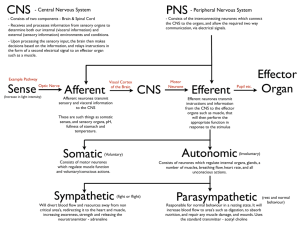
The Nervous System
advertisement

The Nervous System Question What is a group of axons travelling together in the CNS called What does the CNS consist of What does a neurone cell body (soma) contain What are dendrites What is the function of dendrites What is the region called where the axon connects to the cell body, what is its function Which part of a neurone is responsible for releasing neurotransmitters What type of neurones are unipolar Where can you find bipolar neurones What type of neurones are motor neurones & interneurones What are the myelin forming cells called in the brain & spinal cord What forms individual myelin sheaths in the peripheral nervous system What are nodes of ranvier What is the function of a myelin sheath What do glial cells surround What is the function of glial cells What are the different types of glial cells What are the functions of astrocytes What molecules can cross the BBB What are the functions of microglia What do microglia contain to help them perform their function Where do you find ependymal cells What is the function of ependymal cells Where are the meninges What are the 3 membranes making up the meninges What is in the subarachnoid space Answer Pathway or tract – no nerves in CNS Brain Spinal cord Nucleus & ribosomes – has genetic information & machinery for protein synthesis Multiple mitochondria, RER, free ribosomes, golgi apparatus, lysosomes, vesicles Highly branched outgrowths of the cell body Receive most of the inputs from other neurones = site for synapses, ↑ s.area for receiving signals Initial segment/axon hillock – triggers most of the electrical signals Axon terminal Sensory neurones Retina Multipolar Oligodendrocytes Schwann cells Spaces between the myelin sheaths on peripheral nerves Speeds up the conduction of electrical signals along the axon & conserves energy Soma, axon & dendrites of neurones Physically & metabolically support neurones Oligodendrocytes Astrocytes Microglia Ependymal cells Help regulate composition of ECF in the CNS by removing K+ & neurotransmitters around synapses Stimulate the formation of tight junctions between the cells that make up the walls of capillaries – forming BBB Involved in the transport of substances from blood to neurones Sustain neurones metabolically by providing glucose Guide neurones as they migrate to their ultimate destination in developing embryos Small molecules like O2, CO2, H2O They are a specialised macrophage-like cell that perform immune functions in the CNS, they coalesce in areas of damage Lysosomes They line fluid filled cavities within the brain & spinal cord Produce & regulate the production & flow of CSF Between the soft neural tissues of the brain & spinal cord & the bones that house them Thick dura mater next to the bone Arachnoid mater in the middle Thin pia mater next to the nervous tissue CSF 1 What is the subarachnoid space between What are the functions of the meninges What cells is CSF produced by Where are the cells that produce CSF What system does the CSF circulate through to the brainstem Where does the CSF ultimately flow to, why Where do the dural sinuses drain to What are the 2 layers of the dura mater What is the embryological origin of the CNS Where is the tubule structure in the brain Where is the tubular structure in the spinal cord What are the 4 ventricles in the brain How are the lateral ventricles connected to the 3rd ventricle How is the 3rd ventricle connected with the 4th ventricle What forms the dorsal wall of the 4th ventricle What is the volume of CSF What reabsorbs CSF What are the factors favouring reabsorption of CSF What happens to the neural tube in the 4th week of development What are the 3 prominent brain vesicles from the neural tube, what do they become What does the Prosencephalon further divide into, what do these divisions become What does the Rhombencephalon further divide into What is the shell of the cerebral cortex made up of What connects the left & right cerebral hemispheres What divides the diencephalon in 2 What does the diencephalon contain What is the function of the thalamus What is the function of the hypothalamus What is the function of the brain stem What does the brain stem consist of The arachnoid & pia Protect & support the CNS – circulate & absorb the CSF Ependymal cells They make up a specialised epithelial structure = choroid plexus Interconnected ventricular system The top of the outer surface of the brain, where most of it enters the blood stream through one way valves in large veins Internal jugular veins Periosteal layer Internal meningeal layer Neural tube Ventricular system Narrow central canal 2 lateral ventricles 3rd ventricle 4th ventricle Interventricular foramen Cerebral aqueduct Cerebellum 150ml Arachnoid villi – located along the length of the dural venous sinus Higher hydrostatic pressure in the CSF Higher oncotic pressure in the blood 3 areas in the cephalic portion of the tube enlarge creating 3 prominent brain vesicles Prosencephalon = forebrain Mesencephalon = midbrain Rhombenephalon = hind brain Telencephalon = cerebrum Diencephalon Metancephalon = cerebellum & pons Myelencephalon = medulla oblongata Outer layer of grey matter Inner layer of white matter The corpus callosum The 3rd ventricle Thalamus Hypothalamus Collection of several large nuclei that serve as synaptic relay stations & important integrating centres for most inputs to the cortex Maintain homeostasis - Master command centre for neural & endocrine coordination, it lies directly above the pituitary gland & modulates its function, coordinates the limbic system Relays information to or from cerebrum or cerebellum Mesencephalon Pons Medulla oblongata 2 Why is the pons a ‘bridge’ What does the pons contain What is the function of the cerebellum Where does the spinal cord connect to the brainstem What are the functions of the medulla oblongata How does arterial blood reach the brain How does most of the venous blood from the brain leave the cranium What are the internal carotid arteries branches of How do the internal carotid arteries enter the skull What are the vertebral arteries branches of How do the vertebral arteries enter the cranium Where is blood to brainstem & cerebellum derived from Where is blood to the cerebrum derived from What forms the circle of willis What does the anterior cerebral artery supply What does the middle cerebral artery supply What does the posterior cerebral artery supply Which is the largest region of the brain What originates in the cerebral hemispheres (processes) What happens to somatic sensory information relayed to the brain What are gyri What are sulci What are fissures What is the purpose of gyri & sulci Why does the cerebral cortex need lots of neurones What does each cerebral hemisphere receive What does each cerebral cortex generate What is in the Frontal lobe of the cerebral cortex What is in the Parietal lobe of the cerebral cortex What is in the Occipital lobe of the cerebral cortex What is in the temporal lobe of the cerebral cortex Bridge between cerebellum & brain stem Nuclei involved with both somatic & visceral motor control Automatically adjusts motor activities on the basis of sensory information & memories of learned patterns of movement = control of balance, posture/muscle tone, coordination of movement At the medulla oblongata Relays sensory information to the thalamus & to other brainstem centres Contains major centres concerned with the regulation of autonomic function e.g. heart rate, blood pressure, digestive activities Through the internal carotid arteries & the vertebral arteries In the internal jugular veins which drain into the dural sinuses Common carotid artery Through the carotid foramen 1st branch of subclavian arteries Through forum magnum with the spinal cord Branches of vertebro-basilar system Circle of Willis – an anastamosing system of arteries Internal carotid & basilar arteries Medial aspect of cerebral hemispheres excluding occipital lobe Lateral aspect of cerebral hemispheres Inferior aspect of cerebral hemispheres & occipital lobe The cerebrum Conscious thought processes & all intellectual functions It reaches out conscious awareness & cerebral neurones exert direct or indirect control over somatic motor neurones Elevated ridges on the cortical surface Shallow compressions on the cortical surface Deeper grooves on the cortical surface Increase surface area of cerebral hemispheres – providing additional space for adrenal cortical neurones It performs complicated neural functions and analytical activities requiring large numbers of neurones Sensory information form the opposite side of the body Motor commands to the opposite side of the body Primary motor cortex – conscious control of skeletal muscles (& personality) Primary sensory cortex – conscious perception of touch, pressure, vibration, pain, temperature & taste Visual cortex – conscious perception of visual stimuli Auditory & olfactory cortex – conscious perception of auditory & olfactory stimuli 3 Where are the association areas in the cerebral cortex What happens in association areas What happens in the precentral gyrus What happens in the postcentral gyrus