Lab Safety - Mr. Downing Science 10
advertisement

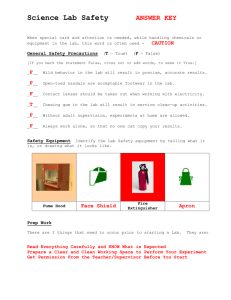
Chapter A1 – Particles & the structure of matter Safety in the laboratory Understanding the rules It’s not enough to remember the rules for conducting a lab experiment safely – you need to understand WHY those rules exist Like any lesson in science, you’ll remember it better if you Understand the importance of the concept, or Experience the concept first-hand Since I’d rather you not learn the importance of safety glasses by blinding yourself, focus on the WHY of the following rules. Rule #1 – Wear the proper safety equipment Rule #1 – Wear the proper safety equipment QUESTION: What safety equipment is mandatory for every lab, and what is optional? Rule #1 – Wear the proper safety equipment ANSWER: Safety glasses are mandatory, however, prescription glasses are an acceptable replacement ** Safety glasses must be worn on the face, not as a hair accessory, even if you find them uncomfortable. ** ** We strongly discourage you from wearing contact lenses into the lab, even if you are wearing safety glasses ** Lab coats are mandatory any time chemicals are being used (e.g. you don’t need them for a bio lab on microscopes) Closed-toe footwear is required – no sandals or flip flops Gloves are only needed during dissections, where you are likely to be handling a biohazardous specimen Rule #2 – Don’t eat or drink in the lab Rule #2 – Don’t eat or drink in the lab QUESTIONS: Suppose you know your three solutions are salt water, sugar water and vinegar, and that none of these substances will hurt you. Why should you still not taste them? Why is it not even a good idea to bring in a water bottle or chewing gum? Rule #2 – Don’t eat or drink in the lab ANSWERS: We cannot guarantee that the solutions are pure, that the glassware is clean, or that there are no chips in the beaker You shouldn’t stake your life on being 100% right about any experiment You could reach for your water and get something else You don’t want anything else on your lab bench other than the required materials Even water can be a dangerous chemical if mixed with the right substance Gum can absorb fumes and increase your chance of ingesting something Rule #3 – Leftover chemicals QUESTION: What should be done with any used or unused chemical once they have been taken from the original container (the source container)? Rule #3 – Leftover Chemicals ANSWER: ALWAYS put any chemicals you’ve taken from the stock beaker in the waste beaker OR If it is safe to do so; with the tap running to dilute it, you can pour it down the drain. WHY: The chemicals could have been contaminated by something already in your beaker so it can never go back to the original container The new chemical you made will have different properties Some solutions, such as those containing heavy metals, will pollute local water supplies, or corrode the pipes Rule #4 – Wash your hands thoroughly QUESTION: When should you wash your hands in the lab? POSSIBLE ANSWERS: A) Before starting the experiment B) After handling each chemical C) At the end of the lab activity D) Before you eat your lunch Rule #4 – Wash your hands thoroughly CORRECT ANSWER: C) At the end of the lab activity unless you spill anything on your hands while you’re working, there’s no need to wash your hands in between each chemical you should wash your hands at the end, even if they appear clean, because some chemicals are invisible and others just plain stink. Rule #5 – Clean up any spilled substances immediately QUESTION: Why should you check with your teacher first before you clean something up? POSSIBLE ANSWERS: A) Because you should collect the spilled liquid and try to reuse it B) Because your teacher wants as many reasons as possible to yell at you C) Because there may be specific instructions for cleaning up that particular solution D) Because your teacher spends less money on her clothes than you, so she doesn’t mind the risk Rule #5 – Clean up any spilled substances immediately CORRECT ANSWER: C) Because there may be specific instructions for cleaning up that particular solution (Although your teacher does probably spends less money on her clothes than you) For instance, if you spill an acid, we will first neutralize it with a basic solution (baking soda solution) if you spill a base, we will first neutralize it with an acidic solution (vinegar solution) some compounds should be diluted first before wiping them up some compounds stain your skin, and you don’t want to be known as the “splotchy kid” Rule #6 – Pour chemicals properly QUESTION: Which photo demonstrates the safe way of pouring chemicals? Rule #6 – Pour chemicals properly ANSWER: Actually none of them! (Photo #1 is the safest) If you spill something, immediate stop pouring and check for a break in your glassware Precise measurements are made at eye level, not above or below your line of sight Rule #6 – Pour chemicals properly ANSWER: Don’t get your lab partner to hold on to your glassware while you pour Especially with test tubes and graduated cylinders (which are narrow), never pour chemicals into containers held in your hands – place the container on the counter or use