CHEM 3001 Laboratory Manual
advertisement

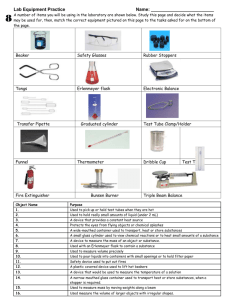
SYLLABUS CHEM 2001 ORGANIC CHEMISTRY LAB Fall 2012 Instructor: Dr. C.A. Hansen Phone, Office, Email: (940) 397-4285 Bolin Science Hall 307C chris.hansen@mwsu.edu Office Hours: MWF 9-11 TR 9-11 Text(s): -CATALYST- Organic Chemistry I and II. -Laboratory Research Notebook, 1994, Jones and Bartlett Publishers, Inc., MA, ISBN 0-86720-877-5. -Approved Safety Goggles for wearing in lab. -Laboratory Marker (Sharpie) Prerequisites: CHEM 1241 and concurrent enrollment in CHEM 2003 Grading Procedure: Lab reports: Lab quizzes: Lab final: Attendance Policy: 55% 15% 30% Attendance is expected as stated in the Student Handbook. General Education Statement: Students in this course must demonstrate their competency in written communication, some fundamental math skills, and basic use of computers through exams and written reports Course Content: This class provides laboratory experiments to accompany CHEM 2003. You will learn separation and purification techniques and quantitative methods in organic chemistry. You will also be introduced to organic synthesis. 1 2 Laboratory Schedule for Fall 2012: Date Exp. # Topic Quiz Techniques Pages Yes or no Week of 27-Aug Laboratory Check-In, Safety 4-Sep 1 10-Sep 2 17-Sep 3 24-Sep 4 1-Oct 5 8-Oct 6 15-Oct 7 22-Oct 8 29-Oct 9 5-Nov 10 12-Nov 10 26-Nov Equipment and Techniques. OP 1, 4, 5, 18 Crystallization OP 7, 8, 10, 28, 33 Extraction OP 18, Appendix Thin Layer Chromatography OP 22 Alkyl Halides from alcohols OP appendix, 34, 39, Klein Chap. 7 Nucleophilic Substitution Reactions Klein Chap. 7 Methylcyclohexene OP 30, 39, Klein Chap. 8 Preparation of Adipic Acid OP 15, 16, Klein Chap. 9 Preparation of 9-Fluorenol Wade, Chap. 13 Grignard Reaction Wade, Chap. 13 Grignard (continue) yes yes yes yes yes yes yes yes yes yes no Laboratory Clean-up and Check-Out Comprehensive Written Final Exam 3 Laboratory Polices and Information: Lab Reports: Lab reports will be STAPLED and turned in to the instructor. Prelab reports are due at the start of the lab. Once a lab is complete you will turn in a post lab and take a quiz over that lab the following lab period. Quizzes: At the end of each tutorial there will be a quiz over the material from the week before. The quizzes will cover the experimental techniques used in lab, the procedure, and the reactions from the lab. Laboratory Safety: Safety Goggles Must Be Worn in the lab at all times. The rules in lab will be discussed in the first class period. Laboratory Notebooks and Reports: Laboratory notebooks contain a top original copy and a carbon copy. Always tear out the bottom carbon copy to turn in. All entries in your notebook should be in ink and legible. Your notebook should be detailed enough for another person to follow. Always include name, date, lab number, etc. on the top of every page. Each new experiment should begin with a new page. In the lab notebook you should record actual measurements. Include calculations, yields, and physical observations for your end products. 4 Write-up Procedures Pre-Lab consists of sections 1-4. Post Lab consists of sections 5-8. Pre labs are to be turned in prior to the start of the experiment. Post labs are due the following lab period after experiment is complete. For the Pre Lab write-up (Always include name, date, lab number, etc. on the top of every page): 1. Main Idea. Write a brief sentence or two describing what you expected to learn or to accomplish along with a reaction (If applicable). 2. Reaction(s). Include all possible reactions that might occur in the lab for that week. If there is no reaction then write there are no reactions for this lab. 3. Table of physical constants. Include a Table of Physicals Constants which should include name of chemicals used or synthesized, MW, mp/bp, density, structure, and any special hazard/cautionary notes. 4. Prelab questions Two web sites that will assist in chemical property data collection are listed below. http://www.chemexper.com/ http://chemfinder.cambridgesoft.com/ Below is a web site for structures which can be used to obtain spectroscopic data http://www.aist.go.jp/RIODB/SDBS/cgi-bin/direct_frame_top.cgi?lang=eng For the Post lab write-up (Always include name, date, lab number, etc. on the top of every page): 5. Data and observations. This is the procedure section. As you perform your lab write down in your lab book the methods you are doing and data you are obtaining. For any melting or boiling point that you measure, the literature value should also be reported. The spectra obtained should be labeled and stapled (IR, NMR, UV) to the back of your report. 6. Calculations. This section must include all calculations. Be sure to label all calculations clearly. 