV. Formation of ketone bodies from acetyl-CoA. Under circumstances that causes acetyl-CoA accumulation (starvation or untreated diabetes, for example), thiolase catalyzes the condensation of two acetyl-CoA molecules to acetoacetyl-CoA, the parent of the three ketone bodies. These reactions all occur within the mitochondrial matrix. The six-carbon compound -hydroxy- -methylglutaryl-CoA (HMG-CoA) is also an intermediate of sterol biosynthesis, but the enzyme that forms HMG-CoA in that pathway is cytosolic. HMG-CoA lyase is present in the mitochondrial matrix but not in the cytosol. The role of oxidation in the conversion of seed triacylglycerols into glucose in germinating seeds. Ketone body formation and export from the liver. Conditions that increase gluconeogenesis (diabetes, fasting) slow the citric acid cycle (by drawing off oxaloacetate) and enhance the conversion of acetylCoA to oxaloacetate. The released coenzyme A continued oxidation of fatty acids. The acetyl-CoA carboxylase reaction. Acetyl-CoA carboxylase has three functional regions; biotin carrier protein; biotin carboxylase, which activates CO2 by attaching it to a nitrogen in the biotin ring in an ATP-dependent reaction; and transcarboxylase, which transfers activated CO2 from biotin to acetyl-CoA, producing malonyl-CoA. The long, flexible biotin arm carriers the activated CO2 from the biotin carboxylase region to the transcarboxylase active site, as shown in the diagrams below the reaction arrows. The four-step sequence used to lengthen a growing fatty acyl chain by two carbons. Each malonyl group and acetyl (or longer acyl) group is activated by a thioester that links it to the fatty acid synthase, a multienzyme complex. 1) The first step is the condensation of an activated acyl group (an acetyl group is the first acyl group) and two carbons derived from malony-CoA, with the elimination of CO2 from the malonyl group; the net effect is extension of the acyl chain by two carbons. The b-keto product of this condensation is then reduced in three more steps nearly identical to the reactions of oxidation, but in the reverse sequence: 2) The -keto group is reduced to an alcohol 3) The elimination of H2O creates a double bond, and 4) The double bond is reduced to form the corresponding saturated fatty acyl group. Subcelllar localization of lipid metabolism in yeast and in vertebrate animal cells differs from that in higher plants. Fatty acid synthesis takes place in the compartment in which NADPH is available for reductive synthesis (i.e., where the [NADPH]/[NADP+] ratio is high). The acetyl group shuttle for transfer of acetyl groups from mitochondria to the cytosol for fatty acid synthesis. (The outer mito-chondrial membrane is freely permeable to all of these compounds.) Acetyl groups pass out of the mitochondrion as citrate; in the cytosol they are delivered as acetylCoA for fatty acid synthesis. Malate returns to the mitochondrial matrix, where it is converted to oxaloacetate. An alternative fate for cytosolic malate is oxidation by malic enzyme to generate cytosolic NADPH; the pyruvate produced returns to the mitochondrial matrix. Overwiew of the catabolism of aminoacids. The separate path taken by the carbon skeleton and the amino groups are emphasized by the bold divergent arrow. The glucose-alanine cycle. Alanine serves as a carrier of ammonia equivalents and of the carbon skeleton of pyruvate from muscle to liver. The ammonia is excreted, and the pyruvate is used to produce glucose, which is returned to the muscle. The "Krebs bicycle", composed of the urea cycle on the right, which meshes with the asparateargininosuccinate shunt of the citric acid cycle on the left. Fumarate produced in the cytosol by argininosuccinate lyase of the urea cycle enters the citric acid cycle in the mitochondrion and is converted in several steps to oxaloacetate. Oxaloacetate accepts an amino group from glutamate by transamination, and the aspartate thus formed leaves the mitochondrion and donates its amino group to the urea cycle in the argininosuccinate synthetase reaction. Intermediates in the citric acid cycle are boxed here. The urea cycle and the reactions that feed amino group into it. Note that the enzymes catalyzing these reactions are distributed between the mitochondrial matrix and the cytosol. One amino group enters the urea cycle from carbamoyl phosphate (step 1), formed in the matrix; the other (entering at step 2) is derived from aspartate, also formed in the matrix via transamination of oxaloacetate and glutamate in a reaction catalyzed by aspartate aminotransferase. The urea cycle itself consists of four steps: 1) Formation of citrulline from ornithine and carbamoyl phosphate. Citrulline passes into the cytosol 2) Formation of argininosuccinate through a citrullyl-AMP intermediate 3) Formation of arginine from argininosuccinate. This reaction releases fumarate, which enters the citric acid cycle 4) Formation of urea. The arginase reaction also regenerates the starting compound in the cycle, ornithine. The nitrogen cycle. The total amount of nitrogen fixed annually in the biosphere exceeds 1011kg. Overview of amino acid biosynthesis. Precursors from glycolysis (red), the citric acid cycle (blue),and the pentose phosphate pathway (purple), and the amino acids derived from them are boxed in the corresponding colors.. http://www.brookscole.com/chemistry_d/templates/student_resources/shared_resources/animations/oxidativ e/oxidativephosphorylation.html