a test tube rack Rule #7 – Always listen to the teacher’s instructions QUESTION: You should know what you’re doing in the lab BEFORE you go in, so what are some reasons why you should listen to the teacher’s instructions during the lab? Rule #7 – Always listen to the teacher’s instructions ANSWERS: There may have been a change to the procedure due to a lack of supplies There could be some kind of emergency Because your teacher charges a “stupid question tax” of 5 marks if you ask her something that is in the lab procedure or was said out loud while you weren’t listening Rule #8 – Label any containers you put chemicals in QUESTION: One of these solutions is sugar water, one is an acid strong enough to dissolve concrete, and one will make you go blind if you drink it. Which is which? Rule #8 – Label any containers you put chemicals in ANSWER: Actually, they’re all water, but hydrochloric acid, methanol, and sugar water are all clear, colourless solutions indistinguishable by sight. SO? Even if you think you can “keep track” of which substance is which, label your containers to minimize your risk, and your error Rule #9 – What to do with broken glassware QUESTION: What’s the proper procedure if you break glassware? ANSWER: 1) Inform your teacher 2) Make sure any other students in the area are alerted 3) Your teacher will collect the large pieces, and we will sweep up the smaller pieces 4) If necessary, we will neutralize any spilled liquid 5) Broken glass goes in a special bin – do not simply throw it out into the garbage. Rule #10 – Safely detect the odor of a substance QUESTION: How should you go about safely detecting the odor of a substance? ANSWER: You hold the container safely away from your face and use your hand to “waft” the scent towards your nose. WHY: The smell may be too strong / irritating to get a nose-full You should never put your face directly over a container in case of splattering or fumes Rule #11 – Tie hair back and roll up loose sleeves Rule #11 – Tie hair back and roll up loose sleeves QUESTION: What are three reasons why it could be dangerous to have long hair or loose sleeves unsecured? Rule #11 – Tie hair back and roll up loose sleeves ANSWER: Both hair or clothing could catch on fire Loose sleeves can knock over glassware, resulting in broken glass or spilling harmful chemicals Both hair and clothing can absorb chemicals and further exacerbate a chemical burn/ irritation Rule #12 – Mixing acids and water QUESTION: What’s the proper way of mixing an acid and water? POSSIBLE ANSWERS: A) Pour the acid into the water – it’s less likely for acid to splash out of the container B) Pour the water into the acid – you’re less likely to get acid on your hands this way C) It doesn’t matter what order you mix two chemicals in, the result will be the same. Rule #12 – Mixing acids and water CORRECT ANSWER: A) Pour the acid into the water – it’s less likely for acid to splash out of the container You always pour any acids into water rather than vice versa because it's much more likely that the liquid being poured into will splash out of the beaker. If it's just water, then it’s not a problem; but if acid gets on your clothes or hands you can get burned badly. Rule #12 – Mixing acids and water CORRECT ANSWER: There’s one other good reason why acid goes in water, and not vice versa If you add water to acid, the first drops of water will react exothermically with the acid, and bubble all over like it’s boiling (very dangerous!) If you add the acid to the water, the acid reacts/ mixes completely with the water and dissipates. So: “Do like you oughta, add acid to wat-ah” Rule #13 – Using distilled water instead of tap water QUESTION: What are three reasons why you get more accurate results if you use distilled water instead of tap water? ANSWER: Tap water will have salts dissolved in it that can skew your results Tap water is not necessarily neutral – in fact at O’Leary it has a pH of about 8.5 Tap water comes out at unpredictable rates, distilled water is easier to control the flow Practice problems: 1. List five steps that should be taken BEFORE entering the lab to perform an experiment? 2. List precautions you can take to prevent: 1. poisoning 2. scalding 3. eye damage Practice problems: 3. The purpose of a lab is to determine what several “mystery solutions are”. You pick up a fresh test tube, and you pour some “mystery solution” into it and add some solute. You give it a good shake by holding your thumb over the opening and shake it up and down. You put the test tube down into the rack and look down into it to make your observations about the reaction that took place. List as many things as you can that were performed unsafely in the lab. Suggest what should have been done instead that would improve the procedure. Practice problems: 4. There are six pieces of emergency equipment in the lab. What are they, where are they, and when/how should they be used? 5. What should be done in the following two scenarios? While your lab partner is boiling some water, the fire alarm goes off. Your lab partner has just poured some acid into what you thought was a beaker of water. It starts to bubble rapidly. Practice problems (Solutions): List five steps that should be taken BEFORE entering the lab or starting to perform an experiment? 