7. Conclusions. This is especially important for investigative type experiments. Discuss results obtained with respect to anticipated findings, any errors that may have caused your results to deviate from what was expected, and any side reactions that may have decreased yields. Summarize what you learned. Discuss your data!!!!!!!! 8. Post lab Questions. Answer the post lab questions from the experiment. 5 EQUIPMENT FOR THE ORGANIC LABORATORY DRAWER Contents of Drawer Contents of MicroWare Box ITEM DESCRIPTION Beaker 50 mL COST $1.75 Beaker 100 mL $1.80 Adaptor, Claisen, $21.74 Beaker 150 mL $1.65 Adaptor, thermometer $13.68 Beaker 250 mL $1.75 Bulbs, amber (3) $.72/ ea Brush, test tube, micro $1.95 Condenser, water jacketed $27.05 Brush, test tube, small $1.95 Conical vials, 3 mL (2) $9.54 /ea Centrifuge tube, glass w/lid $0.75 Conical vials, 5mL(2) $9.54 /ea Clamp 3 prong with holder $62.90 Drying tube, $10.75 Erlenmeyer flasks: 25(2) mL $2.00/ea Erlenmeyer flasks, 10 mL (2) $8.74/ea Erlenmeyer flasks: 50 (2) mL $2.00/ea Hickman still, with sidearm $26.33 ITEM DESCRIPTION COST Erlenmeyer flasks: 125 mL $2.10 Hirsh funnel, with stopper $30.19 Filter flask, 25mL $12.16 Round bottom flask, 10 mL $10.46 Filter flasks, 125 mL $16.00 Spin vane, Teflon $9.15 Funnel, glass stem less $3.70 Spatula, double ended $14.21 Funnels, plastic $0.30 Spatula, micro $8.45 Graduated cylinder, 10 mL $8.10 Stirring rod $.50 Graduated cylinder, 25 mL $8.10 Storage box $15.12 Test tubes: 13 X 100 mm (6) $.20/ea Syringe, glass $18.30 Test tubes: 18 X 150 mm (6) $.35/ea Thermometer $24.87 Test tube holder $9.10 Tongs, crucible $9.81 Thermometer , digital $37.5 Watch glasses: (2) $ 1.50/ea Revised 05/10 6 Laboratory Safety 1. Wear approved eye protection in the laboratory continuously. This means eye covering which will protect both against impact and splashes. (If you should get a chemical in your eye, wash with flowing water from a sink or fountain for 15-30 min.) 2. Perform no unauthorized experiments. 3. In case of fire or accident, call instructor at once. (Note location of fire extinguisher and safety shower now so that you can use them if needed. Wet towels are very efficient for smothering fires.) 4. You must go to the infirmary for treatment of cuts, burns, or inhalation of fumes. (Your instructor will arrange for transportation if needed.) 5. Do not taste anything in the laboratory. (This applies to food as well as chemicals. Do not use the laboratory as an eating place and do not eat or drink from laboratory glassware.) 6. Exercise great care in noting the odor of fumes and avoid breathing fumes of any kind. 7. Do not use mouth suction in filling pipettes with chemical reagents. (Use a suction bulb) 8. Do not force glass tubing into rubber stoppers. (Protect your hands with a towel when inserting tubing into stoppers.) 9. Confine long hair when in the laboratory. (Also, a laboratory apron is essential when you are wearing easily combustible clothing. Such an apron affords desirable protection on all occasions.) 10. No open-toed shoes will be allowed in the laboratory. 11. Never work in the laboratory alone. 12. No smoking or other use of tobacco products in the laboratory. 13. Failure to adhere to rules will result in immediate dismissal from laboratory. I have read the above rules. I have participated in a safety orientation, and I will observe all of the safety rules of my chemistry course. _____________________________________ Name (printed) ________________________ Date _____________________________________ 313 ____________ Signature Room # Drawer # 7 Equipment for the Organic Laboratory Name (print)____________________________________ Day ________ Contents of Drawer Item Description Beakers: 50, 100, 150, 250 mL Brushes, test tube, micro and small Centrifuge tube, glass w/lid Clamp 3 prong with holder Erlenmeyer flasks: 25(2), 50 (2), 125 mL Filter flasks: 25, 125 mL Funnels: glass stem less, plastic Graduated cylinders: 10, 25 mL Test tubes: 13 X 100 mm (6), 18 X 150 mm (6) Test tube holder Tongs, crucible Thermometer , digital Watch glasses: (2) Check in Check out Cost Check in Check out Cost Contents of Microware Kit Item Description Adaptor (2): Claisen, thermometer Bulbs, amber (3) Caps, Green (7) Condenser, water jacketed Conical vials: 3 mL (2), 5mL(2) Drying tube, Erlenmeyer flasks, 10 mL (2) Hickman still, with side arm cap and septum Hirsh funnel, with stopper O-rings (5) Round bottom flask, 10 mL Septa, Teflon coated (5) Spin vane, Teflon Spatulae: double ended, micro Stirring rod Syringe, glass Thermometer Total cost for lost or broken items NC NC NC $ (Complete bottom portion when checking out of lab) Student Signature _________________________ TA Signature_________________ Mustang ID No. __________________________ Date _______________________ 8