Ammonia Gas Absorption - Chemical Engineering
advertisement

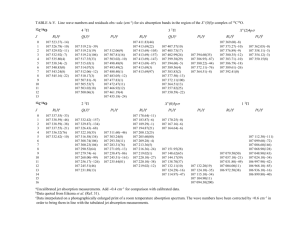
Ammonia Gas Absorption by Oscar D. Crisalle Professor Chemical Engineering Department University of Florida crisalle@che.ufl.edu Revision 12: September 24, 2013 .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 1 CONTENTS 1 Introduction 4 2 Experiment 1: Absorption of Ammonia (N H3) 5 3 Operational Information 6 4 Thermodynamic Phase-Equilibrium 9 5 Henry's Law 11 6 Gas Densities 12 7 Colburn's NTU Equation 13 7.1 Number of transfer units for gas-phase controlled transfer . . . . . . . . . . . . . . . . . . . . . . 13 7.2 Height of a transfer unit for gas-phase controlled transfer . . . . . . . . . . . . . . . . . . . . . . . 14 7.3 Remarks on the NTU Equation (7) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14 8 Overall Mass Transfer Coecient 15 9 Interpretation of the Absorption Factor 16 10 Interpretation of NTU and HTU 17 11 Characteristics of Flow in the Column 18 11.1 Hold-up time (also called residence time) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18 11.2 Number of hold-ups . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18 12 Scrubbing Eectiveness 19 .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 2 13 Theoretical Expectations 21 14 Measurement of N H3 Gas Compositions 22 15 Rotameter: Water Flow Measurement 23 15.1 Liquid solvent rotameter (RTM): water . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 23 15.2 Measurements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24 16 Rotameters: Gas Flow Measurements 16.1 Gas feed-line rotameters (RTM): 25 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 25 16.2 Measurements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26 (N H3 + N2 ) 17 Experimental Details 27 18 Experimental Procedures 30 18.1 Start-up and normal-operation procedures . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30 18.2 Shut-down procedure 31 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19 Anticipated Experimental Problems 32 20 Objectives 33 .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 3 1 INTRODUCTION l This experiment investigates the properties of gas absorption equipment where a gaseous solvent mixed with air or Nitrogen is absorbed by dissolution into a water stream l There l The are two gas absorption experiments in the Unit Operations lab Experiment 1 Absorption of ammonia in water Experiment 2 Absorption of carbon dioxide in water focus of this lab is the Experiment 1 which deals with the absorption of ammonia in water. l It is MANDATORY to read the chapter entitled Gas Absorption in reference [3] before carrying out this experiment. l Remark: Gas absorption is also referred to as gas scrubbing, or gas washing. .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 4 2 EXPERIMENT 1: ABSORPTION OF AMMONIA (N H3 ) Counter-current absorption packed tower Nomenclature l Solute: Raffinate (N2 + NH3) Vout, yout Solvent (W) Lin, xin l Feed Ammonia Carrier Gas: Nitrogen l Solvent: Rotameter (N H3) Water (N2) (W ) Thermodynamic Equilibrium Ammonia Sensor Feed-solvent Phase (or raffinate phase) h N2 + NH3 W Equilibrium y = mx* N2 + NH3 W + NH3 y x* Extract-solvent Phase (or extract phase) Vin, yin Rotameter Feed (N2 + NH3) Lout, xout Extract (W + NH3) Assumption: Nitrogen is insoluble in W .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 5 3 OPERATIONAL INFORMATION l Column available m Height of the column : m Internal m Type diameter: 800 mm 100 mm of packing: Standard 6 mm Raschig rings l Density m The liquid water stream has a higher density than the N2 + N H3 gas stream. That is why the liquid stream is fed from the top. l Insolubility m We are making the assumption that N2 is insoluble in W. This is only an approximation. l Nonvolatility m We W ) is nonvolatile at the temperature of are making the assumption that the solvent ( the experimental conditions .