Unit 3 - Introduction - Scientific Notation and SI
advertisement

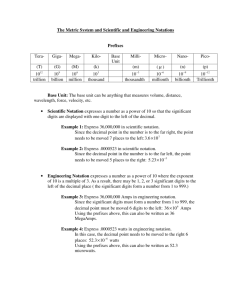
Grade 9 Academic Science - Chemistry Scientific Notation What is this number? 300 000 000 m/s 0.000 000 000 753 kg These numbers are very hard to write. A method has been developed to express very large or small numbers. The method is called scientific notation. Scientific notation is based on powers of the base number 10. The number 300,000,000 is written as 3.0 X 108 The first number 3 is called the COEFFICIENT. It must be greater than or equal to 1 and less than 10. The second number is called the BASE. It must always be 10 in scientific notation. The base number 10 is always written in exponent form. In the number 3.0 x 10 8 the number 8 is referred to as the exponent or power of ten. To write a number in scientific notation, a decimal is put after the first digit and zeroes are dropped. The coefficient in the number 123,000,000,000 is 1.23 To find the exponent count the number of places from the decimal to the end of the number. There are 11 places In 123,000,000,000. Thus, 123,000,000,000 is 1.23 X 1011. Exponents are often expressed using other notations. The number 123,000,000,000 can also be written as 1.23E+11 or as 1.23 X 10^11. Numbers smaller than 1 will have a negative exponent. A millionth of a second (0.000001) is 1.0E-6 or 1.0^-6 or 1.0 X 10-6 Scientific Notation Practice 2,700,000 = 2.7 X 106 260 = 0.16 = 0.965 = 34,657,000,000 = 0.124,000,000 RULE = + exponential - exponential Extremely large and small numbers are difficult to record in full. A standardized reporting format is used called scientific notation. 1. The distance between Mercury and the Sun is 5.8 X 10 7 km. It is not written as 58 000 000 km. What does 107 mean? 2. What is the effect on the exponent by moving the decimal place in the following direction? …to the left __________________________ …to the right _________________________ 3. Rewrite the following measurements in scientific notation. a) 0.000045 km b) 90 200 s c) 0.0076 cm d) 290 000 N e) 0.000042 km f) 20 s g) 0.0623 W h) 456 000 000 g 4. Rewrite the following in full. a) 0.56 X 10-4 b) 3 X 100 c) 3.4 X 10-3 d) 1.6 X 102 e) 9.4 X 104 The Metric System (SI) Rules for SI System of Units Correct Incorrect 1. Leave a space between the number and the unit 75 cm 75cm 2. No periods except at the end of a sentence 4.5 g 4.5 g. 3. Put numbers and symbols together 6 kg six kilograms 6 kilograms six kg 4. Use decimals, not fractions 1.5 L 1½ L 5. Always put a zero in front of the decimal for numbers less than one. 0.0025 cm .0025 cm 6. Group digits in threes for long numbers. Do not use commas. 14 091.205 s 14,901.205 s 7. Do not put “s” after the unit symbol. 10 kg ten kilograms 10 kgs Task Find the SI errors and correct for following statements a. The ribbon was 90cm long b. The sun is 150,000,000 kms away c. The flash in the sky lasted 1 ¼ s. d. Simon weighed 52 g. last time he checked. e. In Canada, one pound is equal to 0.43559237 kilograms f. Each candy costs $ .35. Grade 9 Academic Science – Chemistry Significant Digits / Figures Significant figures are critical when reporting scientific data because they give the reader an idea of how well you could actually measure/report your data. The Rules ALL non-zero numbers (1,2,3,4,5,6,7,8,9) are ALWAYS significant. ALL zeroes between non-zero numbers are ALWAYS significant. ALL zeroes which are SIMULTANEOUSLY to the right of the decimal point AND at the end of the number are ALWAYS significant. ALL zeroes which are to the left of a written decimal point and are in a number >= 10 are ALWAYS significant. Zeroes placed BEFORE other digits are NOT significant (e.g., 0.046 has two