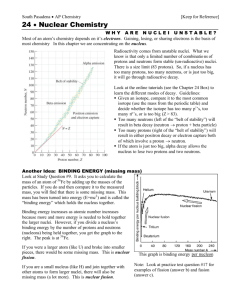
Name
CHAPTER 10
Class
Date
Nuclear Changes
SECTION
21 Nuclear Fission and Fusion
KEY IDEAS
As you read this section, keep these questions in mind:
• What holds the nucleus of an atom together?
• What happens when the nucleus of a heavy atom
splits apart?
• What happens when the nucleus of a small atom joins
with the nucleus of another small atom?
Who Discovered Nuclear Fission?
In 1939, German scientists Otto Hahn and Fritz
Strassman were trying make uranium atoms with heavier
nuclei. To do this, they used a device that shot neutrons
at a sample of uranium. They hoped that at least a few
uranium nuclei would gain one or more neutrons. To
their surprise, new elements formed. Instead of uranium isotopes, they detected barium and krypton. They
thought they had made a mistake.
Soon after, a scientist named Lise Meitner and her
nephew Otto Frisch read the results of the experiments.
Meitner realized that Hahn and Strassman had not made
a mistake. They had actually split the uranium nuclei into
smaller elements.
READING TOOLBOX
Define As you read, write
down any terms in the section that you do not understand. Use the section vocabulary boxes or a dictionary to
find the definitions of these
terms. Write the definitions
in your own words in the text
margin.
READING CHECK
1. Explain What did Lise
Meitner conclude about the
work of Hahn and Strassman?
This nuclear reactor
was used in the late
1940s and early 1950s
to study controlled
nuclear fission.
In the early 1940s, Enrico Fermi and other scientists
at the University of Chicago built a stack of graphite and
uranium blocks. These materials formed the nuclear reactor that was used to create the first controlled nuclear
fission chain reaction. This work launched the Manhattan
Project, which led to the creation of nuclear weapons.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
213
Nuclear Changes
Name
SECTION 2
Class
Date
Nuclear Fission and Fusion continued
What Holds a Nucleus Together?
Protons and neutrons are packed tightly in the tiny
nucleus of an atom. Recall that some atomic nuclei are
unstable and emit nuclear radiation as they decay. The
stability of a nucleus depends on the nuclear forces
that hold the nucleus together. If like charges repel one
another, how can positively charged protons fit together
into an atomic nucleus without flying apart?
THE STRONG NUCLEAR FORCE
READING CHECK
The strong nuclear force holds the neutrons and
protons together in a nucleus. This attraction is much
stronger than the electric force that causes protons
to repel other protons. However, the attraction only
happens over a very short distance (3 × 10–15 meters).
This is less than the width of three protons.
2. Compare Compare the
relative strengths of the
nuclear force and the electric
force.
4USPOHOVDMFBSGPSDF
BDUTPOQSPUPOTBOEOFVUSPOT
&MFDUSJDSFQVMTJPO
BDUTPOQSPUPOT
The strong nuclear force that holds protons and neutrons together is stronger than
the electrical force that pushes protons apart.
THE STRONG NUCLEAR FORCE AND STABILITY
READING CHECK
3. Describe What are
the relative strengths of
attractive and repulsive
forces in a stable nucleus?
Because neutrons have no charge, they do not attract
or repel protons or one another. However, protons repel
one another due to the electric force and attract one
another due to the nuclear force. In stable nuclei, the
attractive forces are stronger than the repulsive forces.
Under these conditions, the element does not undergo
decay.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
214
Nuclear Changes
Name
Class
SECTION 2
Date
Nuclear Fission and Fusion continued
What Causes a Nucleus to Decay?
An element decays when the repulsive forces in the
nucleus are stronger than the attractive forces. A large
number of neutrons in an atom can help hold a nucleus
together. However, there is a limit to how many neutrons
a nucleus can have. Nuclei with too many or too few neutrons are unstable and undergo decay.