1. read the lab COMPLETELY and prepare any pre-lab requirements prepare a pre-lab – predictions, hypotheses, and observations tables should be prepared in advance come dressed in the appropriate clothing bring only the necessary items to the lab (text book, pre-lab, pen/pencil) NO purses, binders, jackets, or iPods put on protective wear: goggles, lab coat, gloves (if necessary) collect solutions/ materials from the front and bring them to your station LABEL all beakers/ materials…keep your station organized! double check with your partners that you ALL understand the procedure – once the lab begins you will lose marks for procedural questions Practice problems (Solutions): List precautions you can take to prevent: 2. poisoning 1. always wash your hands after the lab, don’t touch your face/ mouth during the lab never use your mouth to open containers etc. scalding 2. never touch test tubes/ beakers that are sitting in water in case they are hot use “oven mitts” or tongs to handle any hot glassware eye damage 3. keep your safety glasses on at all times do not wear contact lenses in chemistry labs (some fumes can cause them to melt to your eyes) Practice problems (Solutions): 3. The purpose of a lab is to determine what several “mystery solutions are”. You pick up a fresh test tube, and you pour some “mystery solution” into it and add some solute. You give it a good shake by holding your thumb over the opening and shake it up and down. You put the test tube down into the rack and look down into it to make your observations about the reaction that took place. You shouldn’t mix chemicals unless you have a reasonable idea how they will react You shouldn’t shake glassware vertically You shouldn’t use thumb as a stopper Never look directly into a test tube from above (splatters/ fumes can get in your face). Practice problems (Solutions): Emergency equipment Where it’s located When & how to use Emergency shower In the corner by the door •Use it if you spill a large amount of chemical on your clothes •Pull the handle, strip down clothes and rinse for 15 minutes Eye wash station Over the sink in the second lab bench •Use if you splash chemicals in your eyes •Place your eyes directly over the two faucets, pull the metal paddle towards you, rinse for 15 minutes Practice problems (Solutions): Emergency equipment Where it’s located When & how to use Fire extinguisher In the prep room between •Use if the lab is on fire! the Chem & Bio labs •P – pull the pin •A – aim at the fire •S – squeeze the trigger •S – sweep over the flames Fire blanket In the corner by the green •Use if you are on fire – cupboards “stop, drop and roll” isn’t enough •Wrap yourself in the blanket as tightly as possible and roll on the floor or get someone to pat you out Practice problems (Solutions): Emergency equipment Where it’s located When & how to use Emergency Spill Kit On the wall by the fume hood •Use for a large spill, or corrosive or oxidizing substances, or if fumes are escaping to other rooms •Alert your teacher, who has received special training First Aid Kit On the counter by the green cupboards •Alert your teacher, who will assist you Practice problems (Solutions): What should be done in the following two scenarios? 5. While your lab partner is boiling some water, the fire alarm goes off. Turn off the hot plate/ Bunsen burner. Separate any chemicals that could possibly react while you’re out of the room, then calmly leave the lab. Your lab partner has just poured some acid into what you thought was a beaker of water. It starts to bubble rapidly. The two chemicals are now reacting. Do not touch the beaker. Come get the teacher/ lab tech and tell them what the two chemicals could be Safety symbols Two different systems of safety symbols have been developed to warn users of potential hazards What does the acronym stand for? Where is it used? Number of symbols Colour coded? HSHS WHMIS Household Safety Hazard Symbols Workplace Hazardous Materials Information System For household products (the average consumer) In labs & industry (people with some understanding of chemistry) 4 8 Yes, three shapes and colours are used to indicate degree of hazard No, all symbols are in circles and are printed in black or red HSHS Household Safety Hazard Symbols CORROSIVE POISONOUS EXPLOSIVE FLAMMABLE The product can burn your skin or eyes. If swallowed, it will damage your throat and stomach If you swallow, lick, or in some cases, breathe in the chemical, you could become very sick or die. The container can explode if heated or punctured. Flying pieces from the container can cause serious injury. The product or its fumes will catch fire easily if it is near heat, flames or sparks. HSHS degree of hazard CAUTION WARNING DANGER Yellow inverted triangle Orange diamond Red octagon LOWEST HAZARD HIGHEST HAZARD WHMIS the Workplace Hazardous Materials Information System is a national warning system for dangerous chemicals WHMIS symbols are more specific than HSHS, but require a bit more understanding of chemicals WHMIS SYMBOLS Symbol Meaning Description compressed