Week 9/Th: Lecture Unit '22' VSEPR
advertisement

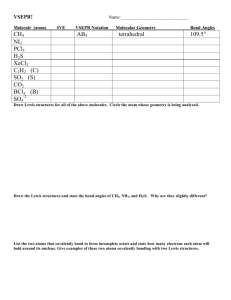
Week 9/Th: Lecture Unit ‘22’ VSEPR Unit 21: Lewis Structures, II -- multiple bonds -- “resonance” Unit 22: VSEPR -- Rules -- Electron Arrangements -- 3D shapes Unit 23,24: Structure & Orbitals -- Molecular Shapes and Orbitals Issues: Test Results, Grade Estimation No Homework this week © DJMorrissey, 2o12 Week 9/Th: Exam Results CEM 141 Fall 2o12 Exam 1 350 CEM 141 Fall 2o12 Exam 2 250 200 150 100 50 0 10 30 50 70 90 110 130 150 170 Score Count Mean Median Mode SD Range Minimum Maximum Exam 1 2344 109.0 110 110 31.7 170 10 180 © DJMorrissey, 2o12 Number of Students Number of Students 300 Exam 2 2226 105.5 107.5 120 34.8 170 10 180 300 250 200 150 100 50 0 10 30 50 70 90 110 130 150 170 Score Grade Point Estimator Week 9/Th: VSEPR Rules, part 1 The ValenceShellElectronPairRepusion (VSEPR) Theory is applied to Covalent Bonds: Electron Sharing, Main group elements Ø focus on the ‘central atom’ Ø construct Lewis electron-pair diagram Ø multiple bonds ?? Ø mutual repulsion of all remaining pairs in 3D à “arrangement” à Only five electronic arrangements on central atom Ø Plus one more rule … © DJMorrissey, 2o12 Week 9/Th: VSEPR – 1st Three “Arrangements” Top Row Hydrides : Lithium Hydride Li – H Beryllium Hydride H – Be – H Boron trihydride H BH3 Methane CH4 © DJMorrissey, 2o12 B H H H H C H H Week 9/Th: VSEPR – All Five “Arrangements” Plus: F F P F © DJMorrissey, 2o12 F F PF5 SF6 F F F S F F F Week 9/Th: VSEPR & Lone Pairs Top Row Hydrides : Methane H H C H CH4 H Ammonia H N H NH3 H Water H2O Tetrahedron, 109.5o Trigonal Pyramid O H H Hydrogen Fluoride H–F © DJMorrissey, 2o12 Bent 104.5o Week 9/Th: VSEPR Rules, part 2 The ValenceShellElectronPairRepusion (VSEPR) Theory is applied to Covalent Bonds: Electron Sharing, Main group elements Ø focus on the ‘central atom’ Ø construct Lewis electron-pair diagram Ø multiple bonds ?? Ø mutual repulsion of all remaining pairs in 3D à “arrangement” à Only five electronic arrangements on central atom Ø LonePair&LP > LP&BondingPair > BP&BP à“molecular shape” à Only eleven 3D shapes for central atom in molecule © DJMorrissey, 2o12 Week 9/Th: VSEPR – Multiple Bonds O N O N2O4 O Bent O NO2- O N N O N NO2 O N N Linear O N N2O Two parts Trigonal Planar O N NO3- O Bent O O N O © DJMorrissey, 2o12 Trigonal Planar Week 9/Th: VSEPR All Rules The ValenceShellElectronPairRepusion (VSEPR) Theory is applied to Covalent Bonds: Electron Sharing, Main group elements Ø focus on the ‘central atom’ Ø construct Lewis electron-pair diagram Ø treat multiple bonds as a single unit Ø mutual repulsion of all remaining pairs in 3D à “arrangement” à Only five electronic arrangements on central atom Ø LonePair&LP > LP&BondingPair > BP&BP à“molecular shape” à Only eleven 3D shapes for central atom in molecule © DJMorrissey, 2o12 Week 9/Th: VSEPR – Large Coordination F F SF6 F S F F F IF5 XeF4 XeF2 ? © DJMorrissey, 2o12 F I F F F F F Xe F F Xe F F F Week 9/Th: VSEPR – All eleven Structures Total # of Electron Pairs à ß Total # of Lone Pairs 2 0 3 Linear Trigonal planar 4 5 6 Tetrahedral Trigonal bipyramidal Octahedral 1 X Bent Trigonal pyramidal See-saw Square pyramidal 2 X X Bent T-shaped Square planar 3 X X X Linear (no examples) © DJMorrissey, 2o12 Week 9/Th: VSEPR – Examples in Notes 1) Thionyl chloride: SOCl2 2) Boron trichloride: BCl3 3) Xenon tetrafluoride: XeF4 [see analog BH3] [done] 4) Bromine trifluoride: BrF3 5) Nitrate anion: NO3- © DJMorrissey, 2o12 [done] Week 9/Th: Nitrogen Triiodide Demo We wrote the Lewis Structure for ammonia, NH3, and we used that to identify its VSEPR structure as a trigonal pyramid. We also have the concept that elements lower in the periodic table with have analogous structures to those containing top-row elements. Nitrogen triiodide, NI3, should have the same structure (Lewis, VSEPR) However, there is a substantial size difference between H & I Covalent Radii H 25 pm N 65 pm I 108 pm N H H H I N I I Nitrogen triiodide is highly strained and tends to decompose … © DJMorrissey, 2o12