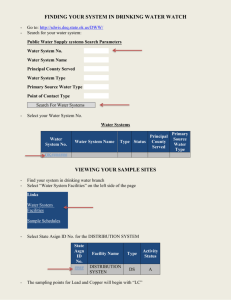
Real-time Monitoring with a
Portable Miniaturized Surface
Plasmon Resonance System
Clement E. Furlong, Research Professor, Departments
of Medicine (Div. Medical Genetics) & Genome Sciences
University of Washington, Seattle, WA
Presented by: Brian Marquardt CPAC/UW
Spreeta sensing components
• Spreeta SPR components
developed in collaboration with
UW with TI
• Miniaturized, robust, high
performance devices.
• Inexpensive in large quantity
• Excellent manufacturing
capabilities and quality control
Each Spreeta chip contains
all of the optical
components needed for
sensitive SPR measurement
of biomolecular interactions
The SPIRIT system
(Surface Plasmon Instrumentation for the Rapid Identification of Toxins)
• Compact, lightweight
(lunchbox size, 6 lb.)
• High performance
• 24 simultaneous
measurements
• Low power (5W)
allows portable
operation
• Automated
Current laboratory
prototype
Touchscreen data display
Sensor channel
Detected levels
(bargraph)
Selected SPR curve
Detected levels
(numeric)
Sensor surface chemistry
Each Spreeta chip has 3
useable channels
Soluble protective coating
Control receptors
(dextran/trehalose)
allows long-term dry storage at
room temperature
(usually antibodies)
Designed NOT to
respond to that agent
Target receptors:
(usually antibodies)
Designed to capture
a specific agent or
analyte e.g.:
•Toxins
•Viruses
•Spores
•Bacteria
YYYYY
YYYYY
Gold layer (50 nM)
Glass substrate
Spreeta
sensor chip
1
Fundamentals of
Reflectivity
0.8
Surface Plasmon Resonance
0.6
0.4
0.2
0
0
20
40
60
80
Ө Degrees
System software
Refractive Index
1.3377
1.3376
1.3375
Sensorgram
1.3374
1.3373
1.3372
1.3371
1.337
1.3369
1.3368
1.3367
0
5
Time, Min
10
SPIRIT performs 24 simultaneous
measurements of antibody
binding
Detection event
Eight sensor chips
Analyte
Three active spots per sensor
Flowcell
Examples of Assays Possible with SPR
• Whole microbial cells
-(F.tularensis, E. coli, Y. pestis)
• Spores
-(e.g., anthrax)
• Viruses with or without amplification
-(e.g. Norwalk, flu)
• Proteins by direct detection with or without
amplification/verification
-(protein toxins, industrial proteins, therapeutics)
• Small molecular weight analytes using displacement or
competition assays
-(e.g., domoic acid, cortisol, insecticides, toxic chemicals, TNT & other small
organics)
Detection of Larger Analytes
• Microbes
• Spores
• Viruses
• Proteins/Toxic Proteins
Analyte Detection and Signal Amplification
Refractive index
1.3382
1.3380
1.3378
1.3376
1.3374
1.3372
1.3370
1.3368
0
50
100
Time, min
Signal Detection
150
Analyte Detection and Signal Amplification
Refractive index
1.3382
1.3380
1.3378
1.3376
1.3374
1.3372
1.3370
1.3368
0
50
100
Time, min
Signal Detection
150
Analyte Detection and Signal Amplification
Refractive index
1.3382
1.3380
1.3378
1.3376
1.3374
1.3372
1.3370
1.3368
0
50
100
Time, min
Signal Detection
150
Analyte Detection and Signal Amplification
Refractive index
1.3382
1.3380
1.3378
1.3376
1.3374
1.3372
1.3370
1.3368
0
50
100
Time, min
Signal Detection
150
Detection of Microbes
Detection and Verification of F. Tularensis (105 cfu/ml)
Amplification/verification
Relative Refractive Index
1.3396
Detection
1.3394
anti-F.T #1
anti-F.T #2
anti-F.T. #3
anti-Bot A NT #1
anti-Bot A NT #2
anti-Bot A NT #3
Active channels
1.3392
1.3390
Reference channels
1.3388
0
2
4
6