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 6 OPERATIONAL INFORMATION l Continuous m Two and dispersed phases phases form inside the column: a CONTINUOUS phase and a DISPERSED phase. When a column is started up, it is FIRST lled with ONLY the gas. This denes the continuous phase. SECOND, the liquid stream is introduced, and it becomes the dispersed phase. m Making the gas the continuous phase creates more interfacial area than when the liquid is the continuous phase (because the gas is constrained to reside in bubbles) l Flooding by the water phase m Occurs when the upward force exerted by the gas is sucient to prevent the liquid from owing downward m The 100% ooding velocity of the gas stream can be determined for a given inlet liquid stream ow: n Set the gas feed ow to a value that oods the column (water level is at the top of the packing surface) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 7 OPERATIONAL INFORMATION l Operation m Start m Set at 0% ooding gas ow 100% ooding conditions m Progressively reduce the gas ow rate until a value where zero ooding (packing base level) occurs m Space below packing base must be covered with water to prevent gas escape via the liquid exit pipe l In the case of unpacked column m For 0% ooding, the height of the column at which the inlet of the feed gas stream is located should be considered as the base level m For 100% ooding, the height of the column at which the inlet of the water solvent stream is located should be considered as the top level .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 8 4 THERMODYNAMIC PHASE-EQUILIBRIUM l Some N H3 from the gas phase (Nitrogen + N H3) absorbs into the N H3) establishing a phase equilibrium after a suciently long time l The liquid phase (Water + x∗ mole fraction of N H3 in the liquid phase at equilibrium y mole fraction of N H3 in the gas phase at equilibrium equilibrium mole-fraction (x∗) of absorbed N H3 is known as the solubility of N H3 in water y N2 + NH3 P Gas Phase Liquid Phase W + NH3 x* T .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 9 THERMODYNAMIC PHASE-EQUILIBRIUM l The solubility of m The l It N H3 in water is high at room temperature and 1 atm of pressure solubility increases with pressure and decreases with temperature x∗ to y , but we are N H3 and nitrogen) is possible to derive a relationship relating of low values of m Focus: y (use of dilute mixture of mostly interested in cases dilute gas-phase regime .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 10 5 HENRY'S LAW l At constant T and at equilibrium, the amount of solute gas that dissolves into the liquid is proportional to the partial pressure (yP ) (x∗) of the solute gas in the gas phase: i.e, yP = H(T )x∗ (1) y = mx∗ (2) or where m= where P H (T ) P (3) is the operating pressure of the column and H (298.15) = 0.885 atm l Valid ( only for dilute solutions and when the solute W) l Temperature does not react with the solvent dependence is given by the van't Ho equation H (T ) = H T and for (N H3) N H3/water ref exp −C 1 1 − ref T T (4) solution C = 3670 K l Resource: http://www.henrys-law.org .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 11 6 GAS DENSITIES l Standard Temperature and Pressure m NIST-STP: National Institute of Standards and Technology T = 20 C = 68 F = 293.15 K m IUIPAC-STP: P = 1 atm = 1.01325 bar = 14.696 psi International Union of Pure and Applied Chemistry T = 0 C = 32 = 273.15 K P = 0.9869 atm = 1 bar = 14.504 psi m Gas rotameter manufactures usually use dierent standards. Refer the instrument manual for details. l Density Models m Density of dry air (model using the specic air constant ) ρAir = where P P Specific T RAir (5) is the feed gas pressure and Specific RAir = 286.689 J/ (kg · K) = 2.829 × 10−3 m3 · atm/ (kg · K) m Density of ammonia gas ρN H3 ≈ M WN H3 17.031 ρAir = ρAir = 0.587ρAir M WAir 29 (6) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 12 7 COLBURN'S NTU EQUATION 7.1 Number of transfer units for gas-phase controlled transfer NOG mxin − yout A mx − yin in ln = 1 mx − y 1−A in out 1− 1− A mxin − yin (7) and L0in A = mVin0 (8) where yout yin xin A Vin0 = Vin/Across L0in = Lin/Across m solute mole fraction in the ranate [dimensionless] solute mole fraction in the feed [dimensionless] solute mole fraction in the solvent [dimensionless] absorption factor solvent molar supercial velocity [dimensionless] 2 lbmole/ min · ft2 lbmole/ min · ft equilibrium constant for dilute solution [dimensionless] feed molar supercial velocity .