What processes do you have dominant hemispheres for What do you have non-dominant hemispheres for What happens in Brocas’ area Where is Brocas’ area What happens in Wernickes’ area Where is Wernickes’ area What does the central white matter of the cerebral cortex contain What are association fibres for in the central white matter What are commissural fibres for in the central white matter What are projection fibres Where are the basal nuclei What are the basal nuclei What processes are basal nuclei involved in Do the basal nuclei initiate movements What do the basal nuclei do in the control of movements For which movements of the body are the basal nuclei particularly important What is the function of the striatum What is the function of the Globus Pallidus Name the basal ganglia/nucleus What is the grey area of the spinal cord composed of What does the white matter of the spinal cord contain On which side of the cord do afferent fibres enter Where are the cell bodies of afferent neurones Where do the efferent neurones leave the spinal cord What forms each spinal nerve What does the peripheral nervous system transmit information between How many pair of nerves are in the PNS In all of the lobes Integration & processing of sensory data, processing & integration of motor activities Initiation of voluntary motor functions It is involved in somatic sensation Language skills Mathematical reasoning Spatial awareness Musical skills Production of words (including written) Inferior frontal gyrus, dominant hemisphere Understanding of words Posterior, superior temporal gyrus Myelinated fibres that form bundles that extend from one cortical area to another, or that connect areas of the cortex to other areas of the brain Interconnect portions of the cerebral cortex within the same cerebral hemisphere They connect the 2 cerebral hemispheres They are tracts which link the cerebrum with other regions of the brain & spinal cord Within the cerebral hemispheres, embedded within the central white matter Paired masses of grey mater The subconscious control of skeletal muscle tone & the coordination of learned movement patterns No Provide general pattern & rhythm once the movement is underway Trunk and proximal limb movements Caudate nucleus & putamen– controls cycles of arm & leg movements that occur between the decision to start & stop walking Controls & adjusts muscle tone, particularly in appendicular muscles to set body position for voluntary movement Caudate nucleus Putamen Globus pallidus Subthalamic nucleus Substantia nigra Interneurones, the cell bodies & dendrites of efferent neurones, the entering axons of afferent neurones & glial cells Groups of myelinated axons Dorsal side via the dorsal roots Dorsal root ganglia On the ventral side via the ventral roots The dorsal & ventral roots combining The CNS & receptors & effectors 43 4 How many of these nerves are spinal What are the remaining nerves How many pairs of cervical nerves What do cervical nerves do How many pairs of thoracic nerves are there What are the thoracic nerves associated with How many pairs of lumbar nerves are there What are the lumbar nerves associated with How many pairs of sacral nerves are there What are the sacral nerves associated with What do all spinal nerves contain How do cranial nerves differ from spinal nerves in what they contain Why can afferent neurones be called primary afferents or first order neurones Why is the efferent NS more complicated than the afferent NS What do somatic nerves do What do autonomic nerves do How do the chains in a pathway run together Where do sensory pathways carry information to (1 exception) What order of neurones are interneurones What are ‘specific ascending pathways’ What happens to specific pathways from the right side of the body Where do you find somatic receptors Where do specific ascending pathways that transmit information from somatic receptors go Where do the specific pathways from the eyes go Where do the specific pathways from the ears go What happens to afferent information after they have been through the primary cortical receiving areas How do neurones in non-specific ascending pathways differ from those in ascending pathways What are polymodal (inter)neurones 31 12 cranial 8 Control the muscles & glands & receive sensory information input from the neck, shoulder, arm & hand 12 Chest & abdominal wall 5 Hips & legs 5 Genitals & lower digestive tract Afferent & efferent fibres Some cranial nerves contain only afferent fibres They are the first cells entering the CNS The efferent NS is subdivided into somatic & autonomic Innervate skeletal muscle Innervate smooth muscle, cardiac muscle, glands & neurones in GI tract In parallel Cerebral cortex – responsible for conscious recognition Second order Ascending pathways in the spinal cord and brain that carry information about single types of stimuli – these pass to the brainstem & thalamus – then to specific sensory areas of the cerebral cortex They cross over to the opposite side of the nervous system – information from receptors on the right side of the body is transmitted to the left cerebral hemisphere Skin, muscles, tendons & joints To the somatosensory cortex The visual cortex in the occipital lobe Auditory cortex in the temporal lobe Continue to association areas in the cerebral cortex where complex integration occurs Neurones in non-specific ascending pathways are activated by sensory units of different types and signal general information (don’t specify what or where) (Inter)neurones that respond to input from several afferent neurones each activated by a different stimulus 5 Where do non-specific ascending pathways end How many neurones are involved in sensory pathways ending in the cerebral cortex How many neurones are involved in sensory pathways ending in the cerebellum What are 2 properties of pathways involving the major sensory & motor tracts of the spinal cord What are first order neurones What are second order neurones What are third order neurones The axon of which neurones is involved in decussation What is a dermatome What is a myotome Which part of a spinal nerve provides sensory innervation Why is cutaneous nerve distribution different from dermatomes What is carried in the spinothalamic tract Where do 1st order neurones in the spinothalamic tract synapse Where do 2nd order neurones decussate What happens to the 2nd order neurones in the brainstem Where do sympathetic nerves leave the CNS Where do parasympathetic nerves leave the CNS What happens above a lesion in a peripheral nerve (lower motor neurone injury) What happens below a lesion in a lower motor neurone injury What happens above a lesion in an upper motor neurone injury What happens at the level of a lesion in an upper motor neurone What happens to muscle tone and reflexes below a lesion in an upper motor neurone What happens to power, sensation and coordination below a lesion in an upper motor neurone Which type of fibres decussate almost immediately on entering the spinal cord In parts of the brainstem & regions of the thalamus & cerebral cortex that are not highly discriminative but are important in controlling awareness & arousal 3 2 The tracts are paired Axons within each tract are grouped according to the body region innervated Sensory neurones that deliver the sensations to the CNS CNS neurones on which 1st order neurones synapse 2nd order neurones synapse onto them in the thalamus Either first or second order neurones A specific region of the body monitored by a pair of spinal nerves = area of skin where sensation is supplied by one spinal nerve Relates to the muscles which share the motor supply of one spinal segment (most muscles belong to more than one myotome) Dorsal ramus Each cutaneous nerve may receive nerve fibres derived from a number of different dermatomes, and/or the nerve fibres from one dermatome may be dispersed amongst a number of cutaneous nerves Fibres related to pain, temperatre, non-discriminatory touch & pressure Within the dorsal horn Prior to ascending in the spinothalamic tract Run in the spinal lemniscus to the thalamus T1 – L2 Cranial 3,7,9 & 10 Sacral 2-4 Above lesion everything is normal Below a lesion everything is absent or decreased Above lesion everything is normal Everything absent or decreased It is increased – because inhibitory neurones from premotor nerves are cut off Absent or decreased Pain and temperature fibres 6 Cranial nerves What are cranial nerves How many pairs of cranial nerves are there Which surface of the brain can they be found on How are cranial nerves numbered What is CNI What is the primary function of CNI What is the origin of CNI What is the destination of CNI What is special about CNI What is CNII What is the primary function of CNII What is the origin of CNII What is the destination of CNII What is CNIII What is the primary function of CNIII What is the origin of CNIII What is the destination of CNIII What is CNIV What is the primary function of CNIV What is the origin of CNIV What is the destination of CNIV What is special about CNIV What is CNV What is the primary function of CNV What are the branches of CNV and what are their functions What is the origin of CNV1 What is