Section Summary 25.1
advertisement

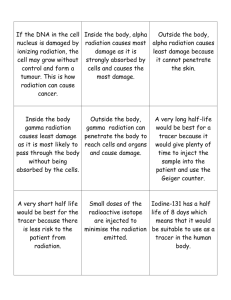
SECTION SUMMARY 25.1 Nuclear Radiation Summary: Isotopes with unstable nuclei are radioactive and are called radioisotopes. The nuclei of radioisotopes decay to stable nuclei plus emission of large amounts of energy. The radiation may be alpha, beta, or gamma. Alpha radiation consists of alpha particles (positively charged helium nuclei) that are easily stopped by a sheet of paper. Beta radiation is composed of fast-moving particles, which are electrons. Beta radiation is more penetrating than alpha radiation. It is stopped by metal foil. Gamma radiation is electromagnetic radiation. Gamma radiation has no mass or electrical charge. It is extremely penetrating. Concrete bricks and lead reduce the intensity of gamma radiation but do not completely stop it. When a radioisotope decays, beta radiation sometimes is emitted. U + 10 e 239 This equation is never true because in beta radiation, the 92 U neutron breaks apart into a proton, which remains in the nucleus, and a fast moving electron which is released. The following beta radiation is correct: 238 92 238 92 U 0 1 e + 238 93 Np Also, in beta radiation when the neutron breaks apart into a proton, causing the atomic number increase by 1 and the mass number stay the same. 25.1 Nuclear Radiation Vocabulary Terms: radioactivity: the process by which nuclei emit particles and rays radiation: the penetrating rays and particles emitted by a radioactive source radioisotopes: an isotope that has an unstable nucleus and undergoes radioactive decay alpha particle: a positively charged particle emitted from certain radioactive nuclei; it consists of two protons and two neutrons and is identical to the nucleus of a helium atom beta particle: an electron resulting from the breaking apart of neutrons in an atom gamma ray: a high-energy photon emitted by a radioisotope The End of the Summary