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 13 COLBURN'S NTU EQUATION 7.2 Height of a transfer unit for gas-phase controlled transfer HOG = lh (9) is the height of the packed bed [ft] 7.3 Remarks on the NTU Equation l Only l It h NOG (7) valid for dilute feed streams is assumed that the solute mole fraction in the solvent is zero, i.e., l Number of transfer units is expressed in terms of concentration in the gas phase m Solubility l As xin = 0 of ammonia in water is high a result, the dominant resistance to diusion (mass transfer) resides within the gas .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 14 8 OVERALL MASS TRANSFER COEFFICIENT l Overall mass transfer coecient on a gas-phase basis Vin 1 = Vin0 HOGAcross HOG m Interfacial area per unit volume of packing, a, is normally transfer coecient (Denition: Ainterf acial = aAcross ) Ky a = l Mass-transfer left lumped with the mass resistance: inverse of the mass-transfer coecient Resistance = l Correlation (10) 1 Ky a (Solve using least-squares regression) Ky a = c1Lcin2 Vinc3 =⇒ ln (Ky a) = c4 + c2 ln Lin + c3 ln Vin where m See (11) (12) c4 = ln c1 tutorial on the Excel function LINEST ( Least-squares regression using LINEST in Excel ) posted in the course web site. You can also use MATLAB or OCTAVE m Verication of correlation n Carry out at least one additional experimental run (Ky a) correlation − (Ky a)run Error = 100% (Ky a)run (13) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 15 9 INTERPRETATION OF THE ABSORPTION FACTOR l The Absorption Factor A is dened as the ratio of the local slope of the operating curve to that of the equilibrium curve A= Slope of operating curve Slope of equlibrium curve (L0in/Vin0 ) L0in = = m mVin0 l For the transfer of N H3 from the gas phase (V ) to the liquid phase (L), the driving force y − y ∗ should be positive, which implies the operating line should be above the equilibrium line. m This is possible when A>1− m Hence, N H3 from A is met. the absorption of the above condition on mxin − yout mxin − yin the gas phase into the liquid phase occurs only when l Observations m When A < 1 − desorption ( or m When A = 1 − mxin − yout mxin − yin mass transfer occurs from the liquid phase into the gas phase stripping ) mxin − yout mxin − yin there is no net mass transfer between the gas and liquid phases .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 16 10 INTERPRETATION OF NTU AND HTU l Number of transfer units (NTU) m Depend on the value of m Measure m If yout desired for a given yin of the diculty of separation a high-level of absorption (separation) is desired, then a larger number of NTUs is needed l Height of a transfer units (HTU) m Depend on the mass transfer coecient and the gas ow rate m Measure m HTU of the separation eectiveness of the packing for the species being absorbed is proportional to the resistance to mass transfer HOG = m HTU 1 Vin Ky a Across (14) is small (lower resistance) when n There n There is a high rate of interface mass transfer is a large amount of interfacial area (better contact) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 17 11 CHARACTERISTICS OF FLOW IN THE COLUMN 11.1 Hold-up time (also called residence time) thold−up = thold−up QW ater VP ack VP ack QW ater (15) hold-up time (residence time) (min) water (solvent) ow rate (GPM) packed volume (gal) 11.2 Number of hold-ups Nhold−up = Nhold−up tSS tSS thold−up number of hold-ups (dimensionless) time to steady-state (min) (16) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 18 12 SCRUBBING EFFECTIVENESS l Denition of Scrubbing Eectiveness := l Formula N H3absorption into the liquid solvent N H3entering via the feed stream Overall rate of Rate of Derivation: Dene Y = and use the mass-balance result Vout = yout yin (18) 1 − yin Vin: 1 − yout = (17) yinVin − youtVout yinVin 1 − yin Vin yinVin − yout 1 − yout = yinVin yin (1 − yout) − yout (1 − yin) yin − yout = yin (1 − yout) yin (1 − yout) yout 1− 1−Y yin = = yout 1 − yinY 1 − yin yin = .