the origin of CNV2 What is the origin of CNV3 What is the destination of CNV What is special about CNV What is CNVI What is the primary function of CNVI What is the origin of CNVI What is the destination of CNVI What in CNVII What is the primary function of CNVII What is the origin of CNVII What is the destination of CNVII Components of the PNS that connect to the brain rather than the spinal cord 12 Ventrolateral surface According to their position along the longitudinal axis of the brain, beginning at the cerebrum The Olfactory nerve Special sensory = smell Receptors of the olfactory epithelium Olfactory bulbs Only cranial nerve attached directly to the cerebrum The Optic nerve Special sensory Retina of the eye Diencephalons by way of the optic chiasma The Oculomotor nerve Motor, eye movements Mesencephalon Somatic motor: superior, inferior & middle rectus muscles, levator palpebrae superioris Visceral motor: intrinsic eye muscles The Trochlear nerve Motor, eye movements Mesencephalon Superior oblique muscle It is the smallest of all the cranial nerves The Trigeminal nerve Mixed – sensory & motor CNV1 Ophthalmic branch = sensory CNV2 Maxillary branch = sensory CNV3 Mandibular branch = sensory & motor Orbital structures, nasal cavity, skin of forehead, superior eyelid, eyebrow, part of nose Inferior eyelid, upperlid, gums, teeth, cheek, nose, palate, part of pharynx, Sensory: lower gums, teeth, lips, palate, tongue (part) Motor: motor nuclei of pons All branches to sensory nuclei in the pons Mandibular branch also innervates muscles of mastication It is the largest cranial nerve The Abducens nerve Motor, eye movements Pons Lateral rectus muscle (6th intrinsic eye muscle) The Facial nerve Mixed – sensory & motor Sensory: taste receptors on anterior 2/3rd of tongue Motor: motor nuclei of pons Sensory: sensory nuclei of pons Somatic motor: muscles of facial expression Visceral motor: lacrimal (tear) gland, nasal mucous glands, submandibular & sublingual salivary glands 7 What is Bells palsy What are the symptoms of Bells palsy What is CNVIII What is the primary function of CNVIII What are the branches of CNVIII and what are their functions What is the origin of CNVIII What is the destination of CNVIII What in CNIX What is the primary function of CNIX What is the origin of CNIX What is the destination of CNIX What is CNX What is the primary function of CNX What is the origin of CNX What is the destination of CNX What is CNXI What is the primary function of CNXI What is the origin of CNXI What is the destination of CNXI What is CNXII What is the primary function of CNXII What is the origin of CNXII What is the destination of CNXII How does CNI enter the cranium How does CNII enter the cranium How does CNIII exit the cranium How does CNIV exit the cranium How does CNV1 exit the cranium How does CNV2 exit the cranium How does CNV3 exit the cranium How does CNVI exit the cranium How does CNVII exit the cranium How does CNVIII exit the cranium How does CNIX exit the cranium How does CNX exit the cranium How does CNXI exit the cranium How does CNXII exit the cranium Which part of the brain does CNIII emerge from Which part of the brain does CNIV emerge from Which part of the brain does CNV1 emerge from An inflammation of the facial nerve Paralysis of facial muscles on the affected side & loss of taste sensations from anterior 2/3 of the tongue The Vestibulocochlear nerve Special sensory Vestibular branch: balance & equilibrium Cochlear branch: hearing Receptors of the inner ear Vestibular & cochlear nuclei of pons & medulla oblongata The Glossopharyngeal nerve Mixed – sensory & motor Sensory: posterior 1/3 of the tongue, part of the pharynx, palate, carotid arteries (carotid body & carotid sinus) Motor: motor nuclei of medulla oblongata Sensory: sensory nuclei of medulla oblongata Somatic motor: pharyngeal muscles involved in swallowing Visceral motor: parotid salivary gland The Vagus nerve Mixed: sensory & motor Visceral sensory: pharynx, auricle, external acoustic meatus, diaphragm & visceral organs in thoracic & abdominopelvic cavities Visceral/somatic motor: motor nuclei in medulla oblongata Visceral Sensory: sensory nuclei & autonomic centres of medulla oblongata Somatic motor: muscles of palate & pharynx Visceral motor: respiratory, cardiovascular & digestive organs The Accessory nerve Motor (soft palate, throat & neck) Motor nuclei of spinal cord & medulla oblongata Internal branch (branchial motor): voluntary muscles of palate, pharynx & larynx External branch (somatic motor): sternocleidomastoid & trapezius The Hypoglossal nerve Motor, tongue movements Motor nuclei of the medulla oblongata Muscles of the tongue Through foraminifera in cribriform plate of ethmoid bone Enters via optic canal, nerves join to form optic chiasma Via superior orbital fissure Via superior orbital fissure Via superior orbital fissure Via the foramen rotundum Via the foramen ovale Via superior orbital fissure Internal acoustic meatus, facial canal & stylomastoid foramen Internal acoustic meatus Via the jugular foramen Via the jugular foramen Via the jugular formanen Through the hypoglossal canal Midbrain Dorsal surface of midbrain Pons 8 Which part of the brain does CNV2 emerge from Which part of the brain does CNV3 emerge from Which part of the brain does CNVI emerge from Which part of the brain does CNVII emerge from Which part of the brain does CNVIII emerge from Which part of the brain does CNIX emerge from Which part of the brain does CNX emerge from Which part of the brain does CNXI emerge from Which part of the brain does CNXII emerge from Which cranial nerve is concerned with smell Which cranial nerve is concerned with sight Which cranial nerves are concerned with taste Which cranial nerve is concerned with hearing & balance Which cranial nerve is concerned with movement of the eyes Which cranial nerve is concerned with movement of face Which cranial nerve is concerned with sensation from the face Which cranial nerve is concerned with chewing Which cranial nerve is concerned with swallowing Which cranial nerve is concerned with movement of the tongue Which cranial nerve is concerned with movement of the vocal cords Which cranial nerve is concerned with parasympathetic visceral sensory input Which cranial nerve is concerned with movement of the neck Where are cell bodies of efferent neurones Where are cell bodies of afferent neurones What is a collection of cell bodies in the periphery called What is a collection of cell bodies in the CNS called What is the Io function of the olfactory nerve What is the Io function of the optic nerve Pons Pons Between pons & medulla Between pons & medulla Between pons & medulla Medulla Medulla Small cranial roots (medulla) and large spinal roots Medulla Olfactory Optic Facial Glossopharyngeal Vagus Vestibulocochlear Oculomotor Trochlear Abducent Facial Trigeminal Trigeminal (mandibular) Glossopharyngeal Accessory Hypoglossal Vagus Vagus Accessory Outside CNS Inside CNS Ganglion Nucleus Sensory (smell) Sensory (sight) 9 What is the Io function of the oculomotor nerve What is the Io function of the trochlear nerve What is the Io function of the trigeminal nerve What is the Io function of the abducent nerve What is the Io function of the facial nerve What is the Io function of the vestibulocochlear nerve What is the Io function of the glossopharyngeal nerve What is the Io function of the vagus nerve What is the Io function of the accessory nerve What is the Io function of the hypoglossal nerve Motor System What are lower motor neurones What happens if you get lesions in lower motor neurones What are upper motor neurones What do upper motor neurone lesions cause What happen in corticospinal lesions What is the direct control of muscles Where do segmental reflexes occur What further control are segmental reflexes subject to Which descending inputs are important in the control of muscle groups of the trunk & limbs What are the brainstem nuclei controlled by what is the direct cortical connection to the spinal cord What are the higher brain centres involved in motor control What is the sensory input to the brainstem from What happens if sensory inputs are damaged at a spinal level How can the segmental reflex be influenced by higher centres How can input to the α motorneurones effect the stretch reflex How can input to the γ motor neurones effect the stretch reflex How can input to the interneurones effect the stretch reflex What is α-γ coactivation Motor (eye movements) Motor (eye movements) Mixed (sensation from face & mouth, motor for muscles of mastication) Motor (eye movement) Mixed (muscles of facial expression, parasymp, taste) Sensory (hearing & balance) Mixed (swallowing, sensation from tongue, parasym) Mixed (muscles of throat, parasym, visceral sensory) Motor (soft palate, throat & neck) Motor (movement of the tongue) Spinal & cranial nerves that directly innervate muscles Flaccid paralysis & atrophy Neurones in the brain & spinal cord that change α motorneurone activity Spasticity, some paralysis Weakness (paresis), rather than paralysis Via α motorneurones in the ventral horn of the spinal cord At the spinal cord level Further control via brainstem nuclei, which