SCH4U -- Unit B -- Intro to Orgo
advertisement

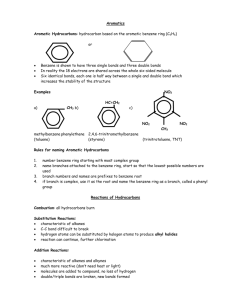
Intro to organic chemistry (orgo) SCH4U – Unit B Organic Compounds • Organic chemistry: chemistry of carbon compounds ▫ Exceptions are oxides of carbon (CO2 and CO), carbonates, bicarbonates, and cyanides • Carbon atoms are generally bonded to: ▫ Each other ▫ Hydrogen atoms ▫ Other specific elements (O, N, S, P and others) Why study organic chemistry? 1. Life is carbon-based. Everyone is made up of organic compounds. There are more known carbon compounds than any other element. 2. There are several million known organic compounds with ~30,000 more being “discovered” each year. 3. Some common organic compounds include plastics, synthetic and natural fibres, dyes, drugs, pesticides, lighter fluid, and gasoline. The carbon atom • Carbon has four valence electrons allowing it to form four bonds with other atoms by sharing its electrons ▫ This results in a tetrahedral shape • Carbon forms covalent bonds (sharing of electrons) with carbon atoms Classification • Since organic compounds are composed almost entirely of carbon, they are sometimes referred to as hydrocarbons • Hydrocarbons can be divided into: 1. Aliphatics: carbon atoms in an open chain structure; may have branches, but no rings 2. Alicyclics: carbon atoms arranged in a ring structure 3. Aromatics: structures related to benzene (C6H6) Classification • Aliphatics can be further categorized into: 1. Alkanes: contains only single bonds (saturated) 2. Alkenes: contains at least one double bond (unsaturated) 3. Alkynes: contains at least on triple bond Isomers Isomers Constitutional Isomers Stereoisomers Diastereomers Enantiomers Isomers Constitutional Isomers Stereoisomers Isomers have same molecular formula Isomers have same molecular formula Atoms bonded in a different sequence Atoms bonded in same sequence Also known as structural isomers Differ in 3D orientation of atoms in space Isomers Diastereomers Enantiomers Based on a double bond Mirror images of each other around a single carbon atom Carbon atoms with different types of atoms At least one carbon atom bonded to four different types of atoms or groups cis isomer when two identical groups or atoms are on the same side trans isomer when two identical groups or atoms are on the opposite side Diagrams & formulas • A number of different (but related) diagrams used to express structures in organic chemistry: ▫ ▫ ▫ ▫ ▫ ▫ Empirical molecular formula Expanded molecular formula Complete structural formula Condensed structural formula Line structural formula 3D structural formula Alkanes # of carbons Prefix # of carbons Prefix 1 Meth- 6 Hex- 2 Eth- 7 Hept- 3 Prop- 8 Oct- 4 But- 9 Non- 5 Pent- 10 Dec- Alkanes • Only single bonds present in the main chain • General formula is CnH2n+2 • Form a tetrahedral shape ▫ Note: more on shapes in the next unit • Create a 109.5° angle between bonds • Considered “saturated” because no other hydrogen atoms can be added • Always end in “~ane” Nomenclature • Three parts to organic chemistry nomenclature: ▫ Prefix: indicates number, type and location of branches and/or functional groups (if any) ▫ Root: indicates the number of carbon atoms in the main (parent) chain ▫ Suffix: indicates the number and location of double or triple bonds (if any) and/or highest priority functional group Let’s draw alkanes! • Draw all ten basic alkanes and name them. Alkyl groups and branching • Each branch of a hydrocarbon is referred to as an alkyl group (if it contains only C and H) • Alkyl groups use the same basic roots as alkanes • Alkyls always end in “~yl” • Also contain a number and hyphen in front to indicate location on the main carbon chain ▫ Example: 2-methyl pentane CH3 CH2 H3C CH CH2 CH3 Alkyl groups and branching • Find the longest carbon chain and assign sequential numbers to each carbon ▫ Assign these numbers temporarily starting from both ends of the carbon chain ▫ Ultimately will select only one set of numbers to use • The goal is to use the numbering along the carbon chain that gives the lowest values assigned to branches Alkyl groups and branching • Let’s try some! Draw 3-ethyl octane… CH2 CH3 H3C CH2 CH H3C CH2 CH2 CH2 CH2 Alkyl groups and branching Alkyl groups and branching • When there is more than one branch, they are listed in front of the root and suffix according to alphabetical order of the name of the alkyl group • Try naming… H3C CH3 CH2 HC • 4-ethyl 3-methyl heptane H3C CH CH2 CH2 CH2 CH3 Alkyl groups and branching • If there is more than one of any given alkyl group ▫ Indicate all the number for the positions using commas to separate the numbers, and ▫ Use prefixes (on the prefixes – confused yet?) to indicate the number present Alkyl groups and branching CH3 • Try naming… H2C CH3 CH3 CH2 H3C CH CH2 CH CH C CH3 CH2 H2C H3C CH3 • 5,6-diethyl-3,3,4-trimethyl nonane Alkenes • At least one double bond present in the main chain • General formula is CnH2n • Form a trigonal planar shape ▫ Note: more on shapes in the next unit • Create a 120° angle between bonds • Considered “unsaturated” because other hydrogen atoms can be added • Always end in “~ene” Nomenclature 1. Identify and name the longest carbon chain that CONTAINS THE DOUBLE BOND(S). 