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 19 l Calculation m 1. From experimental data Information needed: yin and yout Procedure: n Calculate yout yin Y exp = n Calculate m 2. From NTU predictions Information needed: yin , m, Vin , Lin , exp (19) 1 − Y exp = 1 − yinY exp and (20) NOG Procedure n Calculate A and nd the value of Y NOG = by solving (graphically or numerically) from A ln A−1 n Calculate pred = m 3. Prediction error n Calculate the prediction error 1+ 1 (A − 1) Y A 1−Y 1 − yinY (21) (22) PE PE = pred − exp exp 100% (23) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 20 13 THEORETICAL EXPECTATIONS l The mass transfer should increase for larger l The mass transfer should more strongly aected by the gas-feed ow rate solvent ow rate Lin/Vin ratios (Vin) than by the (Lin) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 21 14 MEASUREMENT OF l BACHARACH Ammonia N H3 gas GAS COMPOSITIONS monitor: Model AGMSZ l Measures ammonia gas in the range of 25 to 10, 000 ppm l Detector Type: Single pass, non- dispersive infrared l Sensitivity: 25 ppm l Operating Temperature: 32 to 122°F (0 to 50°C) l Accuracy: ±10 ppm ± 10% from 0 − 1000 ppm of reading l Operating Humidity: 5 to 90% RH, non- condensing l Response Time: 9 to 30 seconds, depend- ing on tube length and gas concentration .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 22 15 ROTAMETER: WATER FLOW MEASUREMENT 15.1 Liquid solvent rotameter (RTM): water l Dwyer Rate-Master Flowmeter: Coarse Fine Model RMC l2 rotameters (coarse and ne adjust- ments) l Measurement m Coarse: m Fine: units Gallons per Minute (GPM) Gallons per Hour (GPH) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 23 ROTAMETER: WATER FLOW MEASUREMENT 15.2 Measurements l Reading: QRT M,solvent l For (graduation mark on the scale) ne rotameter Qsolvent(GP H) = QRT M,solvent l For coarse rotameter Qsolvent(GP H) = l Mass 60 min QRT M,solvent 1 hr (24) (25) ow rate Qsolvent(lb/hr) = ρsolventQsolvent(GP H) (26) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 24 16 ROTAMETERS: GAS FLOW MEASUREMENTS 16.1 Gas feed-line rotameters (RTM): Coarse Fine (N H3 + N2) l Dwyer Rate-Master Flowmeter: Model RMB l2 rotameters (coarse and ne adjust- ments) l Measurement units: Standard Cubic Feet per Hour (SCFH) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 25 ROTAMETERS: GAS FLOW MEASUREMENTS 16.2 Measurements l Reference (from the instrument manual) m Tref = 70 F = 21.111 C = 294.261 K m Pref = 1 atm = 1.01325 bar = 14.696 psi l Reading: RRT M (oat position on the scale) s Qf eed(SCF H) = RRT M Pf eed Tref (K) Pref Tf eed(K) Qf eed(lb/hr) = fSCF H→CF H ρf eed Qf eed(SCF H) where the conversion factor fSCF H→CF H (27) (28) is fSCF H→CF H = Pref Tf eed(K) Pf eed Tref (K) (29) .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 26 17 EXPERIMENTAL DETAILS l Because N H3 is highly soluble in water, one must operate at low solvent-to-feed rations (i.e., low L/V) to prevent complete mass transfer to the liquid (dominant resistance to mass transfer is in the gas phase) l Measure m Take the N H3 composition in the feed and ranate stream using the sensor. repeated measurements to obtain statistical averages. m Report concentration values at steady state (take great care of ensuring steady state is attained) l Measure the volumetric mass ow rates of the the feed and solvent streams using the rotameters and convert the readings to mass and molar ow rates [lbmol/hr]. m Then calculate the corresponding uxes need in Colburn's equation by dividing by the cross sectional area of the column. l Determine the ooding velocity of the feed stream for each solvent ow rate considered. .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 27 EXPERIMENTAL DETAILS l Run l Note the column at various values of the absorption coecient. also that at steady state the N H3 composition in the extract stream is estimated from the following expression (obtained from a mass balance) xout = where Vout yinVin − youtVout Lout (30) is obtained from yet another mass-balance calculation as Vout = 1 − yin Vin 1 − yout (31) l Assumptions m The N H3/N2 mixture behaves as an ideal-gas mixture m The solvent stream contains no absorbed m The extract stream contains no absorbed nitrogen N H3 on inlet to the column .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 28 EXPERIMENTAL DETAILS l Example of a data record m Consider Run recording your data in a table T similar QRT M,f eed QRT M,solvent to the one shown below yin yout ∆P ··· 1 2 3 4 5 .. . .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 29 18 EXPERIMENTAL PROCEDURES 18.1 Start-up and normal-operation procedures 1. Plug the power cable of the N H3 sensor into the outlet. 2. Open the valve for the water outlet (extract) line. 3. Open fully the feed-gas cylinder (N2 + N H3). Set the regulator pressure to the desired setting. (Note: the pressure should not exceed 40 psi) 4. Adjust the rotameters to allow the desired ow of feed gas ow into the column. 