8
11
13
15
Time (min)
17
19
21
23
Virus Detection
Amplification
Detection of Staphylococcal
Enterotoxin B
9.0E-05
1.33525
8.0E-05
SEB binding rates, RIU/min
Sensor response, RIU
1.33520
1.33515
1.33510
1.33505
1.33500
7.0E-05
6.0E-05
5.0E-05
1.0E-05
4.0E-05
8.0E-06
6.0E-06
3.0E-05
4.0E-06
2.0E-05
2.0E-06
0.0E+00
1.0E-05
0
1
2
3
0.0E+00
1.33495
0
20
40
Time, min
60
80
0
20
40
60
80
SEB concentration, nM
100
4
Detection of 5 ng/mL (5 ppb; 33pM)
BotNT (denatured botulinum toxin)
1.3323
1.33228
1.33226
1.33224
1.33222
1.3322
1.33218
1.33216
1.33214
1.33212
1.3321
Anti-Bot-toxin
Reference
RI
Detect
0
Am plify
10
20
Time (min)
30
40
Binding Rate (x10-6RIU/min)
Background-subtracted RIU
Direct Detection of Ricin A
Chain (64 ppb-320 ppb)
100 nM Ricin A Chain
0.00009
50 nM Ricin A Chain
20 nM Ricin A Chain
No Ricin A Chain
0.00007
0.00005
0.00003
0.00001
-0.00001
0
200
400
600
Time (seconds)
800
1000
14
12
10
8
6
4
2
0
0
100
200
300
Ricin A Chain Concentration (nM)
400
Detection of Cortisol by Competition Assay
Cortisol Competition 2-24-04c
1.3389
BSA
BSA-Cortisol
HSA-GD
1.3388
1.3387
RIU
10 nM
750 pM
1 nM
1000 nM
Estriol
2 nM
5 nM
1.3386
Lower arrows indicate return
to no analyte
1.3385
1.3384
0
1000
2000
3000
Time (seconds)
4000
5000
Standard Domoic Acid Concentration
Curve in Clam Extracts
Other Useful Applications of SPR Sensing
• Nucleic Acid Analyses
• Many Other Molecular Interactions
Protein Nucleic Acids as Recognition
Elements for DNA/RNA
Very stable receptor on chip
(Protein Nucleic Acid)
Allows detection of target
Binding of a 79 bp DNA Probe to a Complementary PNA
16 mer on the Sensor Surface
Detection of Analytes in Complex Matrices
(e.g., saliva, plasma, urine, stool extracts, sea
water, fresh water, etc.)
Detection of 1 nM (28 ppb) SEB in
seawater
Staphylococcal enterotoxin B
1.33974
Refractive index,n
1.33973
1.33972
1.33971
1.3397
1.33969
1.33968
1.33967
1.33966
0
20
40
Time, min
60
80
Detection of 500 pM (14 ppb) SEB in urine
Amplification
500 pM SEB
Wash
(urine)
From: Naimushin et al., Biosensors and Bioelectronics 17:573
Detection of cortisol in saliva
using the compound flow cell
Reference-subtracted RIU
0.0074
Saliva plus 28 nM Cortisol
0.0073
0.0072
Saliva plus 14 nM
Cortisol
0.0071
Saliva only
0.007
0.0069
17
27
37
47
57
Tim e (m inute s)
67
77
Detection of Theophylline in Saliva
Using the Compound flow Cell
1.3425
Reference
1.34245
RIU
1.3424
Theophylline
Saliva plus 1 M
Theophylline
Saliva plus 2 M
Theophylline
1.34235
Saliva only
1.3423
1.34225
2000
2500
3000
3500
Time (seconds)
4000
Sequential Detection of 8 Analytes
200
150
Ovalbumin
10 ng/ml
100
F. tularensis 5
x 103 CFU/ml
Norwalk VLPs
5 x 109 particles/ml
SEB 5 ng/ml
RIU
Y. pestis
106 CFU/ml
50
Ricin A
chain 20
ng/ml
B. anthracis
5 x 106 CFU/ml
BG Spores
9 x 104
CFU/ml
0
-50
-100
0
20
40
60
80
100
Time (min)
120
140
160
180
SPIRIT Team & Sponsors
• Medical Genetics
Group:
Dr. Clement Furlong
Scott Soelberg
Dr. Gary Geiss
Dr. Rick Stevens
Steve Near
Matthew Probert
Joshua Probert
Nathaneal Swanson
Dr. Paul Baker
•
• Electrical Engineering
Group:
Dr. Sinclair Yee
Tim Chinowsky
Peter Kauffman
Jared Tritz
Michael Grow
Tony Mactutis
• Texas Instruments:
Jerry Elkind
Dwight Bartholomew
John Quinn
Sponsors:
DOD
Texas Instruments
Center for Process Analytical Chemistry (CPAC), UW, Seattle
Washington State Sea Grant, NIH/NIEHS
 0
0