UNSTABLE NUCLEI
Nuclei with more than 83 protons are always unstable,
no matter how many neutrons they have. These nuclei
always decay and release large amounts of energy and
nuclear radiation. Some of the energy is passed to the
particles that leave the nucleus. The rest of the energy
leaves the nucleus in the form of gamma rays. This radioactive decay helps produce a more stable nucleus.
What Is Nuclear Fission?
The process of splitting atoms with heavy nuclei into
atoms with lighter nuclei is called fission. When the
nucleus splits, both neutrons and energy are released.
In their experiments, Hahn and Strassman used a
device that shot at, or bombarded, a uranium-235 nucleus
with neutrons. One set of products from this type of
fission includes two lighter nuclei, barium-140 and
krypton-93, and neutrons and energy.
235
92
93
1
U + 10 n 140
56 Ba + 36 Kr + 3 0 n + energy
Notice that the products include three neutrons
plus energy. Uranium-235 can also undergo fission to
produce different pairs of lighter nuclei. For example,
uranium-235 can undergo fission to produce strontium-90,
xenon-143, and three neutrons.
How Does Nuclear Fission Produce Energy?
During fission, the nucleus breaks into smaller nuclei.
The process releases large amounts of energy. Each dividing nucleus releases about 3.2 × 10–11 joules of energy. In
comparison, the chemical reaction of one molecule of the
explosive trinitrotoluene (TNT) releases 4.8 × 10–18 joules.
READING CHECK
4. Identify Under what
conditions does a nucleus
decay?
READING CHECK
5. Identify What is the
maximum number of protons
that can be found in a stable
nucleus?
EHHDBG@<EHL>K
6. Analyze How many total
neutrons are on the left side
of the equation? How many
total neutrons are found on
the right?
Math Skills
7. Compare How much
more energy is released by
a dividing nucleus than by a
molecule of TNT?
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
215
Nuclear Changes
Name
SECTION 2
Class
Date
Nuclear Fission and Fusion continued
When a uranium-235 nucleus is bombarded by a neutron, the nucleus breaks apart
into smaller nuclei. The process releases energy through fast moving neutrons.
READING CHECK
8. Explain Why is some
mass missing after fission?
READING CHECK
In their experiment, Hahn and Strassman recorded the
masses of all of the nuclei and particles before and after
the reaction. They found the overall mass had decreased
after the reaction. Hahn and Strassman also found that
the process had released energy. They concluded that the
missing mass must have changed into energy.
What Is Mass-Energy Equivalence?
Recall that, according to Newton’s laws, no mass or
energy can be created or destroyed during physical or
chemical changes. The laws of conservation of mass and
energy do not apply to nuclear reactions such as fission.
During fission, some matter changes to energy.
Albert Einstein explained the equivalence of mass and
energy by the special theory of relativity. This equivalence
means that matter can be converted into energy, and
energy can be converted into matter. Equivalence is
described by the equation below.
9. Explain What does massenergy equivalence mean?
Mass-Energy Equation
Energy = mass × (speed of light)2
E = mc2
The speed of light, c, equals 3.0 × 108 m/s. If you multiply c 2 by even a very small mass, the energy value is very
large. For example, the mass-equivalent energy of 1 kg of
matter is 9 × 1016 joules. That is more than the energy of
22 million tons of the explosive TNT.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
216
Nuclear Changes
Name
SECTION 2
Class
Date
Nuclear Fission and Fusion continued
STABILITY OF MATTER
Obviously, the objects around us do not change suddenly into their equivalent energies. The results of that
would be disastrous. Under ordinary conditions, matter is
very stable.
What Is the Mass Defect?
Suppose you measured the mass of a carbon-14
nucleus, which has six protons and eight neutrons.
Suppose you then measured the mass of six individual
protons and eight individual neutrons. You would expect
the mass of the nucleus to be the same as the total mass
of the individual particles. However, you find that the
mass of the nucleus is less than the sum of the individual
masses. What happened to the missing mass?