5. Open the water inlet valve and adjust the rotameters to obtain the desired solvent (water) ow into the column. DO NOT allow water into the column when the feed ow rate is zero, as water might enter into the feed gas line until it reaches and damages the N H3 sensor. 6. Switch on the dierential pressure gauge to measure the pressure drop across the column. 7. Open the appropriate sensor gas-valves to measure the concentration of N H3 in either the feed stream or the ranate stream. 8. During operation always maintain the water level at the bottom of the column below the feed-gas inlet to prevent feed gas escaping the column through the extract-stream opening. .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 30 EXPERIMENTAL PROCEDURES 18.2 Shut-down procedure 1. Turn o the solvent ow into the column by closing the water valve completely. 2. Close completely the valve of the feed-gas cylinder (N2 + Important note: 3. N H3). DO NOT turn o the feed gas before turning o the water. Wait for the feed-gas and water ow into the column to go to zero on the rotameter scales; then turn o the rotameters. (Closing the inlet valves of water and feed gas before turning o the rotameters helps to release the pressure in the inlet lines in shut-down mode) 4. Switch o the pressure gauge. 5. Unplug the Ammonia sensor from the power outlet. .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 31 19 ANTICIPATED EXPERIMENTAL PROBLEMS l Incorrect l Not start-up sequence (creates the wrong dispersed phase) waiting suciently for steady-state conditions l Experiments l The may not have been carried out at isothermal conditions feed gas may escape through the extract outlet when a small amount of water level is not maintained at the extract outlet .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 32 20 OBJECTIVES NOTE: Address ONLY the objectives identied by the instructor (ignore the rest) l Objective 1 m Characterize the ooding condition of the column at each liquid ow rate by determining the ooding gas ow rate. Plot the ooding gas ow rate as a function of (a) liquid ow rate, (b) the liquid-to-gas molar ow ratios, and (c) the absorption factor l Objective 2 m Determine the hold-up time and the number of hold-up times needed to achieve steady- state as a function of absorption factor l Objective A A. 3 m Characterize the dependence of NTUs and HTUs on the absorption factor A: (a) Plot the NTU and HTU results as a function of A, (b) Plot the natural logarithm of the NTU and HTU results as a function of A. .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 33 OBJECTIVES l Objective 4 m Characterize n Find the mass transfer coecient a correlation for the mass transfer coecient and verify the correlation using additional experimental test n Establish the dependence of the mass transfer coecient on the absorption factor A: (a) Plot the mass transfer coecient as a function of A, (b) Plot the natural logarithm of mass transfer coecient as a function of A. Superimpose on these plots the correlation curve l Objective m Plot the the scrubbing eectiveness as a function of A as a function of the NTUs. l Objective m Plot 5 6 the ranate and the extract compositions as a function of A .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 34 REFERENCES [1] Hodgman, C. D., Weast R. C., and Selby, S. M., editors, CRC Handbook of Chemistry and Physic s, 42nd edition. CRC Press, Cleveland Ohio, (1961). [2] Geankoplis, C. J., Transport Processes and Unit Operations, Third Edition. Prentice-Hall Inc., Englewood Clis, NJ (1990). (Chapter 10) [3] McCabe, W. L., J. C. Smith, and P. Harriet, Unit Operations of Chemical Engineering, Fifth Edition. McGraw-Hill, Inc., New York, NY (1993). (Chapter 22) [4] Foust, A. S., L. A. Wenzel, C. V. Clump. L. Maus. and L. B. Anderson, Operations. John Wiley & Sons, New York, 1960. page 552. Principles of Unit [5] Onda, K., Takeuchi, H., and Okumoto, Y, Mass transfer coecients between gas and liquid phases in packed columns, [6] Treybal, R. E., Journal of Chemical Engineering of Japan, Vol 1, pp. 5662 (1968). Mass Transfer Operations, 2nd. ed., McGraw-Hill, New York (1968). .......................................................................................................................................................... Ammonia Absorption Rev 08 - 04/15/2013 Page 35