gas gas is under pressure and could explode flammable oxidizing Examples propane tank helium tank can burn or burst into flames if placed near a heat source or spark natural gas reacts with oxygen, may not burn itself, but will release gases that make a fire worse chlorine ethanol nitrogen dioxide WHMIS SYMBOLS Symbol Meaning Description poisonous, causing immediate toxic effects a substance that is highly poisonous and will cause immediate health hazards poisonous, causing other toxic effects a substance that is still harmful, but will act over a longer period of time (e.g. carcinogens cause cancer) corrosive can attack (corrode) metals or cause permanent damage to human tissues on contact burning, scarring, and blindness may result Examples cyanide gas some pesticides gasoline fumes cigarettes ammonia hydrochloric acid sodium hydroxide WHMIS SYMBOLS Symbol Meaning Description dangerously reactive may react violently under conditions of shock or an increase in pressure or temperature they may also react vigorously with water to release a toxic gas. biohazard Examples pure hydrogen peroxide aluminium chloride organisms or toxins released blood by organisms that causes E. coli bacteria disease in humans WHMIS - purpose To ensure that all work places across Canada that work with hazardous chemicals have a standardized way for handling and labeling toxic chemicals. Provides: hazard identification product classification ways of safely storing and organizing chemicals labeling methods & material safety data sheets standardized worker training and education Practice problems: 7. For each room listed, suggest one hazardous chemical that might be found in it. Include the WHMIS symbol that would be appropriate for that hazard. a) kitchen b) bathroom c) garage d) bedroom e) classroom http://toxmystery.nlm.nih.gov/ Material Safety Data Sheets MSDS are detailed information sheets describing the particular properties, hazards, and emergency procedures for a specific chemical Each and every single chemical in the lab has its own sheet, and they are organized into binders alphabetically Material Safety Data Sheets Information that can be found on the MSDS: material’s identity (e.g. Clorox bleach) brand name e.g. Clorox chemical name e.g. sodium hypochlorite common name e.g. bleach Material Safety Data Sheets hazardous ingredients lists ingredients as small as 1% e.g. sodium hypochlorite, 5.25% unless listed, the rest of the substance is water physical & chemical hazards / characteristics stability, reactivity, flammability, explosiveness, corrosiveness, compatibility with other materials often listed using WHMIS symbols e.g. stable, incompatible with strong acids, toxic, corrosive, oxidizing Material Safety Data Sheets health hazards and information acute and chronic effects e.g. corrosive, causes eye and skin burns, causes digestive tract burns, harmful if inhaled, causes respiratory tract irritation carcinogen? (may include human and animal summaries) exposure limits how it gets into the body, what organs it targets, symptoms of overexposure e.g. route: blood; organs: eyes, skin, lungs Material Safety Data Sheets precautions for handling and storing any safety equipment required (e.g. gloves, eyewash station) or monitoring equipment emergency and first aid procedures how to deal with cases of inhalation, ingestion, and eye or skin contact e.g. ingestion: do not induce vomiting specific fire-fighting information procedures for cleanup and spills identity of the organization responsible for creating the MSDS, date of issue, contact number Homework Safety worksheet Properties and Classification of Matter Properties of matter “Matter” includes anything that has mass and takes up space To divide up this nearly infinite list of “stuff”, we classify different types of matter based on their properties Properties: the physical and chemical characteristics of a substance Physical properties: appearance and composition of a substance, can usually be determined using the five senses Chemical properties: the reactivity of a substance, can be determined by doing an experiment Physical properties Property Description boiling point temperature of boiling (or condensing) melting point temperature of melting (or freezing) malleability can be flattened into sheets without crumbling ductility can be stretched into wires without breaking colour colour (or colourless) transparency clear (or opaque) state solid, liquid, or gas at room temperature solubility ability to dissolve (usually in water) crystal formation formation of crystals, appearance of crystals conductivity ability to conduct heat or electricity magnetism magnetic attraction between objects Chemical properties Property Description ability to burn combustion (causing flame, heat and light) flash point temperature needed to ignite a flame behaviour in air tendency to break down, react, tarnish reaction with water tendency to corrode or dissolve reaction with acids corrosion, sometimes bubble formation reaction to heat tendency to melt or decompose reaction to red and blue litmus blue red - acid red blue - base no colour change - neutral Matter Pure substances Elements Compounds Mixtures