Mass of 1 proton
= 1.673 × 10−27 kg
Mass of 6 protons
=
Mass of 1 neutron
= 1.675 × 10−27 kg
Mass of 8 neutrons
=
Actual mass of a carbon-14 nucleus
= 2.325 × 10−26 kg
Expected mass of a carbon-14 nucleus
=
The missing mass is called the mass defect. Einstein’s
theory of special relativity explains: the missing mass
changes into energy. When nuclei form, energy is
released. Note, however, that the mass defect of a
nucleus is very small.
What Is a Nuclear Chain Reaction?
Math Skills
10. Calculate Complete the
table to compare the expected and actual values for the
mass of a carbon-14 nucleus.
READING CHECK
11. Define What is the mass
defect?
Have you ever watched balls moving on a pool table?
When one ball hits another, the collision can cause the
second ball to hit another. Some nuclear reactions work
the same way. One reaction triggers another reaction.
A nucleus that splits when it is hit by a neutron forms
smaller nuclei. The smaller nuclei need fewer neutrons
to be held together. Therefore, they release extra neutrons. If one of those neutrons collides with another
large nucleus, that nucleus undergoes fission, or splits. A
nuclear chain reaction is a continuous series of nuclear
fission reactions.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
217
Nuclear Changes
Name
SECTION 2
Class
Date
Nuclear Fission and Fusion continued
1
n
0
93
Kr
36
93
Kr
36
1
n
0
1
n
0
1
n
0
140
Ba
56
EHHDBG@<EHL>K
1
n
0
140
56 Ba
1
n
0
140
Ba
56
1
n
0
1
n
0
235
U
92
235
U
92
235
U
92
1
n
0
235
U
1
n 92
0
93
Kr
36
1
n
0
235
U
92
12. Predict How many total
neutrons will probably be
released in the next stage of
this chain reaction?
1
n
0
93
36 Kr
235
U
92
235
U
92
235
U
92
1
n
0
140
Ba
56
235
U
92
235
U
92
A nuclear chain reaction may be triggered, or started, by a single neutron.
Scientists found that on average, each uranium
nucleus that divides produces two or three extra neutrons. Each one of these neutrons could trigger, or start,
another fission reaction. The ability to start a nuclear
chain reaction depends partly on the number of neutrons
released during each fission reaction.
What Is a Controlled Chain Reaction?
Energy produced in a nuclear chain reaction can be
used to generate electricity. The diagram describes this
process.
Using Nuclear Chain Reactions to Generate Electricity
1
2
Uranium-235
nuclei in the
fuel rod (black)
undergo a chain
reaction. Control
rods (gray)
absorb neutrons.
This keeps the
chain reaction at
a safe level.
A coolant, usually
water, absorbs energy
from the chain reaction.
3
Water absorbs
energy from the hot
coolant and changes
to steam.
EHHDBG@<EHL>K
13. Identify What is the
function of the control rods?
To cooling
tower
5
4
The steam turns a turbine
attached to a generator.
The generator changes the
mechanical energy of the spinning
turbine into electrical energy.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
218
Nuclear Changes
Name
Class
SECTION 2
Date
Nuclear Fission and Fusion continued
NUCLEAR WEAPONS
The chain-reaction principle is also used to make a
nuclear bomb. In a nuclear bomb, two or more quantities
of uranium-235 are packed into a container. The uranium
is surrounded by a powerful chemical explosive. When
the explosives are detonated, or set off, the uranium is
pushed together to exceed the critical mass. The critical
mass is the smallest amount of a substance that provides
enough neutrons to start a nuclear chain reaction.
If the amount of a substance is less than the critical
mass, a chain reaction will not continue. Fortunately, the
concentration of uranium-235 in nature is too low to start
a chain reaction naturally. In nuclear power plants, control rods are used to slow the chain reaction. In a nuclear
bomb, reactions are not controlled.
READING CHECK
14. Define What is a
critical mass?
What Is Nuclear Fusion?
Nuclear fission is not the only nuclear process that can
produce energy. Energy can also be produced when light
atomic nuclei join, or fuse, to form heavier nuclei. This
process is called fusion.