significant digits) A helpful way to check the last two rules is to write the number in scientific notation. If you can/must get rid of the zeroes, then they are NOT significant. Examples: How many significant figures are present in the following numbers? Number Significant Digits Explanation 48,923 5 3,967 4 900.06 5 0.0004 1 8.1000 5 6 1 501.040 6 10.0 3 3 X 10 All non-zero numbers Significant Digits Identify the number of significant digits in the following examples 3560.39 SD = 356.0 SD = 356 SD = 0.0125 SD = 0.01250 SD = Identify the number of significant digits in the following examples. 0.0000067 2001 3.60 4.3 kg 200 393 cm 0.0073 g 0.0050 Tbytes When quantities are being added or subtracted, the number of decimal places (not significant digits) in the answer should be the same as the least number of decimal places in any of the numbers being added or subtracted. Example: 5.67 1.1 0.9378 7.7 2 decimal places 1 decimal place 4 decimal places Thus, the answer has only one decimal place Metric Prefixes and Conversions Like most of the world, Canada uses the metric system of measurement. We know the common metric prefixes such as centimeter, kilogram and liter. Yet, there are many more prefixes, and in science, the scale of measurement can be very large or very small. Metric Prefixes Prefix Symbol Measurement (order of magnitude) Actual Value Example tera T 1 X 1012 1 000 000 000 000 terameter giga G 1 X 109 1 000 000 000 gigameter mega M 1 X 106 1 000 000 megameter kilo k 1 X 103 1 000 kilometer hecto h 1 X 102 100 hectometer deca da 1 X 101 10 decameter BASE UNIT g L m s 1 X 100 1 gram liter meter second deci d 1 X 10-1 0.1 decimeter centi c 1 X 10 -2 0.01 centimeter milli m 1 X 10-3 0.001 millimeter micro 1 X 10 0.000 001 micrometer nano n 1 X 10-9 0.000 000 001 nanometer pico p 1 X 10-12 0.000 000 000 001 picometer -6 Task Give the metric abbreviation for the following meter decigram microsecond kilogram millimeter megameter nanosecond teragram milliliter centiliter decaliter picogram What is the numeric meaning of each metric prefix? mega centi deca deci milli nano kilo tera giga Convert the following metric units 100 m = _______________________ km 62.5 L = _______________________ cL 485 kg = _______________________ g 0.006 m = _____________________ mm 4.23 km = ______________________ Gm 0.24 cm = ______________________ mm 0.24 cm = ______________________ Mm 5 000 L = ______________________ kL 705 ns = _______________________ s 1.5 ML = ______________________ kL 450 Tbytes = ___________________ bytes 0.003 m = ______________________ cm large to small move decimal to the right small to large move decimal to the left Grade 9 Science – Chemistry Measurement and Significant Figures Task - Suggest the best unit to measure each line below, estimate the length of each line and record these in the table below. Then, carefully measure each line below and complete the table (use proper significant figures). 1 2 3 4 5 6 Line # 1 2 3 4 5 6 Line 6 width Text thickness Text page length Floor tile width Best Unit Estimated Length (L1) Measured Length (L2) Difference (L2 – L1) % Error = (L2 – L1) / L2 X 100 Grade 9 Academic Science - Chemistry Measurement, Conversion and Significant Figures Unit Conversions Time Distance 1 h = 60 min, 1 min = 60 s 1 km = 1000 m Conversion Examples Convert 4 h to seconds 1 h = 3600 s (Conversion Factor) 4 h X 3600 s/1 h = 14400 s Convert 5.2 km to metres 1 km = 1000 m 5.2 km X 1000 m/1 km = 5200 m Convert 25 m/s to km/h 1000 m = 1 km 3600 s = 1 h 25 m/s X 3600 s/1 h X 1 km/1000 m = 90 km/h Practice 1. Convert 156 km/h to m/s 2. Convert 100 m/s to km/h 3. Convert 75 km/h to m/yr 4. Convert 170,000 mm/s to m/s 5. Convert 75 cm/ns to Gm/day