Homogeneous Heterogeneous Solutions Mechanical mixtures Suspensions Colloids Classification of matter Pure substances Mixtures all the particles making up combination of pure the substance are identical elements –cannot be broken down into other substances e.g. carbon compounds – made up of two or more elements in fixed ratios e.g. water substances homogeneous mixtures – the separate components are not visible heterogeneous mixtures – the separate components are visible Classification of matter Homogeneous mixtures the prefix “homo” means “the same”, meaning all the parts of the solution look the same Solutions one example of a homogeneous mixture in a solution, one substance is dissolved in another the substance dissolving is the solute and the substance it’s dissolving in is the solvent Classification of matter Mechanical mixtures Suspensions Colloids The different The components Similar to a substances are clearly visible E.g. trail mix of the mixture are in different states E.g. mud in water, aerosol sprays suspension but the suspended substance cannot easily be separated out E.g. whole milk Classification of matter Not every substance can be easily classified because some substances have features of several categories E.g. motor oil doesn’t have constant properties – it can separate out over time so sometimes it behaves as a solution and sometimes a colloid Pure substances are much easier to classify because Elements are classified in the periodic table Compounds are further classified according to the elements that compose them Physical change vs. chemical change Physical change – does not alter the chemical characteristics of the substances involved, e.g. phase changes changing from solid to liquid to gas crystallization / dissolving allowing a substance to crystallize, then dissolve back into solution Chemical change – the substances produced have different chemical properties than the substances that reacted Chemical reactions In a chemical reaction, chemical change occurs when: at least one new substance is formed, with new physical and chemical properties sometimes, those new substances can be observed with phase changes such as bubbles or precipitates sometimes, they can be observed with colour changes or new odours, a change in energy occurs this is often detected by a change in temperature (e.g. it gives off heat) Homework A1.2 – Check and Reflect Pg.17 #1-7, 9 Developing Ideas about Matter Chemistry in our world Even before scientists fully understood the structure of the atom or had the technology to study it, people were using chemistry in their daily lives Food chemistry Metallurgy Alchemy Food chemistry Methods of preserving food heating food – temporarily sterilizes it (kills the microorganisms) canning – heating the food, then sealing it in an air-tight container freezing – low temperatures prevent the grown of microorganisms Food chemistry salting – salt dries the water out of the meat, but also preserves it by drying the water out of any bacteria fermentation – bacteria naturally present on the surface of living organisms converts starches and sugars into acid, preserving the food and giving it a sour flavour Metallurgy the science of producing and using metals annealing the heating of a metal before it’s hammered, which makes it less brittle this technique has allowed copper to be used for tools, weapons and jewellery for thousands of years copper works better than other pure metals because of its hardness and malleability Metallurgy other techniques include alloys – the heating and combination of two or metals to gain the benefits of both e.g. brass, bronze smelting – extracting pure metal from its ore e.g. getting iron from iron ore Alchemy early experiments involving a combination of science and magic was mostly schemes for getting rich, or producing “miracle cures” such as antiaging serums in the process, also resulted in: the discovery of mercury a method for the production of acids the improvement of glassware and lab equipment Our understanding of the atom Accurately describing matter in terms of its atomic structure is something that scientists are still working on today. This section deals with the evolution of our ideas on the atom and its structure, from Aristotle (~400 BC) to today’s theory on Quantum Mechanics As we go through each scientist’s contribution, try to identify What was different about his view compared to previous theory? How did this view contribute to our current understanding? Our understanding of the atom What you learn in Science 10 about the actual structure of the atom is still a simplification of what we believe to be true, however we will present the Bohr model as being “close enough”. Our understanding of the atom Different elements are simply different combinations of the same three particles What makes oxygen different than nitrogen, for instance, is that oxygen has one more of each of the particles, but an oxygen proton looks exactly the same as a nitrogen particle. Our understanding of the atom Features of this model of the atom: Protons (p+) Positively-charged particles Found in the nucleus Neutrons (n0) Neutral particles Found in the nucleus