In the sun and other stars, huge amounts of energy are
produced when hydrogen nuclei fuse. However, a large
amount of energy is needed to start a fusion reaction.
Energy is needed to push nuclei close enough so that the
strong nuclear force can overcome the repulsive electrical force. In stars, extremely high temperatures provide
the energy to bring hydrogen nuclei together.
1
H
1
READING CHECK
15. Describe What happens
during nuclear fusion?
+ 11H → 21 H + other particles
2
H + 11H → 32He + 00γ
1
3FBDUJPO
3
He
2
+ 32He → 42He + 11H + 11H
3FBDUJPO
3FBDUJPO
3FBDUJPO
3FBDUJPO
The process of nuclear fusion releases large amounts of energy.
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
219
Nuclear Changes
Name
Class
Date
Section 2 Review
SECTION VOCABULARY
critical mass the minimum mass of a
fissionable isotope that provides the number
of neutrons needed to sustain a chain reaction
fission the process by which a nucleus splits
into two or more fragments and releases
neutrons and energy
fusion the process in which light nuclei
combine at extremely high temperatures,
forming heavier nuclei and releasing energy
nuclear chain reaction a continuous series of
nuclear fission reactions
1. Summarize Complete the process chart to describe how nuclear fission is used to
produce electricity.
A coolant absorbs energy from the chain reaction.
A generator changes the mechanical energy of the spinning turbine into electrical energy.
2. Predict Suppose you had an atom of
56
26
Fe. Is the mass of its nucleus greater
than, less than, or equal to the combined masses of 26 protons and 30 neutrons?
Explain your answer.
3. Identify Do the following equations describe nuclear fission or nuclear fusion?
Explain your answers.
235
1
140
93
1
92 U + 0 n 56 Ba + 36 Kr + 3 0 n + energy
208
82
Pb +
58
26
1
Fe 265
108 Hs + 0 n
Copyright © by Holt, Rinehart and Winston. All rights reserved.
Interactive Reader
220
Nuclear Changes
The Great Radiation Journey
Today you are going to go on a journey to learn all about radiation and how it
affects you. Here are the rules to follow:
1. Only one group per station.
2. Be quiet in the halls!
3. Leave other classrooms alone.
Station #1
Location:_____
1. What percent of your annual radiation dose is from background radiation?
2. What are some sources of background radiation?
3. Where do you find background radiation?
Station #2
Location:_____
4. What percent of your annual radiation dose is from human-made radiation?
5. What are some sources of human-made radiation?
6. Is there any difference between background radiation and human-made
radiation?
Station #3
Location:_____
7. What is stored in the nucleus of an atom?
8. How do scientists release the energy in an atom?
9. What two things are released when you split an atom?
10. What are the three main types of radiation produced by atoms?
Station #4
11. What are alpha particles?
12. How far can alpha particles travel?
13. What can stop alpha particles?
14. When are alpha particles harmful?
Location:_____
Station #5
Location:_____
15. What are beta particles?
16. How far can beta particles travel?
17. What can stop beta particles?
18. When are beta particles the most harmful?
Station #6
Location:_____
19. What are gamma rays?
20. How far can gamma rays travel?
21. What can stop gamma rays?
Station #7
Location:_____
22. What unit do we use to measure radiation?
23. What is the average person’s dose of radiation each year?
24. How much of the yearly dose is due to background radiation?
25. How much of the yearly dose is due to human-made radiation?
Station #8
Location:_____
26. How much background radiation does a person living on the Atlantic Coast
receive each year?
27. How much background radiation does a person living on the Colorado Plateau
receive?
28. Why does the person on the Colorado Plateau receive more background
radiation?
Station #9
Location:_____
29. Why are you exposed to more background radiation when you fly?
30. How much radiation exposure do you get when you…..
fly? __________,
have dental x-rays? ________
work at a nuclear power plant? ____________
For Stations 10-17, you are going to calculate your yearly radiation dose.
