significant figures

CHEMISTRY
July 30, 2013
Brain Teaser
Turn in Homework: Science Autobiography
Write your name and period number
Define Chemistry using your own words
What is the SI base unit for the following measurements (Example: Volume = Liter (L))
Length = ___________
Mass = __________
Temperature = ___________
Time = __________
Agenda
Brain Teaser
Notes:
Introduction to Chemistry
SI Units and Base Units
Significant Figures
Homework
Intro to Measurement Worksheet 1
Significant Figures Worksheet (?)
Unit 1 Objectives
Introduction to Chemistry
Define chemistry and matter
Units of measurement
SI Units
Base Units
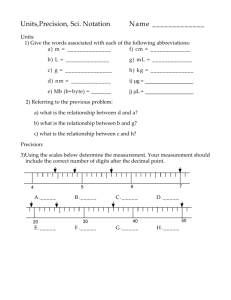
Accuracy versus Precision
Uncertainty in Measurement
Significant Figures
Significant Figures in Calculations
Dimensional Analysis
Introduction to Chemistry
Chemistry: The
Central Science
Chemistry is the science that investigates and explains the structure and properties of matter.
Seeks to explain the submicroscopic events that lead to macroscopic observations
Branches of Chemistry
Branch
Organic chemistry
Inorganic chemistry
Physical chemistry
Analytical chemistry
Biochemistry
Area of Emphasis Examples most carbon-containing chemicals pharmaceuticals, plastics in general, matter that does not contain carbon minerals, metals and nonmetals, semiconductors the behavior and changes of matter and the related energy changes reaction rates, reaction mechanisms components and composition of substances matter and processes of living organisms food nutrients, quality control metabolism, fermentation
Units of measurement
SI Units ( Le Systéme Internationale)
Scientists need to report data that can be reproduced by other scientists. They need standard units of measurement.
Base Units
• A base unit is a defined unit in a system of measurement
•There are seven base units in SI.
Base Units
SI Units and Base Units
Handouts
Kilo, centi, milli
SIGNIFICANT FIGURES
Significant Figures
Significant
Figures
Digits in a measurement that have meaning relative to the equipment being used
Significant Figures
Place What is the increment on the equipment?
What you know for sure.
Significant Figures
Digits with meaning
Digits that can be known precisely plus a last digit that must be estimated.
Scale Reading and Uncertainty
Uncertainty: Limit of precision of the reading (based on ability to guess the final digit).
Existed in measured quantities versus counted quantities
Refer to Example (2 rulers)
Ruler
http://www.funbrain.com/measure/
What are the units?
Graduated Cylinder
http://www.uwplatt.edu/chemep/chem/chemscape
/labdocs/catofp/measurea/volume/gradcyl/gradc yl.htm
What are the units?
Significant Figures
What do you notice?
Depends on type of equipment being used.
Depends on size of equipment used.
Summary
Things to consider
What do significant figures tell you about the measurement on the equipment?
If you wanted to measure the mass of a whale, what scale would you want to use? Would it matter if you know its mass accurately to 1 gram?
If you wanted to measure the mass a grain of sand , what scale would you want to use? Would it matter if you know its mass accurately to 1 gram?
Significant Figures
Raw Data Rules
How do you know how many sig figs?
1.
2.
3.
4.
5.
All digits 1-9 are significant.
Zeros between significant digits are always significant.
Trailing 0’s are significant only if the number contains a decimal point
Zeros in the beginning of a number with a decimal point are not significant.
Zeros following a significant number with a decimal are significant.
Significant Figures
Pacific to Atlantic
Rule
Pacific = Decimal Present
Start from the Pacific (left hand side), every digit beginning with the first 1-9 integer is significant
Examples
20.0 = 3 sig digits
0.00320400 = 6 sig digits
1000. = 4 sig digits
Significant Figures
Atlantic Rule to
Pacific
Atlantic = Decimal Absent
Start from the Atlantic (right hand side), every digit beginning with the first 1-9 integer is significant
Examples
100020 = 5 sig digits
1000 = 1 sig digits
Practice
2.
3.
1.
4.
How many significant figures are in
400.0
4000
4004
0.004
More Practice Problems
Determine the number of significant figures in the following:
1005000
1.005
0.000125
1000.
0.02002
2002
200.200
Review Questions
Determine the number of significant figures in:
72.3 g
60.5 g
6.20 g
0.0253 g
4320 g
0.00040230 g
4.05 x 10 5 g
4500. g
Homework
1.
2.
Measurement Worksheet 1
Significant Figures 1 Worksheet
THE END
Why do we use the metric system?
