Thermochemistry Podcast #2 - Mr. Wood
advertisement

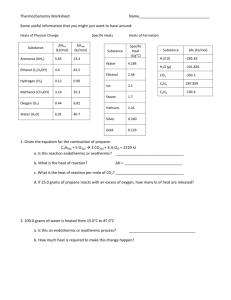
Thermochemistry Podcast #2 Heats of Reaction Heats of Formation Hess’ Law Heat of Solution When a substance dissolves in water energy can be absorbed or released. This is quantity of energy is called the molar heat of solution and is represented by __________ Energetically, solution formation is like a chemical reaction. Sample Problem #1 What is the heat change when 80 g of NaOH dissolves in water? (DHsol=-44.51 kJ/mol) From the data presented below, determine the heat of solution for bengiovannium (molar mass = 217 g/mol). Heat of Formation DHf is the energy change during formation of the substance from its elements. Heats of formation are usually listed on a table. (pg 975 in your book) DHf for elements = ___________ Heat of Reaction DHr can be determined using this equation: A __________ DHr indicates an ___________ reaction A __________ DHr indicates an ___________ reaction Hess’ Law Hess's Law states that "the change in enthalpy for any chemical reaction is constant, whether the reaction occurs in one step or in several steps." Thermochemical data, therefore, may be treated algebraically. Heat of Reaction Sample Problem H2S(g) 4F2(g) 2HF(g) + SF6(g) Determine the DHr for the reaction above. DHf(H2S) = -21 kJ/mol DHf(HF) = -273 kJ/mol DHf(SF6) = -1220 kJ/mol. Pg 572 The enthalpy change for the combustion of methane can be evaluated for the path that decomposes the reactants into elements and then recombines those elements to form the products. A state function does not depend on the path you choose to go from the initial state to the final conditions. Hess’ Law Allows us to measure the heat of reaction indirectly by adding reactions together that have a known DHr Hess's law of heat summation the law that states that if you add two or more thermochemical equations to give a final equation, then you also add the heats of reaction to give the final heat of reaction Hess’ Law Example C + 1/2 O2 CO DHr° = -110 kJ/mol CO + 1/2 O2 CO2 DHr° = -283 kJ/mol C + O2 CO2 DHr° = = Algebra and Hess’ Law Whatever is done to one side of an equation, must be done to the other. If a chemical equation is doubled, tripled, etc. . . then the enthalpy of that reaction is doubled, tripled, etc. . . If a chemical equation is reversed, then the enthalpy is reversed by changing the sign from negative to positive (or vice versa) Hess’ Law Example 2 • Find the heat of formation for: • C(graphite) + 2H2(g) CH4(g) ΔHf = _____ kJ • C(graphite) + O2(g) CO2(g) • H2(g) + ½O2(g) H2O(l) • CH4(g) + 2O2(g) CO2(g) + 2H2O(l) ΔH = -393.5 kJ ΔH = -285.8 kJ ΔH = -890.3 kJ Hess’ Law Practice Find 4 NH3 (g) + 5 O2 (g) 4 NO (g) + 6 H2O (g) using the data below. N2 (g) + O2 (g) 2 NO (g); DHf = 180.6 kJ N2 (g) + 3 H2 (g) 2 NH3 (g); DHf = -91.8 kJ 2 H2 (g) + O2 (g) 2 H2O (g); DHf = -483.7 kJ More Hess’ Law Practice Calculate ΔH for the reaction: CS2(l) + 2H2O(l) CO2(g) + 2H2S(g) Given: H2S(g) + 3/2O2(g) H2O(l) + SO2(g) CS2(l) + 3O2(g) CO2(g) + 2SO2(g) ΔH = +50.0 kJ ΔH = ? kJ ΔH = -562.6 kJ ΔH = -1075.2 kJ More Hess’ Law Problems Calculate ΔHf for C2H2(g) 2C(s) + H2(g) C2H2(g) Given the following thermochemical equations: C2H2(g) + 5/2O2(g) 2CO2(g) + H2O(l) ΔH = -1299.5 kJ C(s) + O2(g) CO2(g) ΔH = -393.5 kJ H2(g) + 1/2O2(g) H2O(l) ΔH = -285.8 kJ ΔH = +226.7 kJ