31. Enter your values on the table below:
Station #
10
11
12
13
14
15
16
16
16
16
17
17
17
Station #18
Station
Location
Radiation Exposure
(mrems)
Item
Elevation
Location
House Materials
Food and Water
Air (Radon)
Flying
Cell Phone
MP3 Player or I-Pod
Computer Use
Watching Television
Smoke Detectors
Nuclear reactors
Medical X-rays
Total Yearly Exposure:
Location:_____
32. What is the average annual radiation dose for Americans?
33. What is the Government limit for workers in the Nuclear Industry?
34. Should you be worried about your exposure? Why or why not?
Station #19
Location:_____
35. Fill in the table below:
Radiation Dose
Effect
Time to Onset
50,000 mrems
75,000 mrems
400,000 mrems
1,000,000 mrems
2,000,000 mrems
Station #20
Location:_____
36. Describe what happened at Three Mile Island.
37. Describe what happened at the Chernobyl Power Plant?
Radioactive Decay
Alright, we are going to continue to learn about the nucleus of the atom.
Here’s what we know so far:
- Protons and Neutrons are in the nucleus.
- Protons have a positive charge and neutrons have no charge.
- Protons repel each other when they get too close.
- The neutrons in the nucleus keep the protons from getting too close to
each other.
- Atoms with too many, or too few, neutrons are unstable.
- The rounded masses on the Periodic Table are the masses of stable
isotopes.
Here’s what we still need to learn:
- What happens to unstable atoms?
- What are the different types of decay?
- How do I write out a decay reaction?
- When do atoms stop decaying?
- What is fission?
- What is Fusion?
- What is meant by “half-life”?
- How does radiation affect us?
Well, that is quite a bit to learn, so we better get started right away. To begin
with we need to practice using our Periodic Tables to determine if an isotope is
stable or unstable.
Directions: Fill in the following table for the elements listed. Make sure to double
check the masses given with the symbol to see if the atom is stable or not.
Symbol
21
10
S
36
17
Cl
40
19
Atomic
Number
Number
Number
from
of
P.T.
Isotope
Number
Number
of
of
Protons
Neutrons
Stable or
Unstable?
K
Na
131
___
238
92
Mass
Ne
32
16
23
11
Name
Mass
Xe
___
Radium226
94
239
Iodine131
11
5
72
Carbon___
102
Stable
Now that you are an expert at finding out if an atom is stable or unstable, you are
ready to learn about what happens to unstable atoms. Quite simply, unstable
atoms break down. Think of unstable atoms as being fragile or weak. They aren’t
strong enough to stay in one piece, so they fall apart. Now they don’t do it all
crazy and chaotic – they actually break down in certain, defined ways until
they become stable. That’s right, unstable atoms break down (or decay) in order
to turn into stable atoms. Along the way they can change from one kind of element
to another. Let me show you what I mean by starting out with the three types of
decay that we need to learn about: alpha, beta and gamma.
Important Note:
When we want to write down what isotope of an element we are talking about here
is how we do it:
atomic mass
C-14
atomic number
The first way shows the atomic number and the mass, the second way just shows
the symbol and the mass.
Alpha (
)Decay:
Uranium-238 is an unstable isotope of Uranium. It has way too many neutrons to
be stable. In the process of becoming stable, parts of the nucleus break off and
shoot off from the nucleus. In alpha decay, an alpha particle is released from the
nucleus. Here is the equation showing U-238 undergoing alpha decay:
Alpha Particle
When an unstable atom undergoes alpha decay, an alpha particle breaks away from
the nucleus and what is left is a new element. In this case, the Uranium atom loses
two protons and it turns into a Thorium atom. The alpha particle is made up of two
protons and two neutrons. It is the exact same like a Helium nucleus.
Alpha Decay Practice
Fill in the missing information:
Beta (β) Decay
Carbon-14 is an unstable isotope of the element Carbon. Instead of having 6
protons and 6 neutrons like the stable isotope of Carbon, Carbon-14 has 6 protons
and 8 neutrons. Since Carbon-14 (we also can write it like this: C-14) is unstable,
it breaks down. In this case it undergoes something that we call beta decay.
