2. Method of Calculations 3.
advertisement
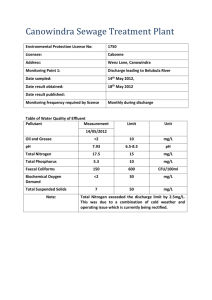
1. Introduction Many mathematical models have been developed for Li- battery cell performance. However, a critical need still do exists for a rather simple battery performance evaluation method. In the present study, such a parameter, Integral Average Voltage (IAV), is being introduced. Its utility, convenience and applicability are validated via existing experimental data derived from lithiated Ni dope Mn spinel 5V cathode materials. IAV is simple to calculate using the cell charge/discharge profile; at the same time, it offers a perceptive analysis of cell features and associated processes. 2. Method of Calculations 3. The Applicability of the Proposed Concept Case Study #1 The energy spent (or gained) in course of charge transfer through the potential difference Vcell(Qfinal)-Vcell(Qinitial) is Single- and double- doped spinels (M = Ni, Co, Cu, Cr, Fe, etc.) have been reported to have operational voltages over 4V and improved cycling performance, compared to pure LiMn2 O4 . The V(Q) curves for these oxides usually have a complicated shape and are inconvenient for comparison and further consideration (figure 1). Figure 2 better assist identifying several correlations between different variables and thus, allow a better comprehensive analysis of the studied electrode system. Q final E V (Q)dQ (1) Qinitial 𝑄𝑖𝑛𝑖𝑡𝑖𝑎𝑙 (𝑄𝑓𝑖𝑛𝑎𝑙 ) stands for the charge of the cell at the beginning (end) of the discharge/ charge process. 5.0 The average integral voltage, over the [𝑄𝑖𝑛𝑖𝑡𝑖𝑎𝑙 , 𝑄𝑓𝑖𝑛𝑎𝑙 ] interval is defined as: Q final 4.6 <Voltage> (V) Vcell 4.8 V (Q)dQ Qinitial Q final Qinitial E Q (2) 3.8 <V(discharge)>; 1st run <V(charge)>; 1st run <V(discharge)>; 50 th run <V(charge)>; 50 th run Li partial intercalation energy 3.6 3.4 0.00 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0.50 0.55 x (Ni content) (3) LiNi0.4Mn1.6O4–cathodes containing different content of conductive additives. 5.0 13% Super P carbon 4% Super P carbon 4.8 <V(discharge)> (V) 4.0 3.0 4. The Applicability of the Proposed Concept Case Study #2 3. 4.2 3.2 The average over-voltage over this interval may be defined as: emf Vcell Vcell Vcell 4.4 4.6 4.4 4.2 tg=0.13 4.0 3.8 Figure 2: 𝑉𝑐 (𝑥 ) (charge/discharge) for Figure 1: Charge/discharge behaviour of LiMn2−xNixO4 cycled in LiPF6 + (EC+DMC)/Li LiMn2−xNixO4/LiPF6 + (EC+DMC)/Li coin cell. 𝑉𝑐 (𝑥 ) (charge/discharge) calculated using coin cell data from figure 1 and equation (2). Li partial intercalation energies resulted from ab-initio quantum-mechanical calculations are presented. Figure 2 reveals that 𝑉𝑐 (𝑥) dependence on Ni-content (x) correlates well with the dependence of Li partial intercalation energies on nickel content. Li+-conductivity in LiMn2−𝑥 Ni𝑥 O4 improves with increase in nickel content and thus, the ohmic component is expected to diminish in course of cycling. In course of cycling 𝑉𝑐 (charge) is practically unchanged, whereas 𝑉𝑐 (discharge) increases for x>0. tg=0.32 3.6 3.4 3.2 3.0 0.0 0.5 1.0 1.5 2.0 2.5 3.0 5. Conclusions Current (A/g) Figure 3: Galvanostatic discharge voltage curves of LiNi0.40Mn1.6O4 sample containing 4 wt % (dashed) and 13 wt % (solid) Super P carbon as a conductive additive Figure 4: 𝑉𝑐 (discharge) for LiNi0.4 Mn1.6 O4 cathode materials for different discharge currents and different conductive additive contents. 𝑉𝑐 (𝑥 ) (discharge) calculated using data from figure 3 and equation (2). Figure 4 clearly demonstrates that the ohmic behaviour prevails for currents over 0.022 A/g for all conductive carbon contents. The conductivity of the cathode containing 13% of carbon is 2.5 higher than the conductivity of the cathode utilizing only 4% of carbon. The integral average voltage (IAV) method is described and being evaluated. IAV provides a convenient and robust approach presenting results of advanced Li metal cell (Li-air) and 5 V Li-ion positive electrode (lithiated Ni doped Mn spine) testing. IAV allows a deeper understanding of electrode reactions and electrode structure. The method, if being adopted will be highly applicable and seems quite useful in battery-related research. Acknowledgments This work was supported by The Nancy and Stephan Grand Technion Energy Program (GTEP), the Israeli Ministry of Energy and Water Resources, Israel Science Foundation (ISF), Israel National Research for Electric Propulsion (INREP), and the Helmsley Charity Fund. References [1] M. Kunduraci, J.F. Al-Sharab, and G. G. Amatucci, Chem. Mater., 2006, 18, 3585–3592; [2] R.Singhal, J. Saavedra-Aries J., R.Katiyar, Y.Ishikawa, M.J. Vilkas, S.R. Das, M.S. Tomar R.S. Katiyar, J. Renewable Sustainable Energy, 2009, 1 , 023102/1 – 023102/11; [3] Copyright © Mr.WESIK Version v2.10 24/10/2001, acquired on 10-2011 from http://www.4shared.com/get/6SeK55iD/grafula3_curve_tracking.html