2. Number the carbons in the chain so that the two carbons involved in the double bond have the lowest possible numbers. a) If the double bond is equidistant from both ends, number the chain so that the substituent(s) have the lowest possibly number. 3. Follow same rules for identifying and naming prefixes. Nomenclature • Try naming this… H3C CH2 CH2 CH2 • 2-ethyl 1-pentene C CH2 CH3 Nomenclature • Now let’s draw 1-butene and 2-butene… CH3 CH2 CH2 H3C 1-butene CH CH CH H3C 2-butene Nomenclature • Let’s try a structure with more than one double bond… CH CH CH H3C 1,3-pentadiene CH2 Nomenclature • Let’s put everything together and name… CH3 CH3 H2C CH2 H3C CH CH2 C CH C C CH3 • 4-ethyl-3,6-dimethyl 1,2,4-octatriene Diastereomers • Consider the following compounds… CH3 CH CH CH H3C CH3 CH C CH3 H3C cis-2-butene H3C trans-2-butene CH2 2-methyl1-propene Diastereomers • Not superimposable upon one another • Result from lack of free rotation around the double bond • Priorities are assigned to the two groups attached to the carbon atoms on either side of the double bond (starting with the carbon to the left of the double bond) based on atomic number ▫ Largest atomic number at the first point of difference has the highest priority Diastereomers • If two highest priority groups are on the same side of the double bond (top or bottom)… ▫ cis configuration • If two highest priority groups are on the opposite side of the double bond… ▫ trans configuration Alkynes • At least one triple bond present in the main chain • General formula is CnH2n-2 • Form a linear shape ▫ Note: more on shapes in the next unit • Create a 180° angle between bonds • Considered “unsaturated” because other hydrogen atoms can be added • Always end in “~yne” Nomenclature 1. Identify and name the longest carbon chain that CONTAINS THE TRIPLE BOND(S). 2. Number the carbons in the chain so that the two carbons involved in the triple bond have the lowest possible numbers. a) If the triple bond is equidistant from both ends, number the chain so that the substituent(s) have the lowest possibly number. 3. Follow same rules for identifying and naming prefixes. Nomenclature • Try naming this… HC CH3 C HC CH • 3,4-dimethyl 1-hexyne H3C CH2 CH3 Nomenclature • Now let’s draw 1-butyne and 2-butyne… CH2 H3C C H3C C C CH 1-butyne 2-butene CH3 Nomenclature • Let’s try a structure with more than one triple bond… H3C C C C 2,4-hexadiyne C CH3 Nomenclature • Let’s put everything together and name… CH3 H3C CH2 CH C C C C C CH3 H3C • 4-ethyl-4,5-dimethyl 2,6-octadiyne CH3 Alicyclics • Connected end-to-end forming a ring • Have single and/or double bonds present in the main chain • General formula is CnH2n • Can be “saturated” or “unsaturated” • Suffix changes depending on presence of double bonds • Prefix used is always “cyclo~” before the root Nomenclature • Try naming… CH2 H2C CH2 H2C CH2 cyclobutane HC CH2 CH CH2 cyclo-1-pentene cyclopent-1-ene Nomenclature CH3 H2C • Try drawing… CH2 CH HC C HC CH CH CH2 CH C CH H3C CH2 cyclo-1,4-hexadiene CH CH 2,7-diethyl-4-methyl cyclo-1,3,5-heptatriene CH3 Aromatics • Based on benzene and the presence of benzenelike rings • Benzene is a six-carbon ring with three double bonds ▫ Cyclo-1,3,5-hexatriene • However, the electrons in the double bonds are actually spread over the whole molecule = delocalized ▫ Benzene actually has six identical “1 ½” bonds Aromatics • Creates greater stability in the molecule • These “1 ½” bonds do not behave/react like double bonds • Molecules that have this type of sharing are referred to as aromatic compounds CH Benzene CH HC CH HC CH HC CH HC CH CH CH Aromatics • Some common aromatics… CH CH HC CH HC CH CH HC C CH HC C CH CH CH benzene CH napthalene CH antracene CH CH HC C C CH HC C C CH CH CH CH Nomenclature 1. Number carbons in benzene ring assigning lowest number to a branch. a. If more than one branch exists, start numbering at the branch that contains the group with the highest priority (most complex). 2. Name branches attached to the benzene ring, assigning numbers to each branch. a. If only one branch is present, a number does not need to be assigned. 3. Branches are placed before the root as a prefix. Nomenclature • Priority of branches on benzene… Highest priority -OH -NH2 -F, -Cl, -Br, -I -CH2CH2CH3 -CH2CH3 Lowest priority -CH3 Nomenclature • When benzene has only two branches, position numbers do not have to be used • Instead, prefixes ortho-, meta-, and parareplace position numbers CH3 C 1,2-dimethyl benzene ortho-dimethyl benzene CH3 HC C HC CH CH Nomenclature CH3 C CH3 HC CH HC CH C HC CH C HC C CH CH3 CH3 1,3-dimethyl benzene 1,4-dimethyl benzene meta-dimethyl benzene para-dimethyl benzene Nomenclature • IUPAC has retained many of the common names for aromatics… Nomenclature CH2 • Let’s try naming… CH H3C C CH CH 3-ethyl-5-methyl phenol C C OH CH3 Nomenclature • Let’s try drawing… H3C CH2 C H3C O C CH2 C C CH CH CH 2,6-diethyl-3-methyl benzaldehyde CH3 Nomenclature • For use when: ▫ Naming compounds with >1 fxn’l group ▫ Naming compounds with a complicated substituent Nomenclature • Try naming… H3C CH2 CH2 CH CH HC 3-phenyl hexane C CH3 CH CH CH2 CH Nomenclature • Try drawing… CH HC CH HC C CH H3C 4,6-diphenyl 2-octene CH2 CH CH CH CH2 C CH HC CH HC CH CH CH3 Up next • That covers the basics of hydrocarbons in organic chemistry. • Next we will cover functional groups!