Atoms that undergo beta decay have too many neutrons. Beta decay takes care of
that problem. Here is the equation showing C-14 undergoing beta decay:
Beta Particle
You will notice that the mass of the atom before and after decay is 14, but the
atomic number has gone up (from 6 to 7). The beta particle is an electron (that is
why its mass is 0 and its atomic number is -1). If you are really observant, you may
have noticed that the number of neutrons went down (from 8 to 7). It ends up
that a neutron is really a proton and an electron combined together. In beta
decay, a neutron breaks down into a proton and an electron. The proton is held
in the nucleus while the electrons escapes.
Beta Decay Practice
Fill in the missing information:
Gamma (
) Decay
Coblat-60 is an unstable isotope of Cobalt. But, unlike in the atoms in the alpha
and beta decay examples, Cobalt doesn’t have too many or too few neutrons, it has
too much energy. During gamma decay, atoms release electromagnetic radiation
(photons). The mass of the atom doesn’t change and you don’t get a new element.
Gamma Decay Practice
Do we really need to practice it if nothing changes?
Practicing Decay Series
Part One: Alpha Decay Problems
Do alpha decays until you reach a stable element.
73
1.
Se
34
48
2.
Ti
22
82
3.
Rb
37
31
4.
P
15
200
5.
Tl
81
Part Two: Beta Decay
Do beta decays until you reach a stable element.
52
6.
V
23
65
7.
Ni
28
51
8.
K
19
32
9.
Al
13
91
10.
Rb
37
Part Three: Find the steps needed to reach the end product.
Start
End
59
11.
55
Fe
Mn
26
25
97
89
12.
Mo
Y
42
181
13.
39
169
Hf
72
Tm
69
Part Four: Super Decay Challenge: These decays involve many steps. Good luck!
Start
End
238
14.
15.
U
Pb
92
82
235
207
U
92
Pb
82
236
16.
206
208
Th
90
Pb
82
Isotopes, the Strong Nuclear Force and the Band
of Stability
What Makes an Atom Stable or Unstable?
The nucleus of an atom is a strange thing. We know that it is made up of protons and neutrons.
We know that neutrons have no charge and that protons have a positive charge. We also know
that charges that are the same (i.e. protons all have a positive charge) repel each other. The
strange part is that the protons in the nucleus repel each other and yet, the nucleus stays
together.
What Makes the Nucleus Stay Together?
Well, scientists figured out that if there was a force of repulsion between protons, there must
be a stronger force holding the nucleus together. Being creative thinkers, they called this force
the Strong Nuclear Force. It ends up that the strong nuclear force only works over small
distances (I am talking like 10-12 meters, here. I mean really short distances). As long as the
protons in a nucleus are really close together, the Strong Nuclear Force overcomes the force of
Repulsion and the nucleus is fine.
Why Are Some Atoms Unstable?
Well, the key is that there are protons and neutrons in the nucleus. The neutrons mix in
between the protons and push them further apart. In fact, the scientists have found out
that the protons and neutrons actually fill in energy levels – just like electrons do and that
some arrangements of protons and neutrons are more stable than others. If an atom gets
too many neutrons in its nucleus, the protons get pushed too far apart and the Strong Nuclear
Force can’t overcome the force of repulsion between the protons. The end result is that parts
of the nucleus break off and leave.
The Band of Stability
Today you are going to look at something called “The Band of Stability”. Basically it just has to
do with the notion that a certain number of protons and neutrons in a nucleus make it stable.
Let’s get started:
1. Fill in the missing numbers on the Table of Stable Isotopes below:
Isotope
Stable Isotopes
Mass
Protons
Neutrons
He–4
C-12
Si-28
Sc-45
Fe-56
Ag-109
Xe-131
Gd-160
W-184
Pb-206
N-15
O-16
Ca-46
Rb-85
Mo-95
2. Make a graph of the number of neutrons (y-axis) vs. the number of protons (x-axis)
3. The black line already drawn on your graph represents the number of neutrons and the
number of protons being equal (this is just the y=x graph).