Prevent the p+ from repelling each other Our understanding of the atom Electrons (e-) tiny negativelycharged particles in orbit around the nucleus hardly have any mass (about 1/2000th that of a p+ or n0), but make up most of the volume of the atom How did we get to this understanding? Aristotle 400 B.C. all matter was composed of 4 DRY elements: earth, air, water and fire what he got right: matter is made up of different combinations of elements elements can be divided up based on their properties what he got wrong: only 4 elements the elements were “continuous”, that is, they weren’t composed of “parts” COLD HOT WET Democritus 400 B.C. working at the same time as Aristotle matter was made up of tiny particles, called atomos, that could not be divided into smaller pieces what he got right: matter is composed of tiny particles the atoms determine the properties of the element what he got wrong: atoms are indivisible, that is, that they aren’t made up of any smaller parts John Dalton early 1800s performed experiments by combining different elements to form new substances (compounds) atoms are like small spheres that varied in size, mass or colour John Dalton early 1800s what he got right: all matter is made of small particles called atoms all atoms of an element are identical in properties such as size and mass atoms of different elements have different properties atoms of different elements can combine to form compounds with new properties what he got wrong: atoms are solid and cannot be divided any further J.J. Thomson experimented with beams of particles produced in a vacuum tube he passed electricity through different samples of elements, and found that a beam of particles was emitted when the element became “excited” showed that the beam was made of negative charges, and that different elements produced the same type of beam thus credited with the discovery of the electron 1890s J.J. Thomson The “Plum Pudding” Model In Thomson’s model, the negatively charged electrons are stuck in a sphere of positive charge so named because it’s like raisins in plum pudding (or chocolate chips in a cookie) 1890s J.J. Thomson what he got right: atoms can be further divided into smaller particles one of those particles is the electron, which carries a negative charge the electrons in one element are the same as electrons in another element, but in different amounts 1890s J.J. Thomson what he got wrong: the electrons are stuck in the positive sphere, instead of around it this was corrected by Japanese Scientist H. Nagaoka in 1904, who said the electrons traveled around the nucleus like Saturn’s rings 1890s Ernest Rutherford begun his research with Thomson, but expanded on his ideas by discovering the nucleus his experiment is called the Gold Foil Experiment, and was so designed: a sample of a radioactive element was placed in a lead chamber with a tiny opening through this opening, a beam of positively-charged particles would be emitted these particles were traveling at a VERY high speed toward a thin sheet of gold foil 1890s Ernest Rutherford because the particles were moving so fast, and the gold was so thin, he expected the gold foil not to slow the particles down at all, and that they would pass through this would be like firing a cannonball at a sheet of tissue paper most of the time, this is exactly what happened, however… once in about every 10000th time, the particle would bounce back this would be like the tissue deflecting the cannonball – a very surprising result! 1890s Ernest Rutherford from this experiment, he realized that the gold atoms had to be made mostly of empty space when the beam hit the 99.99% of empty space, it passed right through however, 0.01% of the time, the beam struck a positive “spot” on the gold foil, which he determined to be the tiny nucleus of the atom 1890s Ernest Rutherford what he got right: the nucleus is very small compared to the empty space around it the electrons occupy some of the empty space the nucleus has a positive charge what he got wrong: the electrons “swarmed” around the nucleus like bees around a beehive 1890s Neils Bohr credited with discovering the orbital levels of the electrons, that is, that the electrons don’t swarm randomly, but rather occupy specific energy levels around the nucleus he discovered this by passing electricity through different elements to excite them, then letting the electrons release that energy in the form of light early 1900s Neils Bohr he then noted the pattern of light bands emitted from the element since the light emitted was related to the electrons, he reasoned that each element had a different number of electrons, and that the electrons occupied certain energy levels early 1900s Neils Bohr early 1900s what he got right: electrons occupy set energy levels around the nucleus electrons cannot fall below the lowest energy level Neils Bohr early 1900s what he got wrong: electrons are not actually a solid particle, but actually a cloud of negative charge – Quantum Mechanical Model there are two types of particles in the nucleus: the proton and the neutron Homework A1.3 Check and Reflect (page 25) #1, 5, 6, 7 A1.0 Section Review (page 27) #3, 5, 9, 15, 17