organise them into higher order reflexes, involved in the control of posture & balance Vestibulospinal and the reticulospinal tracts Higher brain centres Corticospinal tract (AKA pyramidal tract) Cerebral cortex (motor cortex & association areas, premotor & supplementary motor areas) Basal ganglia Cerebellum Vestibular system Paralysis as if the motor neurones themselves had been damaged Directly onto α motor neurones By acting on γ motorneurones By acting on interneurones if they are strongly inhibited by descending voluntary input the stretch reflex can be overridden Descending input onto γ motorneurones can change the sensitivity of the spindles They can influence the balance between agonist & antagonist The α motorneurones are activated to make the desired movement and γ motorneurones are activated at the same time to maintain the 10 What is the crucial cord segment in the biceps jerk What is the crucial cord segment in the triceps jerk What is the crucial cord segment in the patellar tendon What is the crucial cord segment in the Achilles tendon reflex What is the significance of testing reflexes What is the Babinski sign What can cause flexor reflexes in certain sensitive parts of the body Which normal healthy group show the Babinski sign When can the Babinksi sign be seen in epileptics, why What is most of the human cerebral cortex What is the main motor area in the cerebral cortex What is the main motor area right next to What do cortical cells give rise to What are the cell bodies of cortical neurones like What % of the CS tract neurones are myelinated Where is the major decussation of the CS tract What % of the axons cross over What is the motor homunculus What sort of movements is the motor cortex involved in What does damage to the motor cortex/ CS cause Is the motor cortex involved in the initiation of movement What is the readiness potential What happens about 50ms before the movement actually occurs Why are patients surprised if you stimulate the motor cortex What is the function of the premotor cortex What is the function of the supplementary motor area Where can ↑ in electrical activity & blood flow be seen when rehearsing complex motor tasks What are the supplementary motor area and the premotor cortex part of sensitivity of the spindles to length changes C6 C7 L4 S1 Helps detect level of spinal cord damage – ability to evoke reflexes above but not below a certain level Impaired reflexes may indicate nerve damage Shows damage or disruption to CS tract, stroking along lateral aspect of foot causes toes to fan up and out Mild cutaneous stimulation Children < 1 year old whose motor system is not fully developed Transiently following an epileptic fit when cortical function is still disrupted Association cortex involved in complex processing Precentral gyrus Somatosensory area CS projections to α and γ motorneurones Large, pyrimdal shaped 50% In the brainstem 80% A topographic orderly mapping of the motor representation of the body on the motor cortex Fine precision voluntary movements Clumsiness No An ↑ in electrical activity in all cortical and some subcortical ones before a voluntary movement starts (thinking about it) Extra burst of activity in motor cortex which controls muscles needed for the desired movement = signals being sent down CS tract to activate motorneurones They move but without the thinking about it stage It is responsible for identifying goals and choosing a course of action Involved in the programming of voluntary movements Supplementary motor area The association cortex 11 What is the function of the posterior parietal cortex (PPC) What happens if there is a lesion in the PPC What are the 2 main parts of the Basal Ganglia What is the Striatum divided into What does the Striatum receive, what does it do with this Which 2 midbrain nuclei do the basal ganglia connect to What are Basal ganglia involved in What so diseases of the Basal ganglia manifest as What type of disorder in Parkinson’s disease What are the features of Parkinson’s disease What is superimposed on the main disorder in Parkinson’s disease What are the symptoms of this imposed disorder What type of disorders are Huntington’s Chorea, Athetosis and Ballism What are the symptoms of Huntington’s Chorea What are the symptoms of Athetosis What are the symptoms of Ballism Describe the main feature of cells of the Pallidum In mind of the above, what is control of movement What is the controlling feature of the striatum What neurotransmitter does the striatum use Where does organising cortical input arrive via What are the processes going on to keep a person at rest What happens when a voluntary movement is to be initiated What happens to cause Huntington’s Chorea What happens to cause Ballism What keeps the inhibitory pathway from the striatum to the pallidum in check Essential for the processing of sensory stimuli leading to purposeful movements “neglect syndrome” – an inability to respond to stimuli on the contralateral side of the body – patients may deny contralateral limb is theirs Striatum Globus pallidus (pallidum) Caudate Putamen A copy of the readiness potential, which is processed by the striatum & pallidum then relayed via the thalamus to the cortex & premotor cortex Substantia nigra Subthalamic nucleus Processing the readiness potential so that initiation of voluntary movement can occur Difficulties in starting voluntary movements and stopping unwanted movements Hypokinetic disorder – there is akinesia Extreme difficulty in starting voluntary movements, also a lot of associated movements e.g. swinging arms when walking, loss of facial expression Hyperkinetic features Resting tremor = shake at rest, tremor disappears when movement is in progress Rigidity - ↑ muscle tone which affects both flexors & extensors Hyperkinetic disorder Jerky spasmodic movements at ‘rest’ and incorporated into ‘normal’ voluntary movements Slow writhing movements which affect the extremities – adopt grotesque postures Violent flailing of the limbs They have a high level of spontaneous activity Voluntary regulation of this spontaneous activity It exerts a profound inhibitory effect on the pallidum GABA The striatum The ‘organising cortical input’ excites the striatum, which then inhibits the pallidum Patterned inhibition in the form of the readiness potential allows organised excitation to be transferred from the pallidum to the motor cortex There is striatal degeneration, so that the organising cortical input is lost, so the spontaneous activity of the pallidum passes unchecked to the MC There is degeneration of the subthalamic nucleus, releasing pallidal activity The inhibitory pathway from the substantia nigra to the striatum = nigrostriatal pathway 12 What is the function of keeping the above pathway in check What neurotransmitter does the above pathway use What happens to cause Parkinson’s disease What is the main characteristic of Parkinson’s disease What % of SN neurones degenerate before clinical symptoms of Parkinson’s appear In treating Parkinson’s disease what are you trying to compensate for Why do you give L-DOPA What is the problem with giving L-DOPA How can the efficacy of L-DOPA be improved What catalyses the breakdown of LDOPA How long is L-DOPA affective in treating Parkinson’s What can long term treatments with LDOPA or other DA agonists produce What is schizophrenia associated with What are the ‘possible’ new treatments for Parkinson’s What is the pathway that produces DA, what can DA go on to produce What are the 2 parts to the inhibitory striatal pathway to the pallidum Why can drugs which block ACh be effective at ↑ the effective of remaining dopaminergic neurones What is the aim of the treatment of Huntington’s chorea What sort of treatments are used in Huntington’s chorea What can be the side effects of the drugs used to treat Huntington’s chorea How is Huntington’s disease inherited Cerebellum What is the cerebellum responsible for What is co-ordination What is the structure of the cerebellum What are the 3 functional parts of the cerebellum What is the function of the archicerebellum It ensures that the pallidum does not get too much inhibition Dopamine Degeneration of the substantia nigra pathway It is very hard to initiate a voluntary movement >80% Loss of domaminergic neurones L-DOPA is the precursor of dopamine, L-DOPA can cross the BBB but dopamine can’t It is taken up by many tissues in the body so that < 0.01% of an oral dose reaches the substantia nigra Combining L-DOPA with a selective extra-cerebral decarboxylase inhibitor, which reduces peripheral breakdown L-DOPA → dopamine, catalysed by DOPA decarboxylase 5-7 years Symptoms of psychosis Overactivity of dopaminergic pathways Autotransplantation of adrenal medulla (DA is precursor of NE) Foetal transplants Target viruses to carry tyrosine hydroxylase gene to sites of DA synthesis Stem cells Phenylalanine → Tyrosine → L-DOPA → Dopamine → N.E → E Striatal cholinergic interneurones onto inhibitory GABA neurones from the striatum to the pallidum Both the cortical input & nigrostriatal inhibition are onto striatal cholinergic neurones ↑ efficacy of remaining striatal interneurones ACh mimetics & DA blockers They may