4. Draw in the best fit curve for the data that you just plotted.
5. Fill in the missing numbers on the Unstable Isotopes table below:
Isotope
Unstable Isotopes
Mass
Protons
Neutrons
C-14
Si-32
Fe-52
Xe-155
Pb-214
Ra-226
6. Plot these points on your graph. Make sure to label these points with the isotope name.
0
10 20 30 40 50 60 70 80 90 100
Questions
7. Would a small atom (less than 40 protons) be found in nature if it had the same number
of protons and neutrons?
Explain your answer.
8. Would a large atom (more than 40 protons) be found in nature if it had the same number
of protons and neutrons?
Explain your answer.
9. Imagine that a chemist was trying to create an atom with 60 protons and a mass number
of 155. Would this be possible? Why or why not? (SHOW where if would fall on your
graph).
10. If an element had 90 protons, how many neutrons would be a good number for it to have
in order for it to be considered a stable element? What element would this be? (SHOW
where if would fall on your graph).
11. Atoms will decay until their nuclei are in the Band of Stability. The type of decay will be
determined by what they need to do to get to the Band of Stability. For each of the
unstable isotopes that you graphed, write out a decay reaction that will get them closer
to the Band of Stability.
Isotope Decay Reaction (make sure to label it alpha or beta)
C-14
Si-32
Fe-52
Xe-155
Pb-214
Ra-226
12. What changes take place in the nucleus when an alpha particle is emitted?
13. What is the identity of an alpha particle?
14. What changes take place in the nucleus when a beta particle is emitted?
15. What is the identity of a beta particle?
Name:
______________
Half-Life and M&Ms
Fact: Scientists have calculated the age of the Earth to be 4.6 billion years old.
Question: How do we know that when nobody was around at that time?
Scientists use things that were around when the Earth formed - rocks - to find
estimates for the age of the Earth. It all hinges on something called radioactive
decay and half-life.
Here’s how it works: All matter is made up of atoms and in some cases the
atoms are unstable.
Unstable atoms have nuclei that have too many, or too few,
neutrons. When this happens, the nuclei break down. When they break down,
they shoot off particles or energy - this is the radiation. The thing about all of
this that helps with dating the Earth is that radioactive nuclei decay at a steady,
measurable rate. This rate is unique for each kind of radioactive substance and it
has a special name: half-life.
The half-life of a radioactive substance is the time it takes for half of
a radioactive substance’s atoms to decay into stable elements.
Today, you are going to learn about how radioactive decay and half-life work. Have
fun and bon appetit!
Instructions
Each person needs 60 M&Ms for this activity. Count out 60 from your cup and
SAVE them for this activity. You may eat any extras that you have. Make sure
there is writing on one side of each of your M&Ms.
Do not eat your 60 M&Ms until you finish question #6!
Part One: The Mathematics of Decay
1. How many sides does an M&M have?
2. How many sides have an “M” on them?
3. If you shook an M&M and dropped it, what are the odds that the “M” side
would be up?
4. If you had 100 M&M’s and dropped them, how many “M” s would you expect
to have facing up?
Part Two: Graphing Radioactive Decay and Half-life
You will be in groups of 4 for this activity, but you will handle only your own M&Ms.
Put some paper down on the table before you do the next step!
In your groups, please do the following:
a) Assign each team member a number (1, 2, 3 or 4) on the data table below.
b) For the starting point (turn zero) record the total number of M&Ms.
c) Take your M&Ms and shake them up. Spill them out on your paper towel and
count the number of M&Ms that have the “M” side up. Note: For this lab we
will have radioactive nuclei (un-decayed nuclei) be represented by M&Ms
with the “M” side up and decayed nuclei be the ones with no “M” showing.
Record the number of “M” s on your table.
d) Leaving the decayed M&Ms on the paper towel (just push them to the side),
pick up the un-decayed M&Ms, shake them up and spill them out. Record the
number of un-decayed M&Ms left.
e) Repeat step c until all of your nuclei (M&Ms) have decayed.
f) Do three trials of this activity and calculate your averages. Please round
your averages to the nearest whole number. Once you reach zero, stop
calculating the averages.