Advantages
Simple to use
Easy to convert from one unit to another
Dimensional Analysis
Universal – used worldwide
By all scientists to communicate
By all industrialized nations
Except United States
U.S. loses billions of dollars in trade
Mass versus Volume
Question:
What is the difference between mass and volume?
Introduction to Chemistry
• Chemistry: The
Central Science
• A more formal definition of
Matter is anything that takes up space and has mass
•
Mass is the measure of the amount of matter that an object contains
•
Volume is the amount of space an object takes up.
Example
Refer to the two identical spherical objects below
Bowling Ball
Volley Ball
Example
Which of the following weighs more?
A ton of feathers?
A ton of bricks?
Units of measurement
SI Units ( Le Systéme Internationale)
Scientists need to report data that can be reproduced by other scientists. They need standard units of measurement.
Base Units
• A base unit is a defined unit in a system of measurement
•There are seven base units in SI.
Base Units
Why do we use the metric system?
Advantages
Simple to use
Easy to convert from one unit to another
Dimensional Analysis
Universal – used worldwide
By all scientists to communicate
By all industrialized nations
Except United States
U.S. loses billions of dollars in trade
Example
Convert 3400 milliliters to liters
44 centimeters to meters
277 kilograms to grams
Accuracy Versus Precision
What is the difference between accuracy and precision?
Precision versus Accuracy
Precision: is a measure of how closely an individual measurements agree with one another
Can be precise but inaccurate
Accuracy: refers to how closely individual measurements agree with the correct, or “true” value
Examples using the dartboard
An archery target illustrates the difference between accuracy and precision.
Accuracy and Precision
Measurements
Scale Reading and Uncertainty
Uncertainty: Limit of precision of the reading (based on ability to guess the final digit).
Existed in measured quantities versus counted quantities
Example
Which electronic balance below allows you to obtain a more precise measurement? Why?
A B
Triple Beam Balance
http://www.wisconline.com/objects/index_tj.asp?objID=GCH202
What are the units?
Ruler
http://www.funbrain.com/measure/
What are the units?
Graduated Cylinder
http://www.uwplatt.edu/chemep/chem/chemscape
/labdocs/catofp/measurea/volume/gradcyl/gradc yl.htm
What are the units?
Measurement Activity
Objectives
Measure each physical quantity to the maximum accuracy allowed by the appropriate instrument.
Record measurements and calculations with the appropriate units from the international system of units (SI.)
Measurement Activity
Set up assignment
Title, Name, Chemistry, Period, Date
Station Rotation
10 Station
3-4 minutes at each station
Refer to Handout
Do NOT remove any items from the station
Clean Up and Restore each station before switching
Homework
Homework
“Is it worth the risk?” worksheet
Sign Lab Safety Contract
Study for Lab Safety Test (Monday)
Get Supplies for Chemistry
Closure
Reflect on your learning during the lab activity
Concepts learned?
“Ah-ha” moments?
Questions?
SIGNIFICANT FIGURES
Rules for zeros:
All zeros count except placeholder zeros
These are the ones that disappear when you write the number in scientific notation.
Zeros between nonzero digits are always significant
E.g. 1005 kg (4 sig. fig) and 1.03 (3 sig. fig)
Zeros at the beginning of a number are never significant
E.g. 0.02 (1 sig. fig) and 0.0026 (2 sig. fig)
Zeros at the end of a number are significant if the number contains a decimal point
0.0200g (3 sig. fig), 3.0 cm (2 sig. fig), 5000 (1 sig. fig)
Rules for recognizing significant figures
• Non-zero numbers are always significant.
• Zeros between non-zero numbers are always significant.
• All final zeros to the right of the decimal place are significant.
• Zeros that act as placeholders are not significant.
• Counting numbers and defined constants have an infinite number of significant figures.
Review Questions
Determine the number of significant figures in the following:
1005000
1.005
0.000125
1000.
0.02002
2002
200.200
Review Questions
Determine the number of significant figures in:
72.3 g
60.5 g
6.20 g
0.0253 g
4320 g
0.00040230 g
4.05 x 10^5 g
4500. g
Closure
Reflect on your learning today
Homework
Measurement Worksheet 2
Science Safety Test
Wednesday = TOMORROW
Element Quiz
Thurs/Fri
Lab Notebook
Quadrille Lab Notebook (Graphing paper) OR
Carbonless Copy Lab Notebook
Topics on Unit 1 Test
Lab Safety
Measurements
SI units
Significant Figures, Uncertainty
Accuracy and Precision
Understanding Equipments
Dimensional Analysis