produce Parkinsons As an autosomal dominant disease Co-ordination of voluntary movements Control of the timing of sequences of contractions in different muscles to make a movement smooth & reliable Highly convoluted grey matter cerebellar cortex, beneath which is white matter containing axons going to & from the cortex. Embedded in the white matter are the deep cerebellar nuclei Archicerebellum (vestibulocerebellum) Paleocrebellum Neocerebellum Oldest part – concerned with posture & balance, major input from vestibular system 13 What is the function of the Paleocerebellum What is the function of the neocerebellum What is the largest functional part of the cerebellum in man Where is all input to the cerebellum to Where is all cerebellar output from What are the 2 routes via which information can reach the cortex What are the connections from the pons to the cerebellar cortex What are the connections from the inferior olive to the cerebellar cortex What does the cerebellum receive from the CS tract, via what What is the major input to the cerebellum What causes an error signal Where is an error signal sent to What does the cerebellum carry, which is the same simple circuitry repeated millions of times What cells are the only output of the cerebellar cortex What do mossy fibres connect with What happens when mossy fibres make their connections What do indirect climbing fibres make connections with What happens when indirect fibres make their connections What is motor learning about What causes synaptic strengthening What is the result of synaptic strengthening Are climbing fibres needed when the movement becomes automatic Is synaptic strengthening permanent How is co-ordination achieved What is the result of purkinje cell discharge What is passed from the dentate nucleus to the MC What is ataxia How does cerebellar damage affect the appearance movements How can cerebellar damage affect muscles What tremor is associated with cerebellar damage What is dysdiadochokinesia Important in 4 legged animals for the control of gait Unconscious co-ordination of voluntary movements Neocerebellum – 90% in man To the cortex, cerebellar hemispheres The nucleus (dentate in the case of the neocerebellum), relayed via the thalamus to the motor cortex Direct route via pons of brainstem Indirect route, which traverses several brain areas and finally makes connections in the inferior olive of the brainstem Mossy fibres Climbing fibres Collaterals of CS tract pass to cerebellum, copy of signals being sent to the motor neurones Readiness potential – copy of movement desired A discrepancy between the readiness potential and the copy of the signals being sent to the motor neurones The motor cortex Motor homunculus Purkinje cells Each mossy fibre makes connections with > 1000 purkinje cells Mossy fibres raise the level of excitation in purkinje cells, but does not cause APs to be generated (in the purkinje cells) 1:1 contact with purkinje cells Cause APs in Purkinje cells which inhibit the dentate nucleus Selecting the right climbing fibre to give you the effect that you want It is a result of joint activation of mossy fibre & climbing fibre inputs to a purkinje cell Eventually mossy fibre input on its own is enough to cause APs to be discharged in purkinje cells No Yes – it involves ↑ permeability to Ca2+ and protein synthesis Mossy fibre excitation sweeps along the array of purkinje cells, there is separation between their excitation, the purkinje cells picked out by synaptic strengthening will discharge in a temporal pattern It inhibits the discharge of the dentate nucleus (which also caused by mossy fibre excitation) A temporal code of excitation, caused by purkinje cells inhibiting the discharge of the dentate nucleus In-coordination Ataxia Decomposition of movement – e.g. joint by joint, also affects speech Asynergia – lack of cooperation between agonists & antagonists, they may contract together Intentional tremor – occurs during a movement and particularly towards the end – no tremor at rest Inability to make rapid alternating opposite movements 14 Why does cerebellar damage affect posture, gait and balance Vestibular System Where is the vestibular system found What are labyrinths What do labyrinths detect What are the sensory cells in the vestibular system What features do these sensory cells have What causes these sensory cells to discharge action potentials What causes a decreased discharge of AP from the sensory cells What is tilt of the head detected by What is side to side tilt detected by What is forward to back tilt detected by What are the cilia in macula organs embedded in How are otoliths different to endolymph What happens to otoliths when the head is tilted, what does this cause What are the semi-circular canals (SCCs) like What are the swelling in the SSCs, what is in them What do sensory cells SSCs detect What happens to the endolymph in SSCs when the skull initially at rest is turned How does the above affect the cilia What happens if you rotate at a constant velocity What does the posterior SSC detect What doe the anterior SSC detect What does the horizontal SSC detect What does information from the SSC & macula organs pass along Where does information from the SSC & macula organs initially go to What do vestubular nuclei project to in the opposite cortex What do vestibular nuclei also receive input from How do the vestibular nuclei contribute to kinasthesia What is kinaesthesia What tracts are involved in reflexes mediated by the vestibular system What is the function of the ‘Tonic labyrinthine reflex’ Where does the ‘Tonic labyrinthine reflex’ get its information from Relates to use of information from the vestibular system – adopt wide swaying gait In the inner ear, it is a series of fluid-filled membranous tubes, which are embedded in the temporal bone A series of fluid-filled membranous tubes Tilt of the head with respect to the body Angular accelerations Hair cells, whose cell bodies are embedded in the bone of the skull They have a set of cilia, which protrude into the endolymph, there is a large kinocilium and progressively smaller stereocilia Distortion of the cilia in the direction of the kinocilium causes depolarisation & ↑ discharge of AP Distortion of the cilia away from the kinocilium leads to hyperpolarisation & ↓ discharge of AP Macula organs (utriculus & sacculus) Utriculus Sacculus A blob of jelly which contains crystals of CaCO 3 = otoliths Otoliths have a greater density than endolymph and so are not effeted by gravity The blob of jelly (otolith) is distorted causing shearing forces on the cilia They are like closed tubes of endolymph Swellings = ampullae in which are the cupula organs (hair cells) Angular acceleration The endolymph at first tends not to move because of its inertia The base of the cilia move because they are embedded in the bone, this produces shearing forces which bend the cilia The endolymph rotates at the same speed so there are no shearing forces Head over heals rotation Side to side rotation Spinning CNVIII (vestibulochoclear) vestibular nerves Vestibular nuclei & then make lots of connections The other set of vestibular nuclei Proprioceptors signalling limb & body position, also from neck & eye muscles Vestibular nuclei project via the thalamus to the cerebral cortex Perception of movement & body position Vestibulospinal tracts Reticulospinal tracts Keeps the axis of the head in a constant relationship with the rest of the body Macula organs and neck proprioceptors 15 What is the ‘Dynamic righting reflex’ What do the Vestibulocochlear reflexes involve What are the 2 vestibulocochlear reflexes What is the ‘Static reflex’ What is the input to the ‘Static reflex’ What is the ‘Dynamic reflex’ another name for What is the ‘Dynamic reflex’ Why is the reflex dynamic What causes a right nystagmus What is the conventional direction of nystagmus What can nystagmus be used to test What are the 22 tests that can be done What is tested by caloric stimulation What does washing with 44oC H2O cause What does washing with 30oC H2O cause What would be seen if you put warm water into right ear What ahs to be done to visual input to test vestibular function What can result from powerful maintained stimulation of the vestibular system When is motion sickness most likely to occur What happens in motion sickness How do trained ballerinas avoid motion sickness What are the symptoms of acute interference of normal vestibular function What is vertigo What happens in Meniere’s disease What’s Menieres disease associated with Can vestibular impairement be compensated for What can cause nystagmus at rest What is the dominant input to the archicerebellum EEG/Sleep What are the 2 ways of assessing someone’s consciousness Rapid postural adjustments that are made to stop you falling when you trip. Long reflexes, involving extension of all limbs Eye movements & the control of the extraocular muscles Static reflex Dynamic reflex When you tilt your head your eyes roll to compensate, so that over a certain range, the image stays the right way up Macula input plus proprioceptors from eye muscles Vestibular nystagmus A series of eye movements that rotate the eye against the