Data Table
Trial 1
Trial 2
1
2
3
4
Total
60
60
60
60
240
Copy your
totals here
and then
calculate the
averages.
Turn
0
1
2
3
4
5
6
1
60
Trial 3
2
3
4
Total
1
2
3
4
Total
60
60
60
240
60
60
60
60
240
Trial 1
Total
Trial 2
Total
Trial 3
Total
Avg.
240
240
240
240
Graph the
numbers in
the average
column.
Remember to
round your
averages to
the nearest
whole
number!
7
8
9
4. Make a graph of number of M&Ms (y-axis) versus turn number (x-axis) on the
graph paper provided.
The graph you just made is an excellent example of a half-life curve. It starts out
with a large number and rapidly goes down. Use your graph to answer the questions.
Questions
1. Each turn represented a half-life; did half of your M&Ms always decay each
turn?
2. Why didn’t half of your M&Ms decay each turn?
3. After 1 half-life (1 turn) you would expect ½ of your M&Ms to decay and ½ of
your M&Ms to be un-decayed. In other words, after 1 half-life, you have ½ of
the original sample still un-decayed (radioactive). Fill out the table below to find
out the fraction of radioactive material left after each successive half-life:
# of half – lives
Fraction of radioactive material left
1
2
3
½
½x½=¼
½ x ½ x ½ = 1/8
4
5
6
4. What happens to the amount of radioactive nuclei left after each half-life?
5. Do you think the radioactive nuclei will ever all go away? Why or why not?
6. What happens to the nuclei that decay – do they disappear or turn into a new
element? Explain your answer!
Really Cool Fact
One really interesting thing about the half-life graph that you made is that the
graph for any radioactive element would look the same! The only thing that changes
is the length of the turns. Some elements have half-lives that can be measured in
seconds and others take billions of years!
7. The table below lists some common radioactive substances, along with their
half-lives. For each substance, calculate how long it would take for ten halflives to pass. (Hint: The half-life tells you the length of each turn. Multiply this
by the number of half-lives.)
Element
Astatine-218
Half-life
Time you would have to
wait for 10 half-lives
Practice
Half-Life Problems
2 seconds
Bismuth-210
6 days
Carbon-14
5,730 years
Uranium-238
4,500,000,000 years
Use what you have learned to solve these problems.
NOTE: I have done #8 for you (yes, I am a great guy!).
8. What is the half-life of a 100g sample of nitrogen-16 that decays to 12.5g of
nitrogen-16 in 21.6 seconds?
1
2
3
Solution: 100g
50g
25g
12.5g
It took three steps (half-lives) to go from 100g to 12,5g. The total time was 21.6
seconds. 21.6/3 = 7.2 seconds. The half-life of Nitrogen–16 = 7.2 seconds.
9. All isotopes of technetium are radioactive, but they have widely varying halflives. If an 800g sample of technetium-99 decays to 100g of technetium-99 in
639,000 years, what is its half-life?
10. A 208g sample of sodium-24 decays to 13g of sodium-24 within 60 hours.
What is the half-life of this radioactive isotope?
11. If the half-life of iodine-131 is 8.1 days, how long will it take a 50g sample to
decay to 6.25 g?
12. The half-life of hafnium-156 is 0.025 seconds. How long will it take a 560g
sample to decay to one-fourth its original mass?
13. Chromium-48 has a short half-life of 21.6 hours. How long will it take 360g of
chromium-48 to decay to 11.25 g?
14. Potassium-42 has a half-life of 12.4 hours. How much of an 848g sample of
potassium-42 will be left after 62 hours?
15. Carbon-14 has a half-life of 5730 years. How much of a 144g sample of
carbon-14 will remain after 17,190 years?
16. The half-life of uranium-238 is 4,500,000,000 years. How long would it take
until only 1/16th of a sample was left un-decayed?
17. A radioactive substance has decayed until only 1/32nd of its nuclei are still
radioactive. How many half-lives have passed?