HW - pp 282-283
advertisement

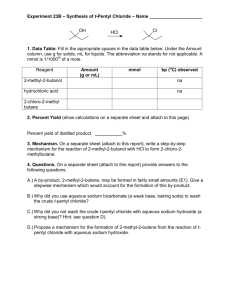
Last night’s homework: Problems 4-6 on page 282 Questions 7-11 on page 283 Write chemical equations for each of the following reactions. 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl 4. In water, iron(III) chloride reacts with sodium hydroxide, producing solid iron(III) hydroxide and sodium chloride. FeCl3 + NaOH → Fe(OH)3 + NaCl FeCl3 + NaOH → Fe(OH)3 + NaCl FeCl3 + NaOH → Fe(OH)3 + NaCl Fe Cl Na O H Fe Cl Na O H FeCl3 + NaOH → Fe(OH)3 + NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 1 1 3 3 FeCl3 + NaOH → Fe(OH)3 + NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 1 1 3 3 FeCl3 + NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 1 1 3 3 FeCl3 + NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 3 3 3 3 FeCl3 + NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 3 3 3 3 FeCl3 + 3NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 1 1 1 Fe Cl Na O H 1 3 3 3 3 FeCl3 + 3NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 3 3 3 Fe Cl Na O H 1 3 3 3 3 FeCl3 + 3NaOH → Fe(OH)3 + 3NaCl Fe Cl Na O H 1 3 3 3 3 Fe Cl Na O H 1 3 3 3 3 5. Liquid carbon disulfide reacts with oxygen gas, producing carbon dioxide gas and sulfur dioxide gas. 5. Liquid carbon disulfide reacts with oxygen gas, producing carbon dioxide gas and sulfur dioxide gas. CS2 + O2 → CO2 + SO2 CS2 + O2 → CO2 + SO2 CS2 + O2 → CO2 + SO2 C O S C O S CS2 + O2 → CO2 + SO2 C O S 1 2 2 C O S 1 4 1 CS2 + O2 → CO2 + 2SO2 C O S 1 2 2 C O S 1 4 1 CS2 + O2 → CO2 + 2SO2 C O S 1 2 2 C O S 1 6 2 CS2 + 3O2 → CO2 + 2SO2 C O S 1 2 2 C O S 1 6 2 CS2 + 3O2 → CO2 + 2SO2 C O S 1 6 2 C O S 1 6 2 CS2 + 3O2 → CO2 + 2SO2 C O S 1 6 2 C O S 1 6 2 6. Solid zinc and aqueous hydrogen sulfate react to produce hydrogen gas and aqueous zinc sulfate. 6. Solid zinc and aqueous hydrogen sulfate react to produce hydrogen gas and aqueous zinc sulfate. Zn + H2SO4 → H2 + ZnSO4 Zn + H2SO4 → H2 + ZnSO4 Zn H S O Zn H S O Zn + H2SO4 → H2 + ZnSO4 Zn H S O 1 2 1 4 Zn H S O 1 2 1 4 7. List three types of evidence that a chemical reaction has occurred. 7. List three types of evidence that a chemical reaction has occurred. Changes in: • Temperature • Color Appearance of: • Odor • Gas bubbles • Solid precipitate 8. Compare and contrast a skeleton equation and a chemical equation. 8. Compare and contrast a skeleton equation and a chemical equation. Both show formulas of reactants and products. Chemical equation also shows relative amounts. 9. Why is it important that a chemical equation be balanced? 9. Why is it important that a chemical equation be balanced? Mass is neither created nor destroyed. 10. When balancing a chemical equation, can you adjust the number that is subscripted to a substance formula? 10. When balancing a chemical equation, can you adjust the number that is subscripted to a substance formula? No 10. When balancing a chemical equation, can you adjust the number that is subscripted to a substance formula? No H2O → H2 + O2 10. When balancing a chemical equation, can you adjust the number that is subscripted to a substance formula? No H2O → H2 + O2 H2O2 → H2 + O2 11. Why is it important that to reduce the coefficients in a balanced equation to the lowest possible whole-number ratio? 11. Why is it important that to reduce the coefficients in a balanced equation to the lowest possible whole-number ratio? Clearly shows the relative amounts. .