direction of rotation of the head so that the original direction of gaze is preserved The extent of eye movement is restricted, when eye muscles come to the end of their range, they rapidly flick back to the zero position straight ahead & the slow phase starts again Right rotation Same as the direction of rapid flick back Vestibular function Post-rotatory nystagmus (opposite direction to nystagmus during rotation) Caloric stimulation – outer ear washed with cold or warm fluid, temp difference (from core temp) sets up convection currents which affect endolymph Horizontal SSC and associated circuitry Nystagmus towards affected side Nystagmus away from affected side Right nystagmus Get rid of visual input – patients wear Freznel’s spectacles which prevent visual fixation Kinetosis (motion sickness), nausea & vomiting, ↓ BP & dizziness, sweating & pallor = all are autonomic NS symptoms If visual & vestibular system inputs to the archicerebellum are in conflict e.g. vestibular system indicates rotation but visual system doesn’t The archicerebellum generates a ‘sickness signal’ to the hypothalamus to bring about the ANS changes They learn to visually fixate All ANS symptoms plus vertigo, there will also be nystagmus The perception of movement in the absence of movement Vertigo, nausea, nystagmus & tinnitus (subjective noise) Over production of endolymph Yes – by visual system Lesions of the brainstem Visual input By their behaviour By their brain activity 16 What can brain activity be analysed in terms of What is the EEG of the awake state characterised by What does amplitude show What does frequency show What is the α rhythm What is the β rhythm When do EEG have a low amplitude & high frequency, why What are the other names for REM sleep What are other names for non-REM sleep What happens during S-wave sleep How many stages of S-sleep are there Which is the deepest stage of S-sleep What NS is dominant in deep S-sleep What happens at each successive stage of S-sleep How does sleep begin (stages) What happens when deep S-sleep is reached What is the EEG of REM sleep like Why is REM sleep called paradoxical sleep When is brain O2 consumption highest What are the physiological signs during REM sleep When is dreaming likely to occur What is the first period of REM sleep like What happens to periods of REM sleep throughout the night What happens to S-sleep throughout the night What is stage 3 & 4 sleep When does stage 3 & 4 sleep occur What happens to total sleep time during childhood & adolescence What happens to REM sleep during life What is chronic insomnia What is the action of many hypnotic drugs, especially barbiturates What happens when the above drugs are withdrawn What are nightmares What can be recorded during dreams What do dreams do to the motor system What are night terrors Amplitude frequency Low amplitude, high frequency waves The bigger the wave the more the activity of the cells is synchronised The higher the frequency, the faster the rate of fluctuations in the cortical cells In relaxed state with eyes closed – frequency becomes slower & amplitude becomes greater In the alert state – higher frequency & smaller amplitude When cells are busy all doing different things rapidly, because they are not synchronised D-sleep, paradoxical sleep Slow wave sleep, S-sleep There is a gradual ↑ in amplitude of EEG waves as the activity of more and more of the cells are synchronised and the frequency is slow 4 4 – person is difficult to rouse Parasympathetic dominance – e.g. ↓HR As you move from 1→4 the EEG pattern develops a slower frequency and higher amplitude Progression from 1→4, normally takes 30-45 mins The process reverses itself – there is a gradual awakening towards stage 1 sleep but instead of stage 1 there is REM sleep EEG waves are desynchronised, high frequency, low amplitude It has an EEG very like the awake state but the sleeper is difficult to rouse During REM sleep Profound inhibition of body muscles Except for eye muscles which show bursts of activity Sympathetic dominance - ↑ HR & BP, ↑ cerebral BF, ↑ brain temperature During REM sleep Short – 5 – 10 mins REM periods increase in duration throughout the night It becomes shallower Delta sleep – restorative, repairing sleep, ↑GH In first half of the night Declines rapidly Declines from 50% at full term to stable 25% in adulthood Chronic inability to obtain the necessary amount of sleep to maintain adequate daytime behaviour Depress REM sleep and delta sleep ‘rebound effect’ – there is a marked and immediate ↑ in the quantity of REM sleep – as if to catch up They have strong visual components & are seen during REM sleep PGO spikes (pontine-geniculo-occipital) They shut it off – maybe to stop us acting out our dreams They are associated with intense feelings of fear & are a non-REM phenomena 17 What is Somnambulism When does somnambulism occur What is Narcolepsy What stage of sleep are narcoleptics most likely to enter What causes narcolepsy What happens to the cerebral cortex if it is left to itself What is a major area of the brain that controls arousal What is the ARAS Where does the ARAS receive sensory input from, where does it connect to What happens if you stimulate the ARAS What happens to the ARAS for sleep Where is it thought that the sleep centres in the brain are What are cycles of S→D & D→S due to What is the GTF What does the GTF consist of What happens in GTF cells How does the GTF affect the cortex Why do cholinesterase inhibitors ↑ the duration of REM sleep What is the locus coeruleus (LC) What is the function of the locus coeruleus How does the LC affect sleep What sort of rhythm does sleep show Where is the ‘master clock’ for biological rhythms What happens to sleep if the SCN is destroyed Limbic System What are the limbic system & the hypothalamus responsible for What happens if you electrically stimulate a reward area What happens if you stimulate a punishment area What is the motivation to learn What are psychoses What is schizophrenia What is schizophrenia characterised by (symptoms Sleep walking Exclusively in non-REM sleep, mainly in stage 4 sleep An irresistible urge to sleep Directly into REM sleep A 90% loss of orexin producing neurones, orexin stimulates wakefulness It reverts to slow waves (intrinsic property), so without other influences cortical cell activity becomes synchronised – so removed cortex from ascending inputs = S-wave sleep The ‘Ascending reticular activating formation’(ARAS) = part of the reticular formation in the mid-brain Part of the reticular formation of the mid-brain All the major sensory systems & it makes connections with the whole of the brain & spinal cord Arousal from sleep It is inhibited Brainstem, hypothalamus, preoptic area Oscillatory activity in 2 areas of the reticular formation Gigantocellularis tegmental field Very large cells with long axons and dendrites that make multiple connections throughout the brain They exhibit oscillatory activity, bursts of APs appear just before & during REM sleep GTF is the origin of PGO spikes which give the visual component to dreams It modifies cortical activity directly through the ARAS Neurones in the GTF are cholinergic, cholinesterase inhibitors cause S→D sleep to occur Another pontine area It acts as an oscillator with the GTF, when LC cells are active GTF are not Opposite to GTF – causes D→S sleep to occur, causes switch from REM sleep to S-wave sleep Circadian = oscillations within a 24 hour period The Suprachiasmatic nucleus (SCN) of the hypothalamus, lying just above the optic chiasma The circadian rhythm of sleep is lost Instinctive & emotional behaviour, e.g. drives for thirst, sex, hunger. It is also important in reward & punishment aspects of behaviour and memory Gives intense feelings of well being, euphoria and sexual arousal Give feelings of terror, anger or pain Learn to gain a reward or avoid a punishment A group of disorders which represent endogenous inherent malfunction of the brain, e.g. schizophrenia & manic depression – relationship between behaviour and reward & punishment areas seems to be impaired A dissociation of the cognitive side of behaviour from the affective or emotional side – not split personality Bizarre delusions – or being persecuted, Auditory hallucinations – hearing voices commenting on their actions 18 What is the action of drugs used to treat schizophrenia What can be the side effects of the above drugs What happens in disruption of nonadrenergic pathways in the limbic system How can NE effect mood How is serotonin related to mood Memory What is episodic memory What is semantic memory What is non-declarative memory Where is non-declarative memory based How long is immediate memory How long is short-term memory How long is long-term memory What is short-term memory Where is short-term memory stored How is short-term memory maintained What is retrograde amnesia What causes retrograde amnesia Where is the memory store How is short-term memory converted to long-term memory What assesses the significance of an event in short-term memory What does the frontal cortex do if the memory is considered useful Where is memory laid down in the cerebral cortex What are the most powerful stimuli at evoking long-term memories What is the hippocampus important in What happens if you bilateral hippocampal damage What is Korsakoff’s syndrome What happens in Alzheimer’s disease What can be used to treat Alzheimer’s How can REM sleep be linked to memory What happens to REM sleep in Alzheimer’s & Korsakoff’s syndrome How may dreaming affect memory Sensory Systems What is the transduction action of all sensory receptors What encodes the intensity of a stimulus They interfere with dopamine transmission, mainly by blocking DA receptors They can interfere with the dopaminergic receptors in Basal ganglia causing Parkinsonism It is thought to be implicated in clinical depression ↑ ECF [NE] causes elation, ↓ ECF [NE] causes depression Serotonin causes elation – so can treat depression by blocking serotonin reuptake Memory for events Memory for words, rules and language Relates to reflexive skills Cerebellum Few 100ms → 20 secs 1 sec → a few hours 30 mins → decades, centuries An electrical phenomenon Largely in the cerebral cortex It needs maintained excitation from reverberating circuits Inability to recall immediate events for up to 4 hours before loss of consciousness Refreshing effect (reverberating circuits) of short term memory is interrupted Sensory & association areas of the cortex By a process of consolidation – this involves selective strengthening of synaptic connection by repeated use The frontal cortex and its association with the reward/punishment centres in the limbic system ‘gates’ Papez circuit, so reverberating activity then continues between the Papez circuit, frontal cortex, the sensory & association areas until consolidation is complete Different components in different places – e.g. visual components in visual cortex Olfactory stimuli Consolidation process You have immediate memory and intact long-term memory but are unable to form new long-term memories In chronic alcoholism, there is vitamin B1 deficiency & malnutrition causes mamillary body damage – the ability to consolidate memory is impaired There is a severe loss of cholinergic neurones throughout the brain, including the hippocampus Anticholinesterases - but underlying degeneration continues Deprived of REM sleep → impairment of memory consolidation for complex cognitive tasks Patients have greatly reduced REM sleep – cholinergic neurones responsible for REM sleep – dreaming may enable memory consolidation, reinforce weak circuits Dreaming may help forget memories that are no longer useful Dreaming may get rid of errors (baby birds!) All sensory receptors transducer their adequate stimulus into a depolarisation, the receptor (generator) potential The size of the receptor potential 19 What enables long distance transmission How do APs encode stimulus intensity What are the 3 types of primary afferent fibres that mediate cutaneous sensations, what are their properties What is proprioception mediated by Where do all Io afferent fibres enter the spinal cord What are the mechanoreceptive fibres Where do the above fibres project Where do the above fibres synapse What happens to the 2nd order fibres What are thermoreceptive & nociceptive fibres What happens if you damage dorsal columns What happens if you damage anterolateral quadrant Where is the ultimate termination of semsory information What produces the sensory homunculus What are the effects of convergence What is lateral inhibition What are the effects of lateral inhibition Physiology of pain What usually elicits a sensation of pain What are the two type of pain How can somatic pain be divided up What are pain receptors called What are pain receptors What are the 2 classes of pain receptors, what are there properties What sensations are part of ‘initial’ pain What sensations are part of delayed pain What nociceptors sense initial pain What nociceptors sense delayed pain What is the neurotransmitter released by first order (C) neurones in pain fibres Do pain fibres cross the mid-line What is hyperalgesia What is the function of substance P Where is histamine released from How does aspirin work What is analgesia Action potentials which are evoked by receptor potentials By frequency of action potentials Aβ – large myelinated (30-70m/s) touch, pressure, vibration Aδ – small myelinated (5-30m/s) cold, ‘fast’ pain, pressure C – unmyelinated fibres (0.5-2m/s) warmth, ‘slow’ pain 2 types of Io afferent fibres – Aβ & Aδ e.g. muscle spindles, GTOs Via the dorsal root ganglia (or cranial nerve ganglia for the head) Aα & Aβ fibres Though ipsilateral dorsal columns In cunate & gracile nuclei Decussate in the brain stem & project to the reticular formation, thalamus & cortex Aδ & C fibres, synapse in dorsal horn, project through contralateral spinothalamic tract to reticular formation, thalamus & cortex Loss of touch, vibration, proprioception below lesion on ipsilateral side Causes loss of temperature sensation below lesion on contralateral side The somatosensory cortex of the postcentral gyrus Nerve endings are grouped according to location of receptors, extent of representation is related to the density of receptors in each location Saves on neurones Reduces acuity Underlies referred sensations Activation of one sensory fibre causes synaptic inhibition of its neighbours Gives better definition of boundaries Cleans up sensory information A stimulus that causes or is on the verge of causing tissue damage Somatic Visceral Superficial – skin Deep = connective tissue, bones, joints, muscles Nociceptors Free nerve endings of primary sensory neurones Aδ – thin myelinated, medium diameter C – unmyelinated, small diameter Sharp, pricking, well localised pain Dull, intense, diffuse Aδ C Substance P Yes Increased sensitivity to painful stimuli, the pain can last for hours after the original stimulus is over Causes release of substances from mast cells, which depolarise C fibre nerve endings – causing action potentials Mast cells & damaged cells It is a cyclo-oxidase inhibitor, cyclo-oxidase is the enzyme that converts arachadonic acid to prostaglandins The selective suppression of pain without effects of consciousness or other sensations 20 What are the 2 components of pathiological pain How are nociceptors different from other receptors Where are the cell bodies of spinal nociceptive afferent fibres How do nociceptive afferent fibres enter the spinal cord, what do they then do What type of receptor is a properties of Aβ fibres What type of receptors are properties of Aδ fibres What type of receptors are a property of C fibres What are the 2 main ascending pathways to the CNS What is the neurotransmitter released in the dorsal horn in the spinothalamic pathway How many neurones are in the spinothalamic tract Which neurone crosses the mid-line What does the 1st order afferent neurones synapse with in the Spino-reticulo-thalamic tract What do touch fibres & pain fibres both synapse onto What is the ‘Gate control theory’ What activates the inhibitory interneurones in the ‘Gate control theory’ What inhibits the inhibitory interneurones How can the gate control theory provide natural pain relief What is the function of descending pathways Where is the Periaqueductal grey (PAG) What does the PAG receive What does the PAG release, what is the effect of this What are enkephalins/endorphins produced by the brain What does the above imply about inhibitory interneurones in the gate control system Why is visceral pain poorly localised What happens in referred pair The peripheral nociceptive afferent neurone, which is activated by noxious stimuli The central mechanisms by which the afferent input generates a pain sensation They have a higher threshold, since they are normally only activated by stimuli of noxious intensity Dorsal root ganglia Via the dorsal roots, ending in the grey matter of the dorsal horn, where they give rise to the main projection pathways from the dorsal horn to the thalamus Mechanoreceptors Mechanoreceptors Nociceptors Mechanoreceptors Nociceptors Thermoreceptors Spinothalamic tract Spino-reticulo-thalamic tract Substance P 2 2nd order afferent neurone An opiate interneurone Inhibitory interneurones It regulates the passage of impulses from the peripheral afferent fibres to the thalamus Descending inhibitory interneurones Non-nociceptive input (Aα/Aβ fibres) Nociceptor C fibre input Rubbing the affected area stimulates the low threshold inputs onto the inhibitory interneurone (Aα/Aβ fibres), which excites the inhibitory interneurone, causing ↓ dorsal horn neural activity to the CNS (thalamus) One of the gating mechanisms that control impulse transmission in the dorsal horn An area of the midbrain – a small area of grey matter surrounding the central canal Input from many other brain regions, including the hypothalamus, cortex & thalamus An endogenous morphine like substance (opiod) which ↓ pain severity = descending inhibitory pain modulation Endogenous morphine-like substance – fits into opiod receptor They must have opiod receptors, opiod interneurones can block pain transmission at 2 sites, block neurotransmitter release from I o afferent, inhibit IIo afferent There are few receptors Visceral pain is referred to a somatic structure from the same dermatome, referred pain is due to convergence – there are many more Io afferent fibres that 2nd order dorsal horn neurones 21 Why does referred pain occur Pharmacology of pain relief What are natural morphine-like drugs What are semi-synthetic morphine-like drugs What are synthetic morphine-like drugs What are the effects of morphine-like drugs What type of pain are morphine-like drugs most effective against What is the evidence that effects of morphone are receptor mediated What are the sub-types of opioid receptors Why does morphine work in the body What are the mechanisms of action of morphine Where are the sites of action of morphine What is the commonest cause of death in opiod poisoning Pharmacology of anaesthesia How do local anaesthetics work What are the 2 types of general anaesthetics What are the stages of general anaesthesia What are the sough after effects of general anaesthesia What are the unwanted effects of general anaesthesia What type of transmission are general anaesthetics most effective against Why are the target for inhalation general anaesthetics probably not a receptor binding site There is somatic & visceral input onto the same dorsal horn neurone Morphine Codeine, heroine Pethidine, methadone Analgesia – against acute & chronic pain Suppression of cough reflex (anti-tussive) Elevation of mood (euphoria) – anxiety & agitation removed Depression of respiration Contraction of pupils – important diagnostic feature in overdose Stimulates histamine release form mast cells – itching at injection site or bronchoconstriction – don’t give to asthmatics Increases tone in smooth muscle Reduces smooth muscle activity e.g. gut Dull constant pain e.g. tissue injury, inflammation & cancer (tumour growth) High potency Marked stereoselectivity Selective antagonists - naloxone High affinity binding sites exist Opioid receptors have been cloned μ,δ,κ,σ – all g-protein coupled receptors, main pharmacological effects of morphine mediated by μ receptors Morphine taps into the endogenous signalling system for endorphins Same as endogenous opiods) It interrupts nociceptive transmission In the spinal cord (substantia gelatinosa)– activation of μ receptors on central terminals of nociceptive afferent inhibits transmitter release In brain stem – activation of μ-receptors in PAG, stimulates descending pathways Respiratory depression – results in ↑ arterial Pco2 associated with a ↓ in the sensitivity of the respiratory centre to Pco2 Block voltage-dependent Na+ channels, preventing APs in afferent & effererent axons Inhalation anaesthetics – nitrous oxide, ether, chloroform Intravenous anaesthetics – thiopental, ketamine Stage I – analgesia Stage II – excitement – dangerous stage Stage III – surgical anaesthesia Stage IV – medullary paralysis - lethal Loss of sensation Loss of consciousness Muscle relaxation Respiratory depression Cardiovascular depression Loss of reflexes More effective against synaptic transmission than axonal conduction – disruption of synaptic transmission in reticular formation & thalamus Low potency No structure-activity relationships No competitive antagonists No high affinity binding sites 22 What does potency of inhalation general anaesthetics correlate with, why? If substances dissolve in oil are they a good anaesthetic What could happen if an anaesthetic dissolved in a membrane How do barbiturates work (intravenous) How does ketamine work (intravenous) Tolerance & Dependence Describe long acting barbiturates Name 2 long acting barbiturates Describe shorter acting barbiturates Name a shorter acting barbiturates Describe very short acting barbiturates Name a very short acting barbiturate What do barbiturates bind to What has to be present for barbiturates to have an effect What is the effect of barbiturates What are the acute effects of barbiturates (↑ dose down list) What are the therapeutic uses of barbiturates Describe long acting benzodiazepines Describe short acting benzodiazepines What do benzodiazepines bind to What is the effect of benzodiazepines What are the acute effects of benzodiazepines What are the therapeutic uses of benzodiazepines What is Tolerance What are the possible mechanisms of tolerance What are the possible mechanisms of dispositional tolerance What are the possible mechanisms of pharmakodynamic tolerance What are the 2 types of dependence Oil:gas partition coefficient – compounds interact with something hydrophobic to produce anaesthesia Yes Increased memory fluidity – unlikely Volume expansion – explains pressure reversal of anaesthesia Bind to GABAA receptors * potentate GABA transmission = IPSPs Blocks NMDA receptor-gated channels, reduces glutamate transmission (glutamate – main excitatory transmitter in brain) Slow metabolism & excretion Barbitone, phenobarbitone Faster metabolism than long acting barbiturates Pentobarbitone Rapid redistribution from CNS to other tissues Thiopental Binds to subunit of GABAA receptor GABA Increases open time of channel when GABA binds – potentiating GABAergic IPSPs – increasing synaptic inhibition Anxiolytic Hypnotic/sedation Muscle relaxant Causes loss of consciousness Depresses respiratory centre – stops sensitivity to CO2 Treat epilepsy - phenobarbitone Induce and/or maintain anaesthesia - thiopentane Metabolised to active metabolites – diazepam Metabolised to inactive metabolites – temazepan Subunit of GABAA receptor ↑ affinity of GABA receptor for GABA – potentiating GABAergic IPSPs – ↑ synaptic inhibition Anxiolytic Hypnotic/sedation Muscle relaxant Anticonvulsant Causes loss of consciousness Relive anxiety Promote sleep Induce basal anaesthesia - diazepam Treat some types of epilepsy - diazepam Reduced responsiveness to a drug caused by previous administration – start needing higher concentrations to get same effect Dispositional – less drug reaches the active site Pharmacodynamic – site of action is less affected by the drug ↓ rate of absorption ↑ rate of metabolism to inactive metabolites ↓ rate of metabolism to activate metabolites ↑ rate of excretion Down-regulation or internalisation of receptors Reduced signalling downstream of receptors Caused by a physiological disorder e.g. diabetes Drug induced dependence 23 What is drug induced dependence caused by What are reward properties caused y What is the psychological component of dependence What is the general physiological effect of drugs of depenedence What is the specific effect of heroin What is the specific effect of amphetamines What is the specific effect of cocaine How can drug seeking behaviour be reduced What is the withdrawal phenomena What is the withdrawal effect of barbiturates What is the withdrawal effect of opioids What is the withdrawal effect of cocaine What factors influence dependence liability of a drug Why is heroine very addictive Brain death What is a coma What signs does a person in a coma exhibit What happens to sleep/wake cycles during a coma What can coma result from What often happens to a person in an irreversible coma When does brain death occur Why must the cause of coma be known for a diagnosis of brain death How long can a respirator be switched off for to check for spontaneous respiration How can you check cerebral & brainstem functions How must a brainstem death diagnosis be made Rewarding properties of the drug Withdrawal phenomena after repeated use Stimulation of central ‘reward pathways’, especially mesolimbic dopaminergic neurones Craving They increase dopamine levels in the nucleus accumbens Increases firing rate Increases dopamine release Inhibits dopamine uptake Lesion of medial forebrain bundle Dopamine receptor antagonists Withdrawal effect is the reverse of the acute effect – happens because of physiological adaptation to the drug effect (tolerance) Acute effect = anticonvulsant, so withdrawal effect = convulsions Acute effect = constipation, so withdrawal effect = diarrhoea Acute effect = elevated mood, so withdrawal effect = depressed mood Strength of effect on reward centres vs strength of any aversive effects Strength of withdrawal symptoms Discriminative effect of the drug It crosses BBB quickly so there is a strong association between heroine & reward A severe ↓ in mental function due to structural, physiological, or metabolic impairment of the brain Sustained loss of the capacity for arousal even in response to vigorous stimulation, no outward behavioural expression of any mental function, eyes are closed They disappear Extensive damage to the cerebral cortex Damage to the brainstem arousal mechanisms Interruptions of connections between the brainstem and cortical areas Metabolic dysfunctions Brain infections Overdose of certain drugs e.g. sedatives, narcotics or ethanol Enter a persistent vegetive state where sleep/wake cycles are present, they can smile, cry but there is no evidence that they can comprehend these behaviours When the brain no longer functions and has no possibility of functioning again Comas caused by drug poisoning are usually reversible 10 mins – longer causes further brain damage due to lack of oxygen No response to painful stimuli above the spinal cord Pupils unresponsive to light Apnea for 10 mins Twice by 2 senior doctors within 